Shutterstock.com #1139100179
Diagnostic Tools and Management
This 2-part series examines the current state of peripheral artery disease. Part 1, which appeared in the August issue, focused on disease burden, risk factors, and clinical presentation. This article reviews diagnostic tools and current management recommendations.
By Aisha Cobbs, PhD
Peripheral artery disease (PAD) is a widespread chronic condition that is most commonly characterized by atherosclerosis, or blockages, in the arteries supplying blood to lower extremities, such as legs, feet, and toes. It is an indicator of systemic atherosclerosis and is a major risk factor for coronary artery disease, myocardial infarction, and stroke. In its most advanced stage, known as critical/chronic limb-threatening ischemia, PAD is a major risk factor for lower extremity amputation.
Properly identifying symptomatic patients can be difficult. Typically, testing for PAD involves a separate office visit to prep the patient for testing, which may involve steps such as making the patient change into a gown. With diabetic patients who may need to come in for nail care every 3 months, encouraging them to come in for another office visit may be challenging. While current tools are addressing the basics of what is needed for diagnosis, raising clinical suspicion of PAD’s presence and then testing for it and treating it are crucial. Earlier diagnosis can improve quality of life and save countless limbs (see “PAD Must Be Caught Early To Prevent Amputation,” page 53).
The first article in this series reviewed disease burden, risk factors, and clinical presentation. This article reviews the diagnostic tools, including newer technologies, and current management strategies for this lethal disease.
Testing For PAD
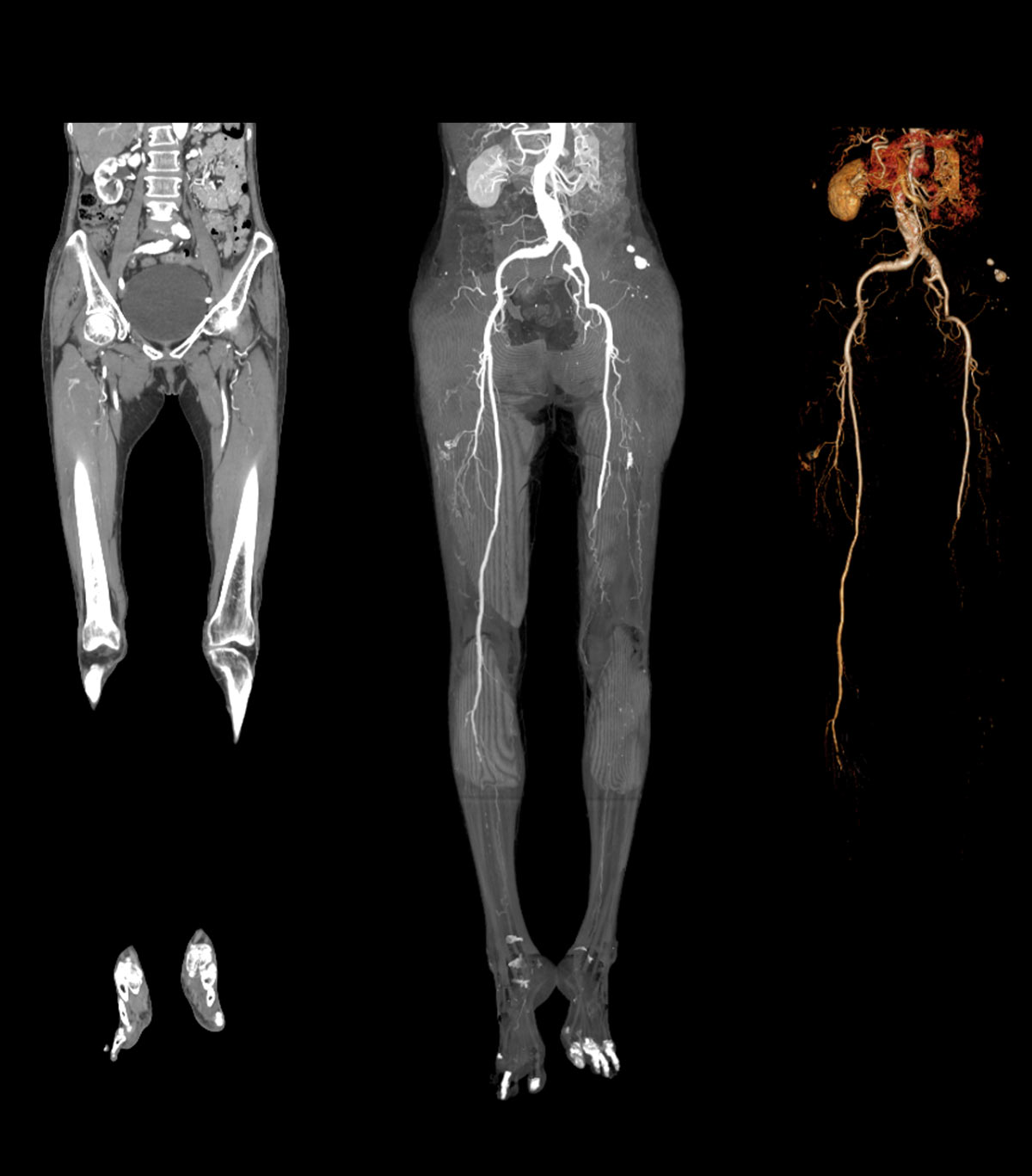
There are several diagnostic tools for PAD. In patients with suspected PAD, the current guidelines from the American Heart Association (AHA) and the American College of Cardiology (ACC) recommend resting ankle brachial testing as the initial diagnostic test.1 Additional testing, such as the toe brachial index (TBI) and pulse volume recordings (PVRs) can also be used to assess arterial occlusive disease. AHA/ACC guidelines recommend Duplex ultrasound or angiography to assess the severity of arterial occlusive disease and to diagnose the anatomical location of stenosis once PAD is confirmed.1
Ankle Brachial Index. The resting ankle brachial index (ABI) is obtained by measuring systolic pressure in arms and ankles while the patient is lying supine.2 The highest of the two blood pressures in the arms is used as the reference blood pressure. To determine the ABI ratio, the reference arm blood pressure is divided by the ankle blood pressure. An ABI ratio ≤ 0.9 is diagnostic of PAD.1
ABI is a non-invasive and reproducible method for assessing arterial occlusive disease in the lower extremities.2 However, there are several limitations with this technology. For instance, diabetes-associated calcification of the arterial walls in the ankles can artificially inflate the ankle blood pressures, or in cases of severe calcification, make it difficult or impossible to get accurate systolic blood pressures.3 Other related conditions, including lymphedema, can also make it difficult to generate blood pressures.4 In some cases, elevated blood pressures due to medial artery stiffness in patients with chronic kidney disease can cause false normal ABI results.5
