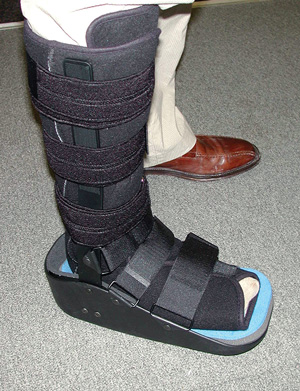
Photo courtesy of James McGuire, DPM, PT, CPed
Increasing body mass index is associated with significantly increased peak plantar pressures regardless of the type of offloading device used. However, it is possible to decrease plantar pressures in some regions of the foot with the use of offloading devices.
By Andrew J. Meyr, DPM, FACFAS; Kelly Pirozzi, DPM, AACFAS; and Matthew R. Wagoner, DPM
Obesity has been deemed a “global epidemic” by the World Health Organization and all evidence points to this problem getting worse before it gets better.1 An interesting investigation on the topic studied two “high risk” elementary schools in Houston, TX.2 The intervention school implemented a yearlong intensive program of daily nutrition education, behavior modification, exercise, and healthy lifestyle counseling for both students and parents, while the control school received no specific obesity-targeted interventions. At the end of the year, the intervention school actually had statistically increased incidences of obesity, weight, and body mass index (BMI) z-scores among students. The authors concluded that “implementation of any school-based obesity intervention programs requires careful planning.” That seems like an understatement.
From a big-picture standpoint, most recent evidence has pointed to a negative effect of obesity on the development, treatment, and outcome of lower extremity pathology. Obese patients are more likely than normal-weight patients to develop symptomatic lower extremity deformity, arthrosis, tendinosis, and other chronic musculoskeletal pathologies.3-10 They are also more likely than their normal-weight counterparts to suffer from fractures of the lower extremity11-13 and experience an increased incidence of postoperative complications when surgery is performed.14-27
The situation becomes increasingly foreboding when considering patients with neuropathy and diabetes. A study by Vela et al28 demonstrated an association between increased weight and increased plantar pressures in the setting of a “normal” foot. They studied the effect of increasing weight in healthy individuals without peripheral neuropathy by loading the front and back pockets of workout vests worn by participants with 9.1 kg and 18.2 kg weights; plantar foot pressures increased 5% and 19% in men and 9% and 25% in women, respectively. These findings were consistent for the various plantar regions (first metatarsal, lesser metatarsal, midfoot, and heel). The authors concluded that reduction in weight could reduce plantar foot pressures and, potentially, the development of ulceration in neuropathic patients.
 Sohn et al also studied the relationship between BMI and diabetic foot ulcers.29 The authors described a J-shaped association between body mass and the development of diabetic foot ulcerations, with obese patients having significantly higher risk for ulceration than normal-weight individuals. Compared with patients with a BMI between 25 and 29.9 kg/m2, the five-year risk of developing a diabetic foot ulcer was 1.4 times higher in those with a BMI between 40 and 44, and 2.1 times higher in those with a BMI of 45 or higher.
Sohn et al also studied the relationship between BMI and diabetic foot ulcers.29 The authors described a J-shaped association between body mass and the development of diabetic foot ulcerations, with obese patients having significantly higher risk for ulceration than normal-weight individuals. Compared with patients with a BMI between 25 and 29.9 kg/m2, the five-year risk of developing a diabetic foot ulcer was 1.4 times higher in those with a BMI between 40 and 44, and 2.1 times higher in those with a BMI of 45 or higher.
Finally, Stuck et al found obesity to be significantly associated with the development of Charcot neuroarthropathy in US Department of Veterans Affairs patients with diabetes, independent of other factors.30 Compared with nonobese patients who did not have neuropathy, those who were obese but did not have neuropathy were 59% more likely to develop Charcot neuroarthropathy.
From an individual practice standpoint, the treatment of lower extremity pathology in the setting of obesity can be a challenging and often frustrating endeavor. The first author of this manuscript has been in private practice for five years in the urban setting of downtown and North Philadelphia. His group’s data indicate that patients presenting to the clinic have a mean BMI of 29.3, meaning that about half of the patients are categorized as “obese” assuming a normally distributed population (Table 1).31 When a patient who is 100 pounds overweight presents to the clinic with a chief complaint of generalized heel pain, it’s not surprising their feet hurt. And when clinicians treat a patient with a plantar diabetic foot wound who is morbidly obese, they often have serious reservations about whether a standard surgical shoe can effectively offload that much extra mass.
