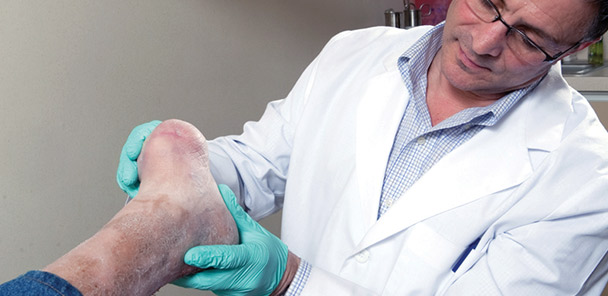
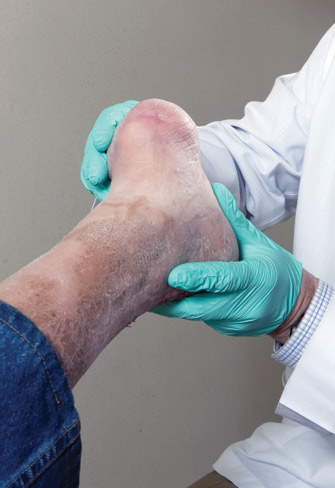
In patients who undergo a minor foot amputation following a diabetic foot ulcer, severe peripheral arterial disease is the primary risk factor for subsequent major amputation, which underscores the importance of early detection and intervention for PAD in this population.
By Vincent S. Nerone, DPM, Kevin D. Springer, DPM, Darren M. Woodruff, DPM, and Said A. Atway, DPM, AACFAS
Patients with diabetes mellitus undergo about 75% of all foot amputations,1 and lower extremity amputation (LEA) greatly increases morbidity and mortality. A retrospective study by Aulivola et al, for example, found a mortality rate of 8.6% at 30 days, 69.7% at one year, and 34.7% at five years after major LEA.2
Risk factors for amputation in diabetic patients have been well documented and include peripheral arterial disease (PAD), loss of protective sensation, lower extremity ulceration, previous lower extremity amputation, insulin dependence, impaired glucose control, microvascular complications, and history of stroke.3-5
The rate of lower extremity reamputation in diabetic patients has also been well documented.6-8 A 10-year observational study by Izumi et al demonstrated reamputation rates of 26.7%, 48.3%, and 60.7% at one, three, and five years, respectively, after a diabetic patient’s initial lower extremity amputation.6 However, there are few studies documenting risk factors for reamputation, especially risks for major amputations (above or below knee amputation) after minor foot amputations (any amputation of the foot distal to the ankle).7-10 We performed the following study to determine risk factors for major LEA (i.e., limb loss) after a minor foot amputation in diabetic patients.
Methods
In the present retrospective study, we identified 172 patients at our institution that had undergone a minor foot amputation and at least one subsequent LEA between January 1, 2000, and January 1, 2011. Surgeons from the vascular surgery, orthopedic surgery, and podiatric surgery services did the amputations. We reviewed patient charts and operative reports to determine the exact surgical procedure performed.
Minor amputations included partial toe amputation, complete toe amputation, partial or full ray resection, or proximal foot amputation (transmetatarsal, Lisfranc, Chopart, or Symes). Below knee and above knee amputations were considered major amputations (limb loss). The laterality of the subsequent amputations was not recorded.
The indications for amputation included soft tissue infection, osteomyelitis, or gangrene. The presence of soft tissue infection was determined by imaging (plain films, magnetic resonance imaging, or nuclear imaging) as well as physical examination. The presence of osteomyelitis was diagnosed using the imaging modalities previously described and often confirmed by bone culture, either preoperatively or intraoperatively. The presence of gangrene was determined by physical examination.
Study exclusion criteria included no documentation of hemoglobin A1c (HbA1c) at the initial amputation, no documentation of peripheral vascular status (documentation of palpable pulses, noninvasive vascular studies, or angiography), and the use of minimal debridement with no true amputation. Nine patients were excluded from the present study, leaving 163 for inclusion. One patient was excluded because the patient had no record of vascular status, four were excluded because they had no record of HbA1c, and four were excluded because a debridement rather than a true amputation was performed.