There are also clinical factors that can make an ABI difficult to perform in a short period of time due to complicated patient preparation procedures. For example, to obtain the most accurate results, it is recommended that the patient be at rest and fully supine for at least 5 minutes before taking measurements.6 In addition, patients need to abstain from tobacco or caffeine consumption for at least an hour prior to ABI testing because these substances can cause inaccurate readings.5,6
Toe Brachial Index. Another limitation of the ABI is that by calculating systolic blood pressures at the ankles, PAD experienced in arteries distal to the ankle (such as the foot or toes) may go undetected since the arteries of the toes tend to retain more elasticity.7 Therefore, some healthcare providers may perform a toe brachial index (TBI), which is like an ABI, except that systolic blood pressures are measured at the toes rather than the ankles. TBIs can be performed in addition to, or as a replacement for the ABIs, especially in patients with non-compressible arteries due to diabetes mellitus and other conditions that reduce arterial wall compressibility.1 The AHA/ACC guidelines recommend TBI to diagnose patients with suspected PAD, if ABI is > 1.40.1 Like ABI, TBI may not be diagnostic of PAD in patients with chronic kidney disease who have a falsely normal ABI due to medial artery calcification.8 A TBI of < 0.6 is abnormal.9
Pulse Volume Recording. Because of the challenges with ABIs described above, healthcare providers will often perform an ABI along with PVRs to assess PAD.1 PVRs are performed by placing a blood pressure cuff at the location where the provider wants to evaluate the blood flow. The blood pressure cuff is then inflated to a pressure that is low enough to avoid restricting arterial blood flow, but high enough to sense changes in blood flow as the artery expands and contracts. The results are captured in a tracing that documents the morphology and amplitude of the recorded waveforms (Figure 1).10
Typically, PVRs are simpler to perform on patients, and when combined with ABI, PVR has an overall 85% diagnostic accuracy rate.11 In addition, PVRs are not susceptible to issues where a patient may have non-compressible arteries, elevated blood pressure, lymphedema, or other complications that may make it difficult to perform an ABI.12
 Duplex ultrasound. A duplex ultrasound test combines imaging and Doppler technology. Ultrasound helps pinpoint the location of the disease and determines the severity and the anatomical location of stenosis.1 While this type of test is highly accurate, it is also very operator-dependent, so it is typically performed in a vascular lab by a Registered Vascular Technologist or other credentialed professional specifically trained in the technology.13
Duplex ultrasound. A duplex ultrasound test combines imaging and Doppler technology. Ultrasound helps pinpoint the location of the disease and determines the severity and the anatomical location of stenosis.1 While this type of test is highly accurate, it is also very operator-dependent, so it is typically performed in a vascular lab by a Registered Vascular Technologist or other credentialed professional specifically trained in the technology.13
