Implementation of these new, highly sophisticated devices can help prevent, diagnose, treat, and monitor wound progress.

Figure 1. A) Using various optical signals, the SnapshotNIR provides images that detail the proportion of oxygenated hemoglobin found within the tissue capillary bed. B) The image produced by the device showing various oxygenation percentages found in tissues throughout the wound bed.
By Windy Cole, DPM
As a medical director in a wound care clinic and the director of wound research for a podiatric medical school, I’ve seen a lot wound imaging in my time. Recently, I have seen some new and exciting imaging innovations enter the wound care space that have the potential to advance wound healing outcomes in a broad base of patients suffering from hard-to-heal wounds. Wound care is not a cookie-cutter specialty and no one therapy fits all. As our knowledge of the pathophysiology of chronic wounds continues to grow, so does the number of advanced dressings and treatment options. An area of wound care that has started to see some exciting innovation is wound diagnostics. Medical imaging, such as X-ray, MRI, and ultrasound, have been important tools for many years throughout all aspects of medicine. Historically, wound care clinicians have not had a lot of similar type technology available at our offices or wound care centers. That appears to be changing. There are new, small, portable, point-of-care devices being developed now that can easily fit into the wound care algorithm. I think the future of wound care is the adoption and implementation of these new theragnostic devices that can help prevent, diagnose, treat, and monitor wound progress.
Changing the Way We Practice
Wound physiology is complex. The practice of wound care is very much a visual specialty. While most wound care centers use standard camera systems to supplement the medical record, track wound progress, or even take wound tracings, the specialty of wound care has not fully realized advances in wound imaging. The development and incorporation into everyday healthcare of these emerging sophisticated technologies will allow clinicians to obtain more information about the wounds we are treating and enable a better understanding of tissue microenvironments. For instance, these new imaging devices can assess tissue oxygenation, microvasculature, overall tissue health, and wound microbiome. Use of these new ‘theragnostics’ will provide more quantitative information aiding clinicians in making more informed treatment decisions. As we better understand the wound healing process and host response to therapy, we will be better able to monitor wound status and deliver adaptive wound therapies.
Detecting Tissue Oxygen Saturation
Adequate tissue perfusion is a known predictor of wound healing. Historically, non-invasive vascular studies are a good screening test, but sometimes the results give clinicians very little information, especially when it comes to determining the functional blood flow to the wound. Patients may appear to have adequate large vessel perfusion, but may actually have poor perfusion present at the area of concern due to microvessel disease. Near-infrared spectroscopy (NIRS) is an emerging imaging technology that can be used to evaluate functional tissue oxygen saturation in the management of chronic ulcerations.1-3 One such commercially available NIRS device is the SnapShotNIR (Figure 1; Kent Imaging, Calgary, Alberta, Canada). This non-contact theragnositic device is hand-held, mobile, and offers repeatable immediate images that can be used to determine site-specific quantifiable levels of tissue oxygenation in and around the wound. This diagnostic tool utilizes differing optical signals based on the proportion of oxygenated hemoglobin found within the tissue capillary bed.3 The images obtained allow clinicians to get a better idea of microcirculation and functional blood flow to the wound as well as the surrounding tissues.

Figure 2. A) Moleculight i:X illuminates the wound and surrounding area with violet light, exciting the wound tissues and bacteria and resulting in endogenous production of fluorescence signals, without additional contrast agents. B) The image produced by the device showing various layers of florescence within the wound bed.
Bacteria Detection and Clinical Decision Making
Increased wound bacteria loads can hinder healing and cause acute infection.4 Bacteria and biofilm may persist in the wounds even with good wound care. Elevated levels of tissue bioburden will further prolong wound chronicity. Though swabs and/or wound biopsies can be used to confirm presence of bacterial burden in wounds, use of these procedures varies widely and results take days to obtain. The lack of objective information at point of care to guide appropriate debridement contributes to delays in wound healing.