We don’t mean to suggest that such clinicians are acting without empathy or sympathy. On the contrary, these are realistic issues we all should be addressing to determine whether we are doing everything we can to appropriately and effectively treat our patients.
It is with this in mind that the first author’s group at the Temple University School of Podiatric Medicine has recently undertaken a series of observational investigations studying obesity and the foot. We have made some interesting observations that have changed the way we evaluate and treat obese patients.
Evaluation of patient BMI
One of our first hypotheses was that physicians in the US could be at risk for a concept or phenomenon that we termed “relative obesity.”32 Although the patients in the first author’s clinic have a mean BMI of 29.3, this doesn’t indicate that a BMI of 29.3 is “normal.” It means that the average patient encountered is overweight and nearly obese. But, because this is the case, it’s possible that clinicians’ perception of what is obese may be skewed accordingly. We may see a patient with a BMI of 41 and think to ourselves that the patient is subjectively and relatively “not that big,” when in fact they are objectively categorized as “morbidly obese.” We suspected this was the case for other clinicians as well.
To test this hypothesis, we had physicians across several levels of clinical experience (student, resident, and practicing physician) provide a visual estimation of a given patient’s BMI without actual knowledge of the patient’s height or weight. We did this across three different clinical situations in which physicians commonly see patients (walking down a hallway, sitting in a treatment chair, and lying in a hospital bed). In our study, the physicians’ visual estimates of patient BMI were inaccurate twice as often as they were correct (77.9% vs 22.1% of the time), and the most common error was to underestimate BMI (48.3% of the time). Assessments were most likely to be underestimated when patients were actually obese or morbidly obese, when they were sitting in a treatment chair, and when they were lying in a hospital bed.
Based on these results, we concluded that physicians are likely to be influenced by the concept of relative obesity if they solely rely on a visual estimation, and that they should be performing an actual calculation of BMI during the patient examination and medical-
decision making processes. We believe if clinicians are unconsciously underestimating patient BMI, they also may be unconsciously underestimating the risks inherent to the treatment of obese patients.
The BOOT data
We have also sought to evaluate whether increasing body mass has an effect on the efficacy of offloading devices that are commonly used in the treatment of acute foot and ankle injuries, postoperatively following elective or traumatic reconstructions, and in the treatment of diabetic foot wounds. In other words, does a CAM (controlled ankle motion) walker have the same level of efficacy in someone who is 5’10”, wears a size 11 shoe, and has a BMI of 22 versus someone of the same height and shoe size who has a BMI of 42?
We have completed the initial portion of our planned series of investigations, which we have termed the “Body mass and Obesity Offloading Trials” (BOOT).33,34
To test our initial hypothesis we replicated the basic structure of a previously published study design investigating the effects of increasing weight on peak plantar pressures,28 making modifications in participant footwear and the amount of added weight. Our participants ambulated down a 12-m hallway in a pair of their own sneakers, a surgical shoe, a CAM walker, and a total contact cast (TCC) with cast shoe. All of our volunteers had normal-range BMIs, which we increased to overweight, obese, and morbidly obese levels by asking them to don military backpacks with adjustable shoulder, chest, and waist straps and loaded with weight evenly that was distributed anteriorly and posteriorly about the trunk for each of the test conditions (Figure 1). For our outcome analysis, we measured mean peak plantar pressures under the heel, midfoot, forefoot, and first metatarsal with an in-shoe pressure measurement system. Comparisons involved foot segments, BMI weight class, and footwear conditions.

Figure 1. Modification of a volunteer’s BMI for the BOOT investigation.
Although this, like any investigation, was not without methodological limitations, we think there were some interesting findings worthy of attention and future investigation. First, we found that increasing BMI did lead to significantly increased peak plantar foot pressures across all plantar foot segments regardless of which, if any, offloading device was used. We also found that it was possible to decrease plantar foot pressures with the use of offloading devices, though this occurred in a manner proportional to body mass. For example, plantar pressure under the first metatarsal for the obese BMI condition was higher for the surgical shoe than the CAM walker, but first metatarsal pressure in the CAM walker was still higher for the obese BMI condition than for the normal BMI condition.
However, this did not occur across the entire plantar surface of the foot in the same manner. We found that the heel was incredibly challenging to offload; none of the offloading devices was associated with a statistically significant difference in heel pressure compared with the sneaker for any BMI category. The offloading devices in all BMI classes were, however, associated with lower midfoot and forefoot pressures than the sneaker, particularly when the ankle was immobilized. Finally, of the offloading conditions in all BMI groups, we found the TCC decreased pressures by the greatest margin, which is consistent with previous studies suggesting that it is the gold standard for offloading.35-46
Future trials in the BOOT series will aim to evaluate the efficacy of offloading devices in individuals whose actual BMI falls within each of the categories, as well the effects of diabetes and peripheral neuropathy on plantar pressures and offloading outcomes.