Angiography. Angiography is an invasive diagnostic test that involves injecting the patient with contrast dye which allows blockages to be imaged. Typically, this type of test is performed in a vascular lab, and is highly accurate, but involves radiation exposure.14 This test is normally performed prior to a revascularization procedure, to help plan the intervention.1
PADnet. PADnet (Biomedix, St. Paul, MN) combines the clinical value of ABIs and PVRs into a non-invasive physiologic study which can be performed by a medical assistant familiar with how to take blood pressures.15 PADnet generates diagnostic data which can be combined with demographic data and sent to a vascular specialist using an integrated and secure cloud-based service platform. The vascular specialist can then remotely interpret the study within the HITRUST-compliant interpretation website and send his/her interpretation and care plan back to the healthcare provider performing the PADnet test. This innovative approach enables community-based collaborative care to optimize patient outcomes.
Biomedix’s newest technology, PADnet Xpress, uses state-of-the-art plethysmographic technology combined with advanced artificial intelligence and machine learning technologies to enable PAD detection in a 2-minute test that requires patients to remove only their shoes and socks. PADnet Xpress serves as a substitute for pedal pulse detections and other physiologic PAD examinations, with clinical study results confirming excellent diagnostic accuracy.16
PAD Treatment
Treatment options for PAD, which depend on its stage at diagnosis, encompass 4 key domains: behavior changes or lifestyle modifications, nutritional counseling, pharmaceutical interventions, and surgical interventions. The goal of all 4 options is to improve
- blood flow to impede progression of the disease
- quality of life and
- short and long-term prognosis.
- Lifestyle Modifications
Once PAD has been identified, lifestyle modifications are low-risk, low-cost interventions that can help address or slow the progression of the disease.17 Early detection of PAD increases the chance that lifestyles modifications, such as the ones detailed below, can improve patient outcomes and may reduce the need for higher-risk, higher-cost interventions.
Smoking cessation. Smoking cessation is one of the best ways to reduce PAD risk, resulting in an 80% lower risk of disease.18 Even more promising are recent studies indicating that smoking cessation can help reduce the occurrence of PAD symptoms such as claudication.19
Exercise therapy. Exercise therapy is another promising lifestyle modification that can address PAD. Several studies have demonstrated that exercise improves flow-mediated vasodilation.16 In addition, exercise therapy has been shown to increase the distance a patient experiencing intermittent claudication can walk before experiencing discomfort.16
Nutritional Counseling
Nutritional counseling or dietary therapy is a useful tool for addressing PAD or slowing the progression of the disease, as obesity is a significant risk factor for PAD.16 Combining nutritional therapy with exercise is preferred to exercise alone.19 Table 1 provides details on nutritional guidance for a general patient population. Nutritional counseling is often combined with pharmaceutical interventions.