We separated the 163 patients into two groups. The minor LEA group consisted of patients who underwent an initial minor LEA, one or more subsequent minor LEAs during the follow-up period, and no major LEA. The major LEA group consisted of patients who underwent an initial minor LEA and had a subsequent major LEA during the follow-up period. We then examined the differences between these two groups and looked for possible risk factors associated with a subsequent major LEA.

Figure 1. Healed Chopart amputation in a diabetic male patient aged 47 years with no evidence of PAD.
Each patient chart was thoroughly reviewed for age, gender, race, diabetes mellitus type (type 1 or 2), amputation dates, exact procedure performed, total number of amputations, interval between each amputation, interval to major amputation, follow-up duration, HbA1c, presence of chronic kidney disease (CKD), treatment with hemodialysis or peritoneal dialysis, history of kidney or kidney-pancreas transplantation, smoking status (current, quit, or never), severity of PAD (determined by palpation of pulses, noninvasive vascular studies, or angiography), and whether peripheral vascular intervention was performed (e.g., stent, angioplasty, or peripheral bypass). These factors were chosen because they were risk factors for amputation and reamputation cited in previous studies.3-10 The presence of peripheral neuropathy was not well documented in the patient charts.
The presence of PAD was only recorded if patients had undergone noninvasive studies or angiography. All patients with documentation of palpable dorsalis pedis and posterior tibial pulses were considered to have normal arterial inflow (no PAD). Patients with nonpalpable or weakly palpable pedal pulses all underwent noninvasive studies or angiography. Patients considered to have mild to moderate PAD had either biphasic Doppler signals or an ankle brachial index (ABI) between .5 and .9.
Patients in the severe PAD group had either monophasic Doppler signals at the level of the ankle or an ABI less than .5, previous lower extremity bypass, or were diagnosed by a vascular specialist based on angiogram findings. The diagnosis of diabetes, HbA1c level, presence of CKD, end-stage renal disease (ESRD), need for dialysis, smoking status, and peripheral vascular disease level were assessed at the initial amputation. Vascular intervention at any time was recorded.
Statistical analyses
A 2-sided p-value less than .05 was considered statistically significant. Continuous and categorical variables from the demographic data are presented as the mean +/- standard deviation and frequency with percentages, respectively. Unpaired t tests were used to compare the major and minor LEA groups for patient age and HbA1c. Chi-square tests were used to compare the categorical variables between the major and minor LEA groups, except for smoking status and transplantation status. Fisher’s exact test was used for these two variables, because of the small number of patients. Analysis of variance was used to compare the PAD groups.
Results
Demographic data. There were 298 reamputations (111 patients) in the minor LEA group, and 211 reamputations (52 patients) in the major LEA group. The average patient age was 53.18 +/- 11.11 years and 56.23 +/- 10.76 years for the minor and major LEA groups, respectively. The percentages of male patients in the minor and major LEA groups were 58.56% (n = 65) and 65.39% (n = 34), respectively. The percentage of patients who were white was 58.56% (n = 65) in the minor LEA group and 53.85% (n = 28) in the major LEA group. The percentage of patients with type 2 diabetes mellitus was 88.29% (n = 98) and 86.54% (n = 45) in the minor and major LEA groups, respectively. The percentage of patients who were alive at the time of the study when the data were recorded was 82.88% (n = 92) and 71.15% (n = 37) in the minor and major LEA groups, respectively. The average follow up was 3.65 years for the minor LEA group and 4.37 years for the major LEA group.
Risk factors for major amputation. No statistically significant differences were found between the minor and major amputation groups for HbA1c (9.03 vs 8.92, p = .806); smoking status (51.35% [n = 57] vs 44.23% [n = 23]; p = .679); CKD (57.66% [n = 64] vs 55.77% [n = 29]; p = .820); ESRD requiring dialysis (16.22% [n = 18] vs 19.23% [n = 10]; p =.634); kidney or kidney-pancreas
transplant (9.01% [n = 10] vs 7.69% [n = 4]; p = .520); or peripheral vascular intervention (20.72% [n = 23] vs 35.69% [n = 17]; p = .098).
