 At a symposium in Cape Town, South Africa, an orthotist demonstrated his technique for treating balance issues in patients with peripheral neuropathy using ankle foot orthoses (AFOs), and a team of researchers theorized about evidence-based concepts that could help explain his findings.
At a symposium in Cape Town, South Africa, an orthotist demonstrated his technique for treating balance issues in patients with peripheral neuropathy using ankle foot orthoses (AFOs), and a team of researchers theorized about evidence-based concepts that could help explain his findings.
By Cary Groner
A symposium at the recent 2017 World Congress of the International Society for Prosthetics and Orthotics (ISPO)1 shed new light on a long-vexing question: How can clinicians help patients with peripheral neuropathy improve their balance using ankle foot orthoses (AFOs)?
People with severe neuropathy typically have trouble with balance and gait, partly because they receive little or none of the sensory information the rest of us get from the plantar surfaces of the feet. That input helps most people manage the body’s ever-swaying center of mass, much of which is controlled at the ankle. If the ankle lacks the necessary plantar feedback, the whole kinetic chain is destabilized, and people must compensate with movements at the knee, hip, and trunk. The result is often difficulty maintaining balance while standing and walking.
Unfortunately, there’s a paucity of research on interventions that might address the problem, so clinicians have largely been left to their own devices. AFOs are often prescribed for conditions that affect joint stability and positioning, pressure distribution, and neuromuscular issues, and research suggests they can be effective for adjusting various gait parameters.2 A 2010 systematic review in the Journal of Prosthetics & Orthotics (JPO), moreover, reported that although rigid AFOs seemed to facilitate static balance, dynamic balance problems were usually better treated with more flexible leaf-spring devices.2 But unfortunately, little of this research is directly applicable to patients with neuropathy, whose concerns include the risk of falls.
LER has previously reported that, although AFOs can be helpful for improving balance, their utility for preventing falls is open to question—and, for most patients with impaired balance, falls present the biggest danger.3 Part of the issue is that balance itself is achieved via a complex system of visual, cognitive, motor, vestibular, proprioceptive, and neurological functions at both the central and peripheral levels.3 The goal is to maintain the body’s center of gravity within the base of support while a patient is stationary (static balance), and to control the center of mass in dynamic situations such as walking (dynamic balance).2
So, when Paul Charlton, MSc, a senior orthotist at Peacocks Medical Group in Newcastle Upon Tyne, UK, showed Stefania Fatone, PhD, BPO(Hons) some of the results he was getting by treating patients with neuropathy using rigid AFOs, she took notice. Fatone, an associate professor in physical medicine and rehabilitation at Northwestern University in Chicago, put together a team of researchers to examine Charlton’s methods and consider a theoretical underpinning that could help explain his findings.
The ISPO symposium was the result; it included Charlton and Fatone, as well as Cleveland Barnett, PhD, a senior lecturer in biomechanics at Nottingham Trent University in the UK, and Nerrolyn Ramstrand, PhD, BPO(Hons), an associate professor of prosthetics and orthotics at Jonkoping University in Sweden and coauthor of the 2010 review in JPO.

