By Larisa Ryskalin, Gabriele Morucci, Paola Soldani, and Marco Gesi
Plantar fasciitis is a chronic and painful disabling condition affecting the inferomedial aspect of the heel, usually extending toward the metatarsophalangeal joints. These authors examined the anatomical and biomechanical substrates of plantar fasciitis with special emphasis on the emerging, though largely neglected, fascial system.
The plantar aponeurosis (PA) is a broad and thick band of connective tissue that arises from the calcaneus and runs longitudinally to attach to the plantar aspect of the forefoot in 3 distinct sites, thus creating 3 distinct components (i.e., medial, central, and lateral bands). The central aponeurotic band is the thickest component. It divides distally into 5 longitudinal digitations, which insert at the level of each metatarsophalangeal joint capsule (Figure 1). Even from a biomechanical viewpoint, the central band of the PA is often cited as the key supporter of the longitudinal arch of the foot, acting like a windlass and preventing arch flattening. Furthermore, it assists subtalar joint supination during propulsion. This band of the PA is therefore the 1 most often involved in the development of plantar fasciitis.
Plantar fasciitis is the most common cause of adult heel pain, accounting for over 1 million physician visits per year in the United States. It primarily affects the PA enthesis and leads to a chronic, painful, and self-limiting condition. It is the most prevalent running-related musculoskeletal disorder among athletes, but it also affects both physically active and sedentary middle-aged and older adults as well as workers whose activity impacts the foot strongly.
Patients usually complain of persistent inferior heel pain. The classic clinical presentation is a sharp stabbing heel pain, usually more intense during the first steps following long non-weight bearing periods (i.e., first standing after rest; typically, the first steps in the morning or after being seated for a long time). The pain tends to reduce gradually once the patient starts walking. However, it can worsen at the end of the day or be exacerbated by prolonged weight-bearing activities.
Despite its high prevalence, the exact etiology and pathological mechanisms underlying plantar heel pain remain unclear. Plantar fasciitis is classically considered an “overuse injury.” Among extrinsic factors, mechanical overload is considered a major contributor to the development of plantar heel pain, presumably because excessive and repetitive tensile strain causes PA micro-traumas. Although the previous literature indicated that micro-tears can result in chronic inflammation, histological evidence does not reveal inflammatory cell invasion or inflammatory markers. Thus, the term “fasciitis” seems misused. Nowadays, PA can more appropriately be deemed “plantar fasciosis,” implying that its pathological signs are more consistent with a chronic degenerative process and tissue degeneration than with an inflammatory response.
Increased tension on the Achilles tendon (AT) resulting from intense muscle contraction is a major mechanical factor in PA overstraining. Previous studies reported a positive correlation between AT loading and PA tension. As a proof of concept, AT tightness is found in almost 80% of patients affected by plantar fasciitis. There was a strong statistically significant correlation in this condition between isolated gastrocnemius contractures or increased gastrocnemius-soleus complex tightness and heel pain severity. Increased tightness in other posterior leg muscles (eg, hamstring) can also induce prolonged forefoot loading and increase repetitive injury to the PA through the windlass mechanism.
Conversely, if AT flexibility is increased through stretching exercises, the pain symptoms of plantar fasciitis decrease. Similarly, restoring muscle strength and flexibility has reduced the symptoms associated with this condition. Both gastrocnemius and soleus muscle stretching exercises are recommended most often in the current literature.
However, the biomechanical importance of the fascial system cannot be neglected. Indeed, fascial tissue cannot be considered a passive bystander in musculoskeletal dynamics, merely enveloping the muscles. Current literature indicates that the central importance of the fasciae in movement and postural control systems is being recognized. Increasing amounts of experimental evidence demonstrate that the fascial system provides a pathway for force transmission, transmitting, and receiving mechano-metabolic information and thus influencing movement perception, peripheral motor coordination, and proprioception. Thus, one can expect that biomechanical abnormalities within the myofascial unit place excessive stress on the PA and ultimately contribute to heel pain and plantar fasciitis.
Therefore, in the present review, after a brief overview of the anatomy of the suro-Achilles-calcaneal-plantar complex of the superficial posterior compartment of the leg, we discuss the complex structural and functional coupling between muscles and fascial tissue, and the possible correlation between myofascial abnormalities, muscle/tendon tightness, and heel pain severity in plantar fasciitis.