PAD Must Be Caught Early To Prevent Amputation
“Atherosclerosis that affects the limbs is called peripheral artery disease (PAD), the complication that has been shown to cause the most significant long-term disability and economic burden in patients with diabetes. It is estimated that 1 in 3 people 50 years or older with diabetes has PAD, yet millions of people with the disease do not realize they are at risk for losing a limb until it is too late. Someone is given a diabetes diagnosis every 17 seconds in the United States, and every day, 230 Americans with diabetes will undergo an amputation. Each year, approximately 200,000 nontraumatic amputations occur in the United States. African Americans are 4 times more likely to experience diabetes-related amputation than white individuals, and it is estimated that throughout the world, a leg is amputated every 30 seconds; 85% of those amputations are the result of a diabetic foot ulcer. Unfortunately, even as the diabetes and PAD epidemics worsen, people do not recognize the symptoms. Early detection and treatment of PAD is crucial to saving limbs. With timely screening, in-depth assessment of symptoms, and thorough physical examinations, millions of amputations can be prevented. Like cancer, PAD must be caught early and treated to prevent progression and suffering.”23
Foluso A. Fakorede, MD, is a practicing cardiologist and CEO of Cardiovascular Solutions of Central Mississippi as well as cochair of the PAD Initiative for the Association of Black Cardiologists.
Pharmaceutical Interventions
Cholesterol reduction medications such as statins, ezetimibe, and proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors approved to address atherosclerosis in other parts of the body, such as the heart or head, have demonstrated efficacy in PAD.16 Abnormal platelet activation and thrombi development are commonly observed in patients with PAD, so anti-platelet therapy is commonly prescribed as are anticoagulants.16
Endovascular/Surgical Interventions
If lifestyle modifications and pharmaceutical interventions fail, the next line of therapy is revascularization via endovascular or open surgery. Endovascular revascularization for PAD in the lower extremities has progressed rapidly in the last decade with lower morbidity and mortality when compared to open surgery, making it the preferred treatment for many clinical centers.
Revascularization. Atherectomy is an endovascular surgical procedure that removes arterial plaque with a rotating cutter which then deposits the debris into a chamber that can be extracted. Revascularization is indicated in patients with acute limb ischemia and a salvageable limb.1
Amputation. Patients with severe complications of PAD, such as acute limb ischemia, typically develop ulcers that become infected and progress to gangrene. Lower extremity amputation (LEA) is recommended in these circumstances.1 An LEA results in a 200-fold increase that the patient will have to undergo a subsequent LEA at a later date;20 the death rate after an LEA is 48% at 1 year, and 71% at 3 years.21
Conclusion
Our healthcare system needs to undertake more proactive measures to properly identify and address chronic conditions such as PAD in the early stages of the disease progression. Studies have shown that an earlier PAD diagnosis drives a higher success rate of prescribed lifestyle modifications such as smoking cessation, nutritional counseling, and exercise therapy, thereby improving clinical outcomes. Although traditional PAD diagnostic methods have proven to be effective in accurately detecting PAD, there is demand for newer technologies that can assess at-risk patient populations more quickly for proper risk stratification and triage. Such new technologies will play a major role in helping to improve the outcomes for patients who suffer with PAD.
Aisha Cobbs, PhD, is a medical writer from Atlanta, Georgia.
- Gerhard-Herman MD, Gornik HL, Barrett C, et al. 2016 AHA/ACC Guideline on the management of patients with lower extremity peripheral artery disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2017;135(12):e726-e779.
- Firnhaber JM, Powell CS. Lower extremity peripheral artery disease: diagnosis and treatment. Am Fam Physician. 2019;99(6):362-369.
- Abouhamda A, Alturkstani M, Jan Y. Lower sensitivity of ankle-brachial index measurements among people suffering with diabetes-associated vascular disorders: A systematic review. SAGE Open Med. 2019;7:2050312119835038.
- Todd M. Best practice: Doppler assessment in lymphoedema. Br J Community Nurs. 2016;21(12):612-613.