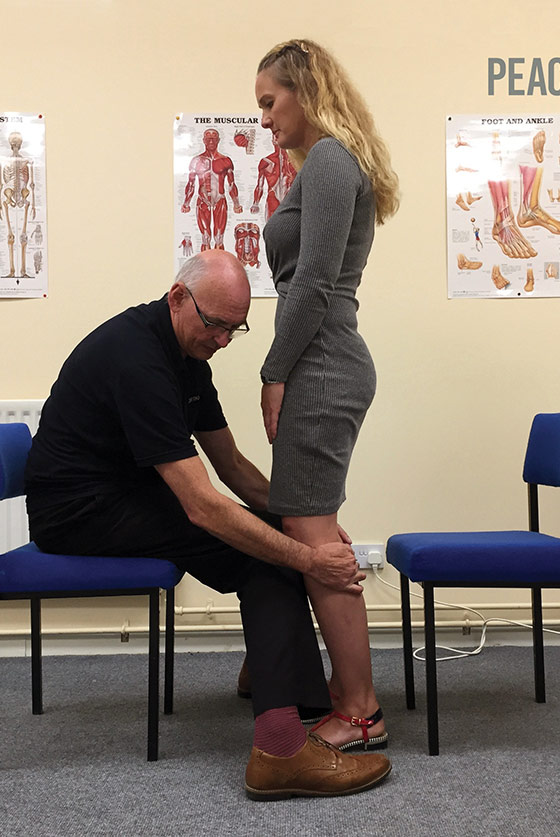
Paul Charlton, MSc, and a healthy volunteer demonstrate his technique for assessing balance in patients with peripheral neuropathy. He starts by asking patients to stand still, to establish a baseline (left). He then has the patient sit. He sits opposite them and positions his knees just below theirs, at the top of their tibias. He grasps the back of the patient’s calves near the top and pulls them toward him, so the tibias are slightly inclined. He has the patient stand as he does this—essentially mimicking the effects of a rigid AFO (center and right). (Photos courtesy of Paul Charlton, MSc.)
Assessment
“Paul’s videos of his patients’ results are really startling, because you see dramatic changes,” Fatone said. “We wanted to try to elucidate the mechanism by which he was getting those effects.”
Charlton’s four presented case studies comprised different diagnostic pathologies, Fatone explained, but all the patients had peripheral neuropathy in common.
“Initially, we wanted to talk about orthotic function with regard to a diagnosis; if you have a stroke you do this, if you have Parkinson’s you do this, if you have MS [multiple sclerosis] you do this,” she continued. “But Paul’s approach was less about diagnosis and more about evaluating the person’s sensation and how that influenced balance. He developed a way to assess whether the peripheral neuropathy is the primary contributor to balance problems.”
Charlton explained his process (see images above) to LER.
“Balance is so complex that as a clinician, I have to start by determining which mechanism is affecting it,” he said. “I can’t address all of those causes, but I know I can have an impact on peripheral neuropathy. So, I want to start by confirming that condition and assessing their proximal control.”
He starts by asking his patients to stand still; it’s difficult for many of them, but those who do it more easily are then asked to move their feet closer together, which increases the postural challenge. Those who manage that reasonably well are then asked to close their eyes.
“That baseline isn’t just for me,” Charlton said. “It also gives the patient a better understanding of their level of impairment, of how poor their balance is. If they can stand still with their feet together and their eyes closed, the [rigid] AFO intervention I use is probably too aggressive.”
Once he’s established the baseline, Charlton has the patients sit. He sits opposite them and positions his knees just below theirs, at the top of their tibias. He then grasps the back of the patient’s calves near the top and pulls them toward him, so the tibias are slightly inclined forward and clamped between his hands and his knees. He then has the patient stand as he does this—essentially mimicking the effects of a rigid AFO.
Patients with peripheral neuropathy are usually much steadier during this process than when standing on their own, he said.
“They’ll say, ‘Well of course I’m steadier, you’re holding me still,’ but I’m only holding them below the knees,” Charlton said. “The point is that if I make their ankle rigid this way, then they can more effectively use the balance mechanisms at their knees, hips, and proximally, because they’re on a stable base. My proposition is that their balance is actually normal, apart from that distal segment. When their ankle is stabilized, they use their knees more effectively, and they become less dependent on their eyesight to maintain their balance.”
Charlton emphasized the importance of the forward tibial incline, as well.
“It doesn’t have to include ankle dorsiflexion,” he said. “I usually use heel lifts to pitch them forward to what I feel is optimal, which is usually around seven degrees. My feeling is that the ground reaction force [GRF] vector should be posterior to the center of the knee joint through midstance. I think what we’re doing with alignment is putting more appropriate demands on the neuromuscular system; you want a flexion moment at the knee, so you’re using your quadriceps and gluteals to keep yourself up.”