FUNCTIONAL ANATOMY OF ACHILLES TENDON
A brief overview of Achilles tendon anatomy
Also termed the calcaneal tendon owing to its attachment to the calcaneus, the AT is one of the thickest, largest, and strongest tendons in the human body. Nevertheless, because it serves as the primary plantar flexing mechanism of the ankle, it is at the greatest risk of rupture, accounting for nearly 20% of all large tendon injuries. Indeed, AT injuries are a very common clinical picture in sports medicine, and they have also become significantly more common within the middle-age, physically active population (i.e., 40–59 years) during the last decade, probably because of the growing popularity of competitive and recreational sports.
Both comparative and ex vivo studies demonstrate the architectural complexity of this tendon. For instance, variations in its twisting structure and the degree of rotation near its insertion on the calcaneal tuberosity have been reported. Reports of the arrangements of the attachments of the AT fascicles to the facets of the calcaneal tuberosity are inconsistent. Also, the relative contributions of the medial and lateral heads of the gastrocnemius and the soleus to the AT can differ among subjects, making the internal AT force distribution even more complicated to understand.
Anatomically, the AT represents the conjoined junction of the triceps surae muscle, which is located at the superficial posterior compartment of the leg (i.e., the calf). This complex consists of the soleus and the medial and lateral heads of the gastrocnemius (Figure 2).
The gastrocnemius comprises 2 heads at its origin, medial, and lateral, which insert proximally in the posterosuperior region of the corresponding femoral condyle. The soleus lies deep to the gastrocnemius and superficial to the muscles of the deep posterior compartment of the leg. It originates from 2 heads, tibial and fibular, which are united by a tendinous arch from which additional fibers arise. The tibial origin is at the inferior border of the soleal line, while the fibular origin is on the posterior aspect of the head and about the upper fourth of the diaphysis. Besides the posterior margins of the tibia and fibula, the soleus can also arise from the surrounding deep fascia of the leg. The medial and lateral intramuscular aponeuroses (originating from the tibia and the fibula, respectively) are continuous with the epimysium of the soleus muscle and penetrate distally into the main muscle belly. Distally, from the thickest point of the muscle belly, a central aponeurotic tendon arises within the soleus. These latter fibers then contribute to the AT, along with those of the gastrocnemius, generally inserting on to the most distal part of the AT while the gastrocnemius fibers insert more proximally (Figure 3).
Anatomical dissection studies show that the AT comprises 3 distinct bundles of tendon fascicles (i.e., subtendons), which are distinguishable at the level of the proximal end of the AT, where the distal aponeuroses of the muscles can be separated from each other. One subtendon arises from the soleus and lies deep, and 2 independent subtendons, which lie superficial, originate from each of the 2 heads of the gastrocnemius. The full incorporation of the tendinous portions of the gastrocnemius and soleus is evident almost 10 cm above the AT calcaneal insertion. However, it has been reported that the soleus can remain separated from the gastrocnemius as far down as the calcaneal insertion. Sometimes there is also a small contribution from the tendon of the plantaris muscle (PM). Often dismissed as a vestigial (accessory) muscle, the PM consists of a small, thin muscle belly and originates from the popliteal surface of the femur. A close connection between the PM tendon and the mid-portion of the calcaneal tendon has been reported in 65% of the adult population.
Unlike other tendons, such as those of the wrist and the hand, the AT lacks a true synovial sheath, but is surrounded by a thin sheath of dense connective tissue called a “paratenon.” Besides providing a certain degree of tendon gliding, the paratenon is an important source of blood supply and nutrition to the tendon. Histologically, Golgi tendon organs, free nerve endings, and Pacinian-like corpuscles were identified in AT tissue samples from healthy pigs, suggesting a role for the tendon in proprioception.
The paratenon is a thick fibrous layer with few elastic fibers, continuous with the crural fascia, as evidenced by an anatomical study of non-embalmed legs from cadavers. The deep investing crural fascia is a layer of connective tissue enclosing the posterior structures of the calf and connected to the paratenon and AT. However, another magnetic resonance imaging (MRI) study suggested that the fascia cruris and the paratenon exist as 2 separate layers around the AT, though they appear less demarcated toward the AT calcaneal insertion, where they seem to fuse with the posterior subcutaneous structures. Another anatomical and radiological study revealed that the mean distance to the confluence of the AT paratenon and the fascia cruris from the postero-superior calcaneal tubercle is 37.3 mm.