Scaling theory
Perhaps the most provocative thing we’ve investigated has been the concept of “scaling theory” as it relates to obesity and the diabetic foot. This is a recognized theory within the field of animal biology that proposes, first, that tissue systems of organisms cannot maintain constant morphology as body mass increases, and second, that constraints of locomotion and support differ between large and small organisms. This theory has been primarily applied to evolutionary morphologic changes that have occurred over centuries in the so-called graviportal animals (elephants, rhinoceroses, hippopotami, etc), but we have studied the potential for applying the same concepts to the human foot.47-50 Our supposition is that the musculoskeletal anatomy, physiology, and biomechanics of the lower extremity of an active and healthy patient with a BMI of 22 are not, and probably should not, be the same as the musculoskeletal anatomy, physiology, and biomechanics of the lower extremity of a neuropathic diabetic patient with a BMI greater than 40.
We identified three unique anatomic differences in graviportal animals that may have application to diabetic foot disease in obese patients. The first is differences in the plantar soft tissue structure, specifically the rearfoot, which contains a highly structured network of adipose tissue organized into compartments and reinforced with collagen, retinculin, elastic fibers, and even a cartilaginous rod which has been termed the “sixth ray” or “prehallux” to add support during heel strike. The second is differences in the osseous structure with metatarsal orientation in a thickened and more horizontal tripod configuration. The third is differences in graviportal tendon structure with muscle tendon thicknesses and insertions that appear to be moving away from pronation and supination function and are more consistent with strictly flexion and extension.
At the very least we have found information that correlates with some of the clinical problems that frequently occur with the obese diabetic foot. These include differences in soft tissue structure such as the calcaneal fat pad, osseous structure such as the tripod foot type, and tendinous structures such as the Achilles tendon and equinus. Although we are certainly not comparing our obese patients to these animals studied by others, it is interesting to see how their evolutionary adaptations to increased mass may have potential application to reconstructive surgery of the Charcot foot.
Conclusion
From a big picture standpoint, all of these findings have changed to some degree the way we evaluate and treat obese patients, particularly in the setting of peripheral neuropathy and diabetes. First, we attempt to be very active in the recognition of obesity, as well as patient education. We will specifically discuss with our patients their BMI weight class, the known lower extremity complications of obesity, and how much weight loss is needed to transition into a different BMI weight class. We refer specifically to weight in our discussions of offloading, particularly of plantar foot wounds, and believe that weight loss may have as much of an effect as a specific surgical shoe or offloading boot.
It is also fair to say that we are more likely to immobilize the ankle during offloading in obese patients. Based primarily on the BOOT data, we feel that ankle immobilization may have a significant effect on plantar foot pressures when compared with something like a surgical shoe that does not immobilize the ankle. And finally, we have come to view Charcot as an “anatomic failure.” What we mean by this is that the patient’s normal anatomy has essentially failed them to some degree. We approach a Charcot reconstruction not as an attempt to give the patient back a “normal” foot and ankle structure, but rather one that can withstand the stresses that caused the problem in the first place. This is a matter of expectations, and we believe physician expectations may be just as important as patient expectations.
Andrew J. Meyr, DPM FACFAS, is an associate professor in the Department of Podiatric Surgery at Temple University School of Podiatric Medicine in Philadelphia, PA. Kelly Pirozzi, DPM, AACFAS, is in private practice with Valley Foot Surgeons in Scottsdale, AZ. Matthew R. Wagoner, DPM, is chief resident in the Temple University Hospital Podiatric Surgical Residency Program at Temple University Hospital in Philadelphia.
[1] James WP. WHO recognition of the global obesity epidemic. Int J Obes 2008;32(Suppl 7):S120-S126.
[2] Klish WJ, Karavias KE, White KS, et al. Multicomponent school-initiated obesity intervention in a high-risk, Hispanic elementary school. J Pediatr Gastroenterol Nutri 2012;54(1):113-116.
[3] Weil L Jr. Obesity, feet, and the impact on health care. Foot Ankle Spec 2012;5(3):148-149.
[4] Butterworth PA, Landorf KB, Smith SE, Menz HB. The association between body mass index and musculoskeletal foot disorders: a systematic review. Obes Rev 2012;13(7):630-642.