- Casey SL, Lanting SM, Chuter VH. The ankle brachial index in people with and without diabetes: intra-tester reliability. J Foot Ankle Res. 2020;13(1):21.
- WOCN Clinical Practice Wound Subcommittee, 2005. Ankle brachial index: quick reference guide for clinicians. J Wound Ostomy Continence Nurs. 2012;39(2 Suppl):S21-S9.
- Bunte MC, Jacob J, Nudelman B, et al. Validation of the relationship between ankle-brachial and toe-brachial indices and infragenicular arterial patency in critical limb ischemia. Vasc Med. 2015;20(1):23-29.
- Tehan P, Bray A, Keech R, et al. Sensitivity and specificity of the toe-brachial index for detecting peripheral arterial disease: initial findings. J Ultrasound Med. 2015;34(10):1737-1743.
- Sibley RC 3rd, Reis SP, MacFarlane JJ, et al. Noninvasive physiologic vascular studies: a guide to diagnosing peripheral arterial disease. Radiographics. 2017;37(1):346-357.
- Kupinski, AM. (2002). Segmental pressure measurement and plethysmography. Journal of Vascular Technology, 26(1):32-38.
- Lewis JE, Williams P, Davies JH. Non-invasive assessment of peripheral arterial disease: Automated ankle brachial index measurement and pulse volume analysis compared to duplex scan. SAGE Open Med. 2016;4:2050312116659088.
- Benitez E and Sumpio BE. Pulse volume recording for peripheral vascular disease diagnosis in diabetes patients. J of Vasc Diagn. 2015;3:33–39.
- Shabani Varaki E, Gargiulo GD, Penkala S, et al. Peripheral vascular disease assessment in the lower limb: a review of current and emerging non-invasive diagnostic methods. Biomed Eng Online. 2018;17(1):61.
- Willner S. The Role of Imaging in Peripheral Arterial Disease, Peripheral Arterial Disease – A Practical Approach. 2018. Nishtha Sareen and Abhishek Ojha, IntechOpen, DOI: 10.5772/intechopen.75675. Available from: https://www.intechopen.com/books/peripheral-arterial-disease-a-practical-approach/the-role-of-imaging-in-peripheral-arterial-disease.
- Biomedix. PADnet delivers collaborative vascular care for improved outcomes. Available at https://biomedix.com/products/padnet/. Access Sept. 25, 2020.
- Duval S. Diagnostic accuracy of PADnet Xpress® in the detection of peripheral artery disease. Lower Extremity Review. 2020;12(8):39.
- Bevan GH, White Solaru KT. Evidence-based medical management of peripheral artery disease. Arterioscler Thromb Vasc Biol. 2020;40(3):541-553.
- Ding N, Sang Y, Chen J, et al. Cigarette Smoking, Smoking Cessation, and Long-Term Risk of 3 major atherosclerotic diseases. J Am Coll Cardiol. 2019;74(4):498-507.
- Ratchford EV. Medical management of claudication. J Vasc Surg. 2017;66(1):275-280.
- Delaney CL, Smale MK, Miller MD. Nutritional considerations for peripheral arterial disease: a narrative review. Nutrients. 2019;11(6):1219.
- Anand SS, Caron F, Eikelboom JW, et al. Major adverse limb events and mortality in patients with peripheral artery disease: the COMPASS trial. J Am Coll Cardiol. 2018;71(20):2306-2315.
- Swaminathan A, Vemulapalli S, Patel MR, et al. Lower extremity amputation in peripheral artery disease: improving patient outcomes. Vasc Health Risk Manag. 2014;10:417-424.
- Fakorede FA. Increasing Awareness about Peripheral Artery Disease Can Save Limbs and Lives. AJMC. Dec 2018:SP609. Available at https://www.ajmc.com/view/contributor-increasing-awareness-about-peripheral-artery-disease-can-save-limbs-and-lives. Accessed Sept. 20, 2020.
- Delaney CL, Smale MK, Miller MD. Nutritional Considerations for Peripheral Arterial Disease: A Narrative Review. Nutrients. 2019;11(6):1219.