Charlton explained, further, that the tibial incline angle varies with the length of the shank; if the ideal is to position the knee joint 1 cm ahead of the GRF vector, a long-shanked person would require a smaller inclination angle to achieve that position than someone with a shorter shank.
“That’s my hypothesis, anyway, for which I have no proof,” he said with a laugh.
But there’s proof, and then there’s proof. Does Charlton have randomized controlled trials? No. But he does have compelling videos of patients walking with dramatically more normalized gait.
“I can show what I see clinically, anyway,” he added. “Then I ask the academics to prove it.”
Constraints
At the ISPO symposium, the academics did their best to comply.
“My role was to look at things from a theoretical perspective,” said Cleveland Barnett. “I’m a biomechanist by trade, so I was trying to explain Paul’s results using biomechanical principles and motor control theories. My orientation is dynamical-systems theory, which is based on the constraints-led approach.”
Briefly, the approach notes humans have “motor abundance”—that is, lots of ways to achieve a given task. If you want to scratch your nose, there are essentially infinite ways in which you can bring the tip of your fingernail to the skin atop your alar cartilage, involving positional changes at the hand, wrist, elbow, and shoulder. A “constraint,” in this context, is just what it sounds like—the placing of a restriction on one or more aspects of that movement menu.
“If you have peripheral neuropathy, you’ll have poor control at the ankle, but your more proximal control may be very good,” Barnett said. “If you fix the ankle joint with an AFO, you reduce the need for neuromuscular control at that joint. When we’re learning something, we are usually quite rigid and stiff; then, as we get better at it, we loosen up, allow for more variation. So, if someone finds a given task difficult, you can help by imposing a constraint, which helps them explore how to coordinate their other movements. Paul freezes the ankle joint with an AFO, which allows patients to stop worrying about controlling the ankle and use the control they have at the knee and hip. In watching his videos, I’ve never seen interventions work so quickly.”
Stef Fatone concurred.
“If you can reduce a task to its least complex form and block redundant degrees of freedom, then people can learn the task more easily,” Fatone said. “In Paul’s approach, we’re using constraint theory to reduce the degree of freedom at the ankle joint and let the person control what they can easily control, where they have feedback and sensation. We’re not necessarily training them, but we’re taking away the thing they can’t control and letting them work with everything else more effectively.”
The literature
Part of the challenge involved in conducting a broad theoretical inquiry into the problem is that what scant literature there is tends to be uneven in its findings. As LER has reported previously, for example, researchers aren’t always certain whether AFOs work by affecting gait mechanics, sensory feedback, or both.3-5
“It’s really difficult to divorce those two things,” Fatone acknowledged. “If someone was completely paralyzed, with no sensation, and the AFO provided a corrective response to postural perturbations, you could say, ‘OK, we’re definitely seeing a mechanical input.’ I suppose you could test that using a nerve block, but does the benefit of what you might learn from that outweigh the risks? It’s a tough study to do. We know that our population of neuropathy patients has diminished sensation, but the amount and kind of sensation loss varies. Is it proprioceptive, touch-receptive, mechanoreceptive— what, exactly, is diminished? It’s very hard to discriminate. The orthosis could be acting in different ways, and teasing those out in any given study is extremely challenging.”
Fatone argues the lack of appropriate studies doesn’t mean researchers are flying completely blind, however.
“We haven’t found any studies that investigated rigid AFOs directly in patients with peripheral neuropathy,” she said. “The study that would test the scenarios Paul is enacting in his clinic, with the exact kind of people he works with, hasn’t been done. But we do have studies that have investigated rigid AFOs more broadly, in mixed populations, and we can infer certain things from that—
including that it’s not unreasonable that rigid AFOs would facilitate static balance in those with neuropathy, because they’ve done it in other populations.”
For example, some studies have shown that AFOs have positive effects on gait regularity6 and postural stability5 in neuropathic patients, and one found that auxiliary sensory cues improved automatic postural responses in those with diabetic neuropathy.7 However, a 2016 systematic review reported that in the absence of randomized controlled trials, the literature offered little consistent evidence of efficacy.8
Since coauthoring the 2010 review mentioned earlier, Nerrolyn Ramstrand has continued to monitor the literature, and she told LER that it offers little justification for changing her conclusions.