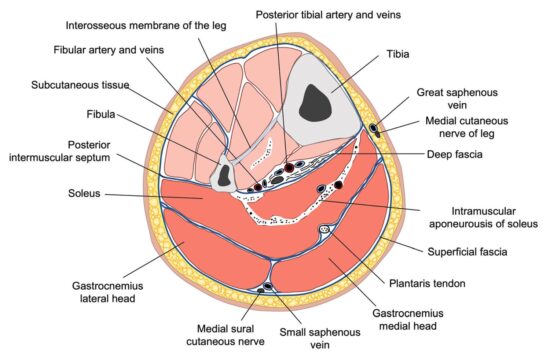
Figure 2. Schematic representation of a cross-section of the lower leg showing all four compartments: anterior, lateral,deep posterior, and superficial posterior. Blue lines indicate the fascial network. Triceps surae muscle components (i.e., medial and lateral heads of the gastrocnemius and the soleus), located within the superficial posterior compartment of the leg, are highlighted in a more intense red.
Achilles tendon pathophysiology
As part of Achilles tendinopathy, tendinosis and tendinitis are defined as clinical pictures of tissue degeneration and inflammation, respectively. Although the term tendinosis is universally accepted since the disease has commonly been described as “degenerative,” the role of inflammation in tendinopathy is controversial and is still much debated. Some authors argue that the term “tendinitis” could be inaccurate and misleading because infiltration and inflammatory cells (such as neutrophils and macrophages) are not found in chronic tendon disorders. In contrast, other studies have demonstrated macrophages, T and B lymphocytes, and increased levels of inflammatory markers such as interleukin-1 (IL-1), IL-6, cyclooxygenase- 1 (COX-1), COX-2, and TGF-β in chronic Achilles tendinopathy. Tissue biopsies from AT patient cohorts show a complex inflammation signature, expressing target molecules from the interferon, NF-κB, STAT-6, and GCR activation pathways, which suggests established (chronic) inflammation and ongoing tissue repair. Recent data suggest that excessive/pathological levels of force can constitute the mechanical stress that triggers tissue microinjury and a local immune system–mediated tendon repair response. This mechanotransduction-mediated dysregulation of the immune response, which ultimately leads to failed healing, is typical of chronic tendinopathic lesions.
Ultrasonography of subjects complaining of Achillodynia demonstrated that AT symptoms could also be associated with increased paratenon thickness with no sign of tendon tissue involvement. Histological examination of the affected tissue showed that metabolic and inflammatory changes within the Achilles paratenon could precede or parallel those within the tendon tissue itself. Acute exercise or excessive loading can result in alterations of AT paratenon structures known as “paratendinitis,” featuring edema, swelling, and lymphocyte infiltration. These alterations result in increased paratenon thickness, which is clearly detectable as increased signal intensity in both sonography and MRI investigations.
Anatomical and structural continuity between the plantar aponeurosis and Achilles paratenon
Functionally, the AT is pivotal in transmitting the contractile forces generated from the triceps surae muscle and producing the ankle plantar flexion torque required for load distribution in the foot. The elastic spring-like properties of the tendon also allow it to store and release energy explosively during walking and running. These functions are closely tied to the morphological and biomechanical relationship between the AT and PA.
A number of randomized control trials have revealed that AT- or calf muscle–stretching exercises can reduce plantar heel pain and increase the range of ankle motion. In 2003, DiGiovanni et al reported statistically significant pain relief at 8-week follow-up in patients affected by plantar fasciitis who were managed with a standard AT-stretching protocol. Another study reported long-term improvement in symptomatic pain following an AT-stretching program in patients with chronic plantar fasciitis. Similarly, patients without previous treatments for plantar fasciitis obtained significant short-term pain relief by using night splints, which keep the foot in a neutral or slightly dorsiflexed position at rest.
Although the effectiveness of AT/calf-stretching exercises and night splints for treating plantar heel pain substantiates a functional link between the AT and PA, anatomical continuity between these structures is still a matter of debate.
In 2002, Milz et al demonstrated a conspicuous bundle of highly oriented trabeculae in the postero-inferior part of the calcaneus. These trabeculae, which were clearly visible in thick resin sections of hindfeet removed from fresh cadavers, appeared as regularly aligned structures oriented along the lines of force transmission from the AT toward the proximal attachment of the PA. Further morphological and histological examination of plastinated slices revealed a band of calcaneal trabeculae running between the AT paratenon and the superficial posterior and inferior calcaneus toward the PA, surrounded by collagens and adipocytes. Continuity of the PA with the AT paratenon was also confirmed in an anatomical dissection study of unembalmed human leg specimens, which revealed a thin layer of periosteal fibers in the heel.
In line with this, MRI data obtained from patients complaining of Achilles tendonitis and from healthy people highlighted a strong correlation between the thickness of the AT paratenon and that of the PA, further strengthening the anatomical and structural relationship between those structures. There is also evidence of a strong statistically significant correlation between cross-sectional measurements of the AT and PA at their calcaneal insertion. Other studies have argued instead for partial contiguity between AT and PA fibers. This could be because anatomical continuity between these collagenous structures seems to vary with age; it is particularly evident in neonates and younger adults but diminishes during adulthood. The number of AT superficial fibers that become continuous with PA fibers seems to decrease with increasing age, along with calcaneal ossification of the tendon into the bone, which further separates these 2 structures.