[5] Runhaar J, Koes BW, Clockaerts S, Bierma-Zeinstra SM. A systematic review on changed biomechanics of lower extremities in obese individuals: a possible role in development of osteoarthritis. Obes Rev 2011;12(12):1071-1082.
[6] Shultz SP, Sitler MR, Tierney RT, et al. Consequences of pediatric obesity on the foot and ankle complex. J Am Podiatr Med Assoc 2012;102(1):5-12.
[7] Bostman OM. Prevalence of obesity among patients admitted for elective orthopaedic surgery. Int J Obes Relat Metab Disord 1994;18(10):709-713.
[8] Fabris SM, Faintuch J, Brienze SL, et al. Are knee and foot orthopedic problems more disabling in the superobese? Obes Surg 2013;23(2):201-204.
[9] Finkelstein EA, Chen H, Prabhu M, et al. The relationship between obesity and injuries among U.S. adults. Am J Health Promot 2007;21(5):460-468.
[10] Wearing SC, Hennig EM, Byrne NM, et al. Musculoskeletal disorders associated with obesity: a biomechanical perspective. Obes Rev 2006;7(3):239-250.
[11] Kessler J, Koebnick C, Smith N, Adams A. Childhood obesity is associated with increased risk of most lower extremity fractures. Clin Orthop Relat Res 2013;471(4):1199-1207.
[12] Chaudhry S, Egol KA. Ankle injuries and fractures in the obese patient. Orthop Clin North Am 2011;42(1):45-53.
[13] Moayyeri A, Luben RN, Wareham NJ, Khaw KT. Body fat mass is a predictor of risk of osteoporotic fracture in women but not in men: a prospective population study. J Intern Med 2012;271(5):472-480.
[14] Armstrong M. Obesity as an intrinsic factor affecting wound healing. J Wound Care 1998;7(5):220-221.
[15] Felcher AH, Mularski RA, Mosen DM, et al. Incidence and risk factors for venous thromboembolic disease in podiatric surgery. Chest 2009;135(4):917-922.
[16] Dodson NB, Ross AJ, Mendicino RW, Catanzariti AR. Factors affecting healing of ankle fractures. J Foot Ankle Surg 2013;52(1):2-5.
[17] Shibuya N, Frost CH, Campbell JD, et al. Incidence of acute deep vein thrombosis and pulmonary embolism in foot and ankle trauma: analysis of the National Trauma Data Bank. J Foot Ankle Surg 2012;51(1):63-68.
[18] Graves ML, Porter SE, Fagan BC, et al. Is obesity protective against wound healing complications in pilon surgery? Soft tissue envelope and pilon fractures in the obese. Orthopedics 2010;33(8):555.
[19] Japour C, Vohra P, Giorgini R, Sobel E. Ankle arthroscopy: follow-up study of 33 ankles—effect of physical therapy and obesity. J Foot Ankle Surg 1996;35(3):199-209.
[20] Bostman OM. Body-weight related to loss of reduction of fractures of the distal tibia and ankle. J Bone Joint Surg Br 1995;77(1):101-103.
[21] Friedman RJ, Hess S, Berkowitz SD, Homering M. Complication rates after hip or knee arthroplasty in morbidly obese patients. Clin Orthop Relat Res 2013;471(10):3358-3366.
[22] Greenblatt DY, Rajamanickam V, Mell MW. Predictors of surgical site infection after open lower extremity revascularization. J Vasc Surg 2011;54(2):433-439.
[23] Dodson NB, Ross AJ, Mendicino RW, Catanzariti AR. Factors affecting healing of ankle fractures. J Foot Ankle Surg 2013;52(1):2-5.
[24] Charalampos GZ, Christensen T, Rigopoulos N, et al. Infection following operative treatment of ankle fractures. Clin Orthop Relat Res 2009;467(7):1715-1720.
[25] Wukich DK, Kline AJ. The management of ankle fractures in patients with diabetes. J Bone Joint Surg 2008;90(7):1570-1578.
[26] Egol K, Tejwani N, Walsh M, et al. Predictors of short-term functional outcome following ankle fracture surgery. J Bone Joint Surg 2006;88(5):974-979.
[27] Still GP, Atwood TC. Operative outcome of 41 ankle fractures: a retrospective analysis. J Foot Ankle Surg 2009;48(3):330-339.
[28] Vela SA, Lavery LA, Armstrong DG, Anaim AA. The effect of increased weight on peak pressures: implications for obesity and diabetic foot pathology. J Foot Ankle Surg 1998;37(5):416-420.