There is now a handheld fluorescence imaging device, MolecuLight i:X (Toronto, Ontario, Canada), that is easy to use, non-invasive and portable (Figure 2). This technology visualizes potentially harmful bacteria on the wound surface and surrounding tissues not otherwise visible with the naked eye. The device emits a violet light that illuminates the wound and surrounding area, exciting the wound tissues and bacteria and resulting in endogenous production of fluorescence signals, without the need for additional contrast agents.5 Optical filters built into the device remove non-informative colors, without any digital processing, and the resulting image is viewed on the display touch screen in real-time.6
The fluoresce signals produced are tissue specific: endogenous tissue components such as collagen will fluoresce green, while pathologic bacteria fluoresce red, and pseudomonas will fluoresce cyan.7 The information captured in the images can aid in more targeted and thorough wound debridement, support clinical decisions and dressing selection, and aid in determining the need for topical and or oral antimicrobial therapy to improve clinical outcomes.8
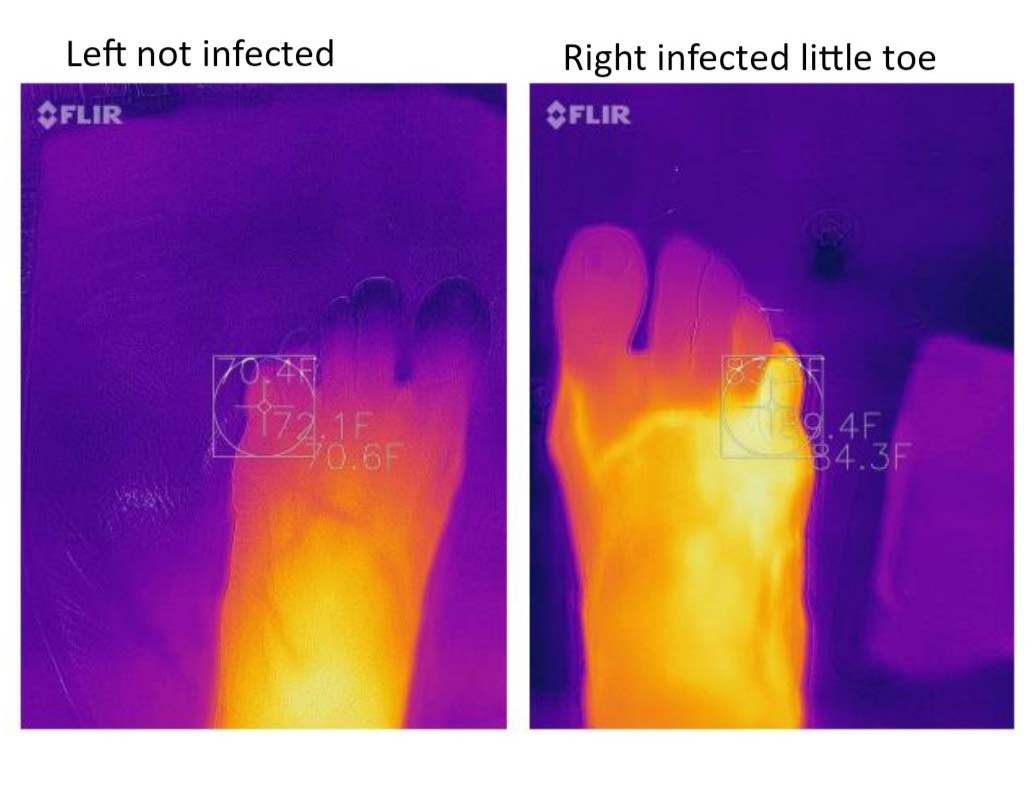
Thermal Imaging May Also Be Useful to Wound Care Clinicians
Thermal imaging devices can potentially be used to provide information on systemic or local temperature abnormalities in tissue caused by things such as ischemia, trauma, inflammation, or infection prior to clinical manifestations. The technique consists of comparing images obtained on both limbs (Figure 3) and performing an asymmetrical analysis by subtracting mean temperature of the nonulcerated limb from the corresponding value of the ulcerated one.9 Long-wave infrared thermography can measure radiant heat from a body surface and has been accepted as a valuable adjunct to standard investigations in the early detection of inflammation and infection.9 Research has shown that a temperature difference between a chronically infected wound and normal tissue has a specific elevated thermal gradient range of 3 degrees C to 4 degrees C.10 The theragnostic value is limited if the contralateral limb also has an ulceration.10
Final Thoughts
Issues of cost, ease of use, and reimbursement remain unknown factors that may stand in the way of implementing these devices as standard of care. As these new technologies advance and evolve, I believe they will gain wider acceptance across the board. These devices have the potential to help clinicians collect more accurate and objective wound data allowing for better documentation and the formulation of more effective wound management. This is a key area of new research to keep our eyes on.
Windy Cole, DPM, serves as Medical Director of the Wound Care Center, University Hospitals Ahuja Medical Center and Adjunct Professor and Director of Wound Care Research at Kent State University College of Podiatric Medicine, both