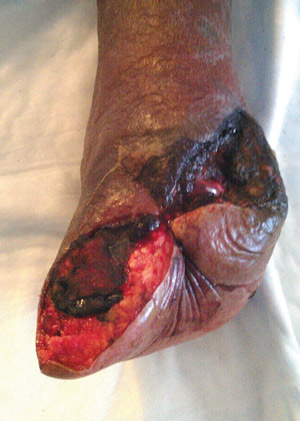
Figure 2. This diabetic man, aged 59 years, with severe PAD underwent an open Chopart amputation, then progressed to a Syme amputation, which eventually healed.
Comparing the vascular status between groups, 59.46% (n = 64) of the minor LEA group and 21.15% (n = 10) of the major LEA group had no PAD, 15.32% (n = 18) and 7.69% (n = 5) had mild to moderate PAD, and 22.23% (n = 29) and 71.15% (n = 33) had severe PAD, respectively. The prevalence of major LEA after initial minor foot amputation was significantly associated with the presence of peripheral arterial disease (p < .001).
Interval to amputation. The interval to major amputation after the initial minor foot amputation was statistically significantly longer (1180.9 days) for the no PAD group than the mild to moderate PAD group (591 days) or the severe PAD group (559.6 days). No statistically significant differences between groups were found with regard to the intervals from the initial minor amputation to the second, third, fourth, or fifth amputations.
Discussion
Our study examined the characteristics of diabetic patients who underwent an initial minor foot amputation and then subsequently underwent either a minor LEA or major LEA. We believed that patients in the major LEA group would have a greater incidence of previously described risk factors than patients in the minor LEA group. The results of the study, however, partially refuted our hypothesis and showed the only significant risk factor for diabetic patients with a minor foot amputation subsequently requiring a major LEA was severe PAD. Additionally, the presence of PAD significantly decreased the time interval to major LEA after an initial minor LEA.
Previous studies have documented risk factors for initial amputation in diabetic patients. The Seattle Diabetic Foot study identified PAD (ABI < .8 or transcutaneous oxygen measurement < 50 mm Hg), loss of protective sensation, lower extremity ulceration, previous lower extremity amputation, and insulin dependence as risk factors.3 Selby et al found that impaired glucose control, longer duration of diabetes mellitus, microvascular complications (retinopathy, neuropathy, nephropathy), and history of stroke were risk factors.4
Our study population of 163 patients did show a high prevalence of PAD (54.6%), impaired glucose control (mean HbA1c of 9%), and CKD (50%), which is consistent with these studies. We did not include neuropathy in our analysis, as this was not consistently documented, but almost all patients did demonstrate some degree of peripheral neuropathy. Duration of diabetes, history of stroke, and evaluation of the microcirculation also were not consistently documented. Impairment of the microcirculation would have been an interesting variable to evaluate, as this has been shown to impair wound healing, but measurement of transcutaneous oxygen or skin perfusion pressure was not routinely performed.11

Figure 3. This diabetic man, aged 79 years, with severe PAD had an open midfoot amputation for gas gangrene but went on to below knee amputation.
Although the previous studies were consulted to determine variables for evaluation, direct comparison with the present study is likely not of value as they evaluated initial rather than reamputations.
Few studies have documented risk factors for reamputation after a minor foot amputation.
Skoutas et al followed 121 patients for 18 months after a minor foot amputation and found that 26 required reamputation.7 Risk factors for reamputation included age older than 70 years, concomitant heel ulcerations, and initial amputation involving only one or two toes. Unfortunately, the researchers’ analysis did not distinguish between major and minor reamputation.
Morbach et al performed a recent study prospectively following 247 diabetic patients with foot ulcerations and no previous major amputation.10 Thirty eight patients required a major LEA in a 10-year follow-up period. All but one patient going on to major LEA had evidence of PAD, and 51.4% had severe PAD (ABI < .4). The researchers also found that increased age and hemodialysis were associated with increased risk.