“We said that the rigid AFO was good for static balance, and that a leaf-spring design was better for dynamic balance, and I think the literature still supports those conclusions,” Ramstrand said. “I’m a researcher, I’m interested in evidence-based practice, and from a purely academic point of view, there’s no evidence to support the theory that rigid AFOs will help neuropathic patients with dynamic balance. But the modern definition of evidence-based practice includes both the literature and individual clinical experience, and obviously Paul can’t ignore his own experience. You have to consider that, as well as the patient’s desires, in addition to the literature.”
Ramstrand noted, too, that there’s an inherent tension between the demands of a research environment, in which conclusions can be drawn only in the presence of experimental uniformity across a cohort of participants, and the practical exigencies of a clinic, where treatments must be customized according to the clinician’s experience and the patient’s needs.
“People haven’t been using rigid AFOs in neuropathy patients because they didn’t think they were useful,” Ramstrand said. “But now we may need to go back and say, ‘OK, there are people having success with these, so why is that?’”
She noted, too, that clinicians may need to distinguish pathology from clinical presentation in their patient evaluations.
“We have to stop talking about pathologies with this cookbook approach to orthotic prescription,” she said. “Instead, we need to ask what’s behind the patient’s clinical presentation—muscle strength, range of motion, all those things. Then we can consider what might be the most appropriate orthosis for that condition: How well are its mechanical properties suited to managing the clinical presentation of that patient? Different orthosis designs can achieve the exact same goal. There’s lots to figure out, and I think we’ve really just started to identify some of the problems.”
Barnett agreed.
“Instead of talking about individual AFOs, you may say that this type of AFO may have this type of effect,” he said. “But beyond that, I think one of Paul’s points is that a given patient’s treatment depends very much on how that patient presents. Two people with different diagnoses can have similar functional deficits, and for the deficits Paul was seeing, I think rigid AFOs are definitely beneficial; they clearly allow people to do things they couldn’t do before. Never mind the intricacies of the biomechanics: If they’re able to walk farther or faster, it’s likely they’re going to feel better about themselves. If they’re able to walk to the shops unaided with a rigid AFO rather than a flexible one, it’s better for their physical and mental health.”
Improvement?
In Barnett’s presentation, he suggested that once patients become comfortable and better balanced with rigid AFOs, it might be possible to loosen them up and allow more of their natural flexibility.
“Long-term, my feeling is that if you put a constraint like that on someone and they make progress, it’s a question of how long you let them use that before you try to challenge them again,” he said. “The theory would suggest that if you slowly allow an increase in articulation, reduce the rigidity of the AFO, they would start to learn to control that to some extent. They might find it difficult at first—their neuropathy might limit them—but they could gradually get used to it.”
Paul Charlton respectfully differed, however.
“I’m slightly skeptical of that,” he acknowledged. “If the patient has a range of motion in a joint that they can’t control, then I see benefit from fixing them in an optimum position. If you give them more movement, it’s fine if they have the control to use it, but if they don’t, you’re not doing them any favors. It depends on the pathology and the patient’s potential. Some peripheral neuropathies can improve, such as in Guillain-Barré syndrome, or in patients with an insult to the central nervous system such as a stroke or a brain injury. For such patients to progress, they should be given more range of movement by reducing the stiffness of the AFO. But in patients where there isn’t that potential for improvement, I doubt if it would work.”
Fatone emphasized the degree to which Charlton customizes his AFOs, for that matter. In his presentation, only one of the cases involved a patient with diabetic peripheral neuropathy, for example, and he fabricated a specially designed external orthosis for her.