[29] Sohn MW, Budiman-Mak E, Lee TA, et al. Significant J-shaped association between body mass index (BMI) and diabetic foot ulcers. Diabetes Metab Res Rev 2011;27(4):402-409.
[30] Stuck RM, Sohn MW, Budiman-Mak E, et al. Charcot arthropathy risk elevation in the obese diabetic population. Am J Med 2008;121(11):1008-1014.
[31] World Health Organization (2000) Obesity: Preventing and Managing the Global Epidemic. WHO Obesity Technical Report Series 894: World Health Organization, Switzerland.
[32] Wagoner MR, Van JC, Nolan CK, et al. Can foot and ankle surgeons accurately estimate patient body mass index? J Foot Ankle Surg 2014;53(1):36-40.
[33] Pirozzi K, McGuire J, Meyr AJ. Effect of variable body mass on plantar foot pressure and off-loading device efficacy. J Foot Ankle Surg 2014;53(5):588-597.
[34] Pirozzi K, McGuire J, Meyr AJ. A comparison of two total contact cast constructs with variable body mass. J Wound Care 2014;23(7):S4-S14.
[35] Armstrong DG, Nguyen HC, Lavery LA, et al. Off-loading the diabetic foot wound: a randomized clinical trial. Diabetes Care 2001;24(6):1019-1022.
[36] Beuker BJ, van Deursen RW, Price P, et al. Plantar pressure in off-loading devices used in diabetic ulcer treatment. Wound Repair Regen 2005;13(6):537-542.
[37] Caravaggi C, Sganzaroli A, Fabbi M, et al. Nonwindowed nonremovable fiberglass off-loading cast versus removable pneumatic cast (AircastXP diabetic walker) in the treatment of neuropathic noninfected plantar ulcers. Diabetes Care 2007;30(10):2577-2578.
[38] Meyerson M, Papa J, Eaton K, Wilson K. The total-contact cast for management of neuropathic plantar ulceration of the foot. J Bone Joint Surg Am 1992;74(2):261-269.
[39] Armstrong DG, Lavery LA, Bushman TR. Peak foot pressures influence the healing time of diabetic foot ulcers treated with total contact casts. J Rehabil Res Dev 1998;35(1):1-5.
[40] Begg L, McLaughlin P, Manning L, et al. A novel approach to mapping load transfer from the plantar surface of the foot to the walls of the total contact cast: a proof of concept study. J Foot Ankle Res 2012;5(1):32.
[41] Patry J, Belley R, Côté M, Chateau-Degat ML. Plantar pressure, plantar forces, and their influence on the pathogenesis of diabetic foot ulcers: a review. J Am Podiatr Med Assoc 2013;103(4):322-332.
[42] Helm PA, Walker SC, Pullium G. Total contact casting in diabetic patients with neuropathic foot ulcerations. Arch Phys Med Rehabil 1984;65(11):691-693.
[43] Walker SC, Helm PA, Pullium G. Total contact casting and chronic diabetic neuropathic foot ulcerations: healing rates by wound location. Arch Phys Med Rehabil 1987;68(4):217-221.
[44] Birke JA, Novick A, Patout CA, Coleman WC. Healing rates of plantar ulcers in leprosy and diabetes. Lepr Rev 1992;63(4):365-374.
[45] Birke JA, Pavich MA, Patout Jr CA, Horswell RA. Comparison of forefoot ulcer healing using alternative off-loading methods in patients with diabetes mellitus. Adv Skin Wound Care 2002;15(5):210-215.
[46] Sinacore DR, Mueller MJ, Diamond JE, et al. Diabetic plantar ulcers treated by total contact casting. A clinical report. Phys Ther 1987;67(10):1543-1549.
[47] Miller CE, Basu C, Fritsch G, et al. Ontogenetic scaling of foot musculoskeletal anatomy in elephants. J R Soc Interface 2008;5(21):465-475.
[48] Weissengruber GE, Egger GF, Hutchinson JR, et al. The structure of the cushions in the feet of African elephants (Loxodonta africana). J Anat 2006;209(6):781-792.
[49] Fisher RE, Scott KM, Adrian B. Hind limb myology of the common hippopotamus, Hippopotamus amphibius (Artiodactyla: Hippopotamidae). Zool J Linnean Soc 2010;158(3):661-682.
[50] Meyr AJ, Pirozzi K, Creech C. Can scaling theory aid in Charcot foot reconstruction in obese patients? Podiatry Today 2013;26(3):16-20.