Biomechanically, there is increasing evidence for a strong correlation between the AT and PA. Thus, the AT-calcaneus-PA-complex can be seen as a part of a broader myofascial system, where adjacent structures collaborate to spread and transmit the load force. If we consider each of these structures as representing respectively the proximal, intermediate, and distal parts of the conjoined tendon of the calf muscles (i.e., the gastrocnemius and soleus), then it can assumed that even triceps surae structures are involved in the development of plantar fasciitis.
Part II, which focuses on the critical coordination between the triceps surae muscle, Achilles tendon, and plantar aponeurosis and the structural and functional coupling between muscles and fascia, will appear next month.
This article has been excerpted from “Do the fasciae of the soleus have a role in plantar fasciitis?” from Clin Anat. 2023 Aug 4. doi: 10.1002/ca.24102, by the same authors. Editing has occurred, including the renumbering or removal of tables, and references have been removed for brevity. Use is per CC Attribution 4.0 International License. Part II, which focuses on the critical coordination between the Triceps surae muscle, Achilles tendon, and plantar aponeurosis and the structural and functional coupling between muscles and fascia, will appear next month.

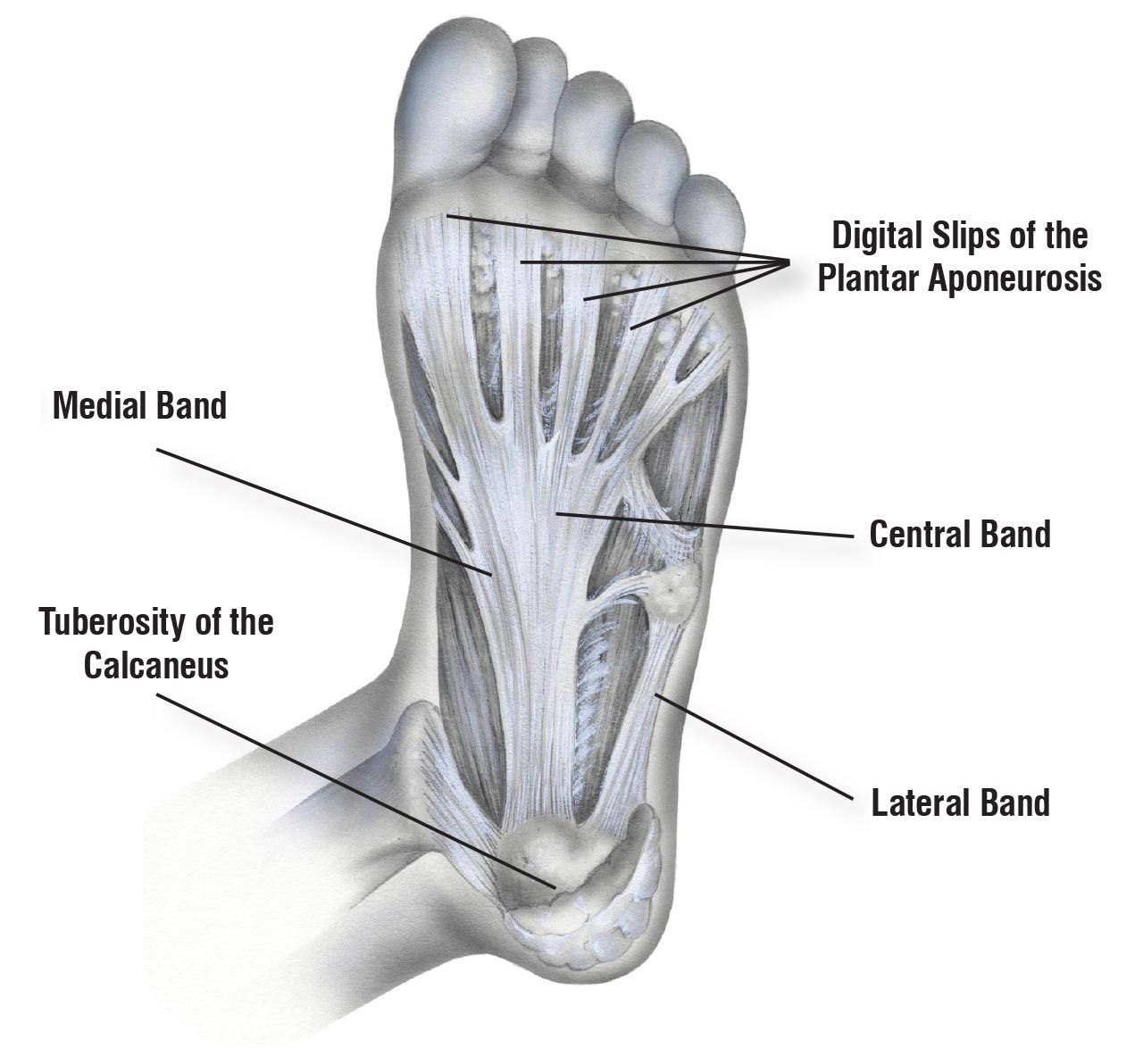
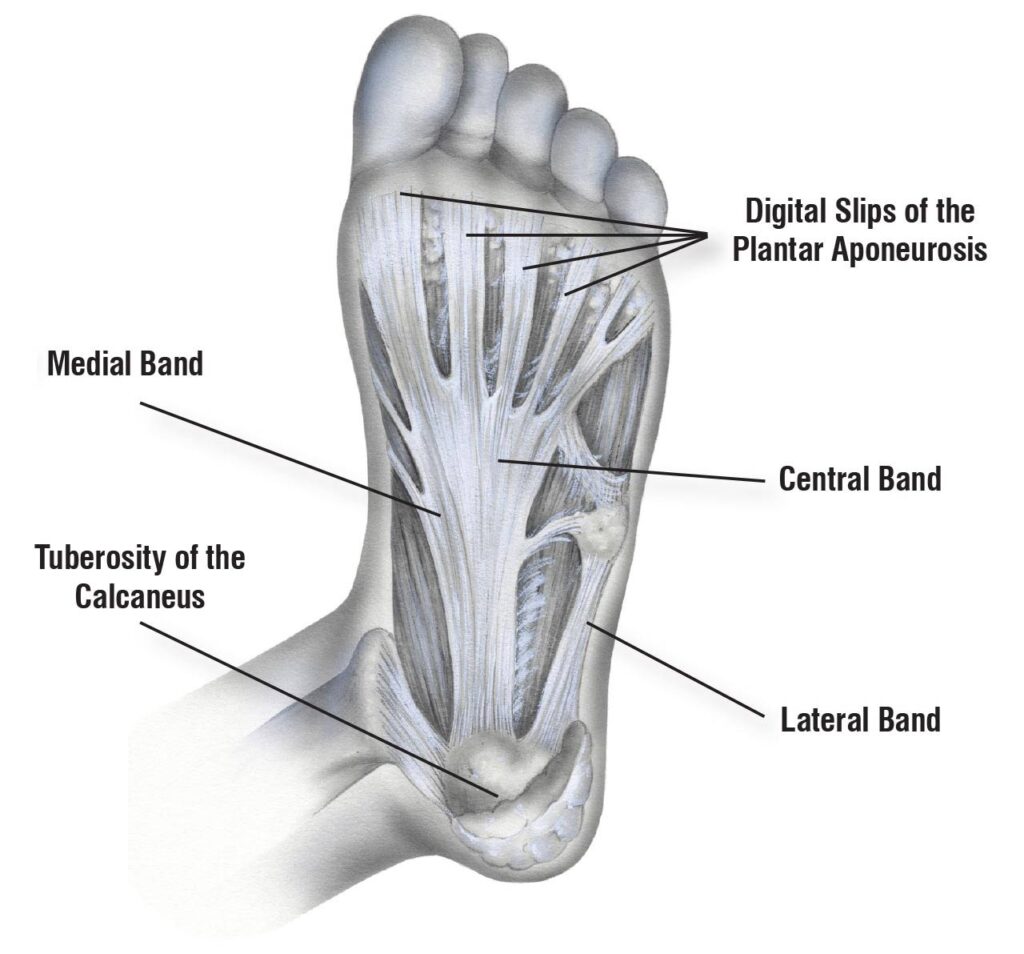







Medial and lateral band are mismarked in the illustration.
Without knowing anything about the authors, the article lacks credibility.
The fascia extends upwards up to and around the skull and ends in the forehead. This is the reason that you can eliminate equinus with cranial sacral work.
Thank you for pointing out this error. We will be sure to correct for the next issue.
Appreciate your support of LER!
Might want to correctly label your diagram, with respect to medial and lateral bands.
Thank you for pointing out this error. It has been corrected.
Thank you for this comment. We have fully identified the authors in the second portion.