“In that case, he made an AFO with a rigid, lateral carbon-fiber strut that attached to the base of her diabetic footwear,” Fatone explained. “The others had more typical AFOs that went into the shoe and had plastic directly in contact with the foot, but in her case he used a completely different strategy to achieve the same control, so that it didn’t change the pressure experienced by her foot. The only problem was that she started walking so much more that her shoes weren’t able to protect her from the increased activity level, and she started to have tissue breakdown in her feet. So you can never win, right?”
Charlton acknowledged another problem, as well: His patients are sometimes intimidated by their new capabilities.
“Quite often the patients have developed a lot of compensations in their gait,” he said. “Then, suddenly, they’re in a situation where they get a real heel strike. They go from that to foot flat, and their tibia is pushed forward, and it’s a shock to their system. They’re walking more normally, but it’s scary, it takes practice, and I really have to persuade them to stay with it. But if I can convince them to do that, a more normal gait comes very quickly.”
Charlton emphasized that his approach isn’t out of the reach of most clinicians.
“None of this is high-tech,” he said. “It’s simple, it’s practical, and it uses existing technology. And it provides a framework as to the patients for whom it might best be applied.”
Down the road
Fatone said that Charlton’s work, along with the admittedly vague messages from the literature, are pointing a way forward for future research.
“I think we’re evolving a theoretical framework for the kinds of hypotheses we want to test,” she said. “We know what the gaps in the literature are, and by looking at those, as well as at the work that’s been done well, it allows you to design a better research project. That, coupled with the fact that we now have a good hypothesis based on a theoretical understanding of what we think is going on, puts us in a better position to design a study to answer the question of how Paul is getting such good results.”
Cary Groner is a freelance writer in the San Francisco Bay Area.
- Fatone S, Charlton P, Barnett C, Ramstrand N. Balancing act: Exploring clinical, theoretical and evidence-based perspectives in the optimization of balance using orthoses in peripheral neuropathy. Presented at the International Society of Prosthetics & Orthotics World Congress, Cape Town, South Africa, May 2017.
- Ramstrand N, Ramstrand S. AAOP state-of-the-science report: the effect of ankle foot orthoses on balance — a systematic review. J Prosthet Orthot 2010;22(10):P4-P23.
- Groner C. Can AFOs help prevent falls? LER 2012;4(8):16-22.
- Rao N, Aruin A. The effect of ankle foot orthoses on balance impairment: single case study. J Prosthet Orthot 1999;11(1):15-19.
- Rao N, Aruin A. Automatic postural responses in individuals with peripheral neuropathy and ankle foot orthoses. Diabetes Res Clin Pract 2006;74(1):48-56.
- Richardson JK, Thies SB, DeMott TK, Ashton-Miller JA. Interventions improve gait regularity in patients with peripheral neuropathy while walking on an irregular surface under low light. J Am Geriatr Soc 2004;52(4):510-515.
- Rao N, Aruin A. Auxiliary sensory cues improve automatic postural responses in individuals with diabetic neuropathy. Neurorehabil Neural Repair 2011;25(2):110-117.
- Paton J, Hatton AL, Rome K, Kent B. Effects of foot and ankle devices on balance, gait and falls in adults with sensory perception loss: a systematic review. JBI Database System Rev Implement Rep 2016;14(12):127-162.









Amazing. It sounds counter intuitive but it isn’t. The approach is nearly the opposite of AFOs without sensory loss. It makes perfect sense to me. I always felt at a loss when working a peripheral neuropathy clients who had devastating sensory loss.
We are always looking to get knee extension near midstance and then rocker into toe off. Without sensation the person cannot balance themselves and falling backwards is the consequence of trying to get knee extension at midstance with a vertical tibia.
Paul Charlton finds the individual’s sweet spot with the tibia/ankle joint in dorsiflexion. The person then learns to balance themselves by leaning into the solid ankle. Fine tuning with lifts makes perfect sense as variations in sensory loss are different from person to person. Once they learn to lean forward and are confident the ankle is stable they can fire their quads and gluts to obtain a vertical and stable pelvis to balance the trunk. My compliments to Mr. Charlton for thinking outside the box and even going against standard protocols and figuring this out. I am impressed.