Although not all patients had previous minor foot amputation, this study demonstrates some similarities to our current study, in particular the finding that severe PAD is a significant risk factor for major LEA. Contrary to Morbach’s study, we did not find hemodialysis or increased age to be statistically significant risk factors. This divergent pattern may be due the relatively younger age of participants in our study, which had no individuals in either group older than 70 years. Wide variations among countries have been observed with regard to the risk of amputation, with the lowest prevalence in Japan and the highest in Belgium, France, and Germany.12
Conclusions
In conclusion, the present study demonstrates that, in diabetic patients with minor foot amputations, severe PAD is a significant risk factor for progressing to limb loss. Also, the presence of PAD significantly decreases the interval to limb loss after a minor foot amputation. These findings demonstrate the importance of the early detection of PAD in patients with diabetes as well as early intervention for PAD, which may save limbs.
Consideration should be given to major amputations in patients with severe PAD in which revascularization attempts have failed. This is a difficult subject to discuss with patients but is a conversation that needs to happen as patients with minor amputations due to severe PAD are very likely to end up with a major amputation in a short time. The present study brings to light the fact that severe PAD needs to be considered when the surgeon is deciding on the level of amputation.
Vincent Nerone, DPM, is on staff at Marietta Memorial Hospital in Ohio. Kevin Springer, DPM, is in private practice at Podiatry Center Inc. in Canton, OH. Darren Woodruff, DPM, is currently a third-year resident and Said Atway, DPM, AACFAS, is clinical assistant professor of orthopaedics, Section of Podiatry, at the Ohio State University Wexner Medical Center in Columbus.
1. Toursarkissian B, Shireman PK, Harrison A, et al. Major lower-extremity amputation: contemporary experience in a single Veterans Affairs institution. Am Surg 2002;68(7):606-610.
2. Aulivola B, Hile CN, Hamdan AD, et al. Major lower extremity amputation: outcome of a modern series. Arch Surg 2004;139(4):395-399.
3. Adler AI, Boyko EJ, Ahroni JH, Smith DG. Lower-extremity amputation in diabetes. The independent effects of peripheral vascular disease, sensory neuropathy, and foot ulcers. Diabetes Care 1999;22(7):1029-1035.
4. Selby JV, Zhang D. Risk factors for lower extremity amputation in persons with diabetes. Diabetes Care 1995;18(4):509-516.
5. Pecoraro RE, Reiber GE, Burgess EM. Pathways to diabetic limb amputation: basis for prevention. Diabetes Care 1990;13(5):513-521.
6. Izumi Y, Satterfield K, Lee S, Harkless LB. Risk of reamputation in diabetic patients stratified by limb and level of amputation: a 10-year observation. Diabetes Care 2006;29(3):566-570.
7. Skoutas D, Papanas N, Georgiadis GS, et al. Risk factors for ipsilateral reamputation in patients with diabetic foot lesions. Int J Low Extrem Wounds 2009;8(2):69-74.
8. Kono Y, Muder RR. Identifying the incidence of and risk factors for reamputation among patients who underwent foot amputation. Ann Vasc Surg 2012;26(8):1120-1126.
9. Nather A, Bee CS, Huak CY, et al. Epidemiology of diabetic foot problems and predictive factors for limb loss. J Diabetes Complications 2008;22(2):77-82.
10. Morbach S, Furchert H, Gröblinghoff U, et al. Long-term prognosis of diabetic foot patients and their limbs: amputation and death over the course of a decade. Diabetes Care 2012;35(10):2021-2027.
11. Ogrin R, Woodward M, Sussman G, Khalil Z. Oxygen tension assessment: an overlooked tool for prediction of delayed healing in a clinical setting. Int Wound J 2011;8(5):437-445.
12. Combe C, Albert J, Bragg-Gresham J, et al. The burden of amputation among hemodialysis patients in the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am J Kidney Dis 2009;54(4):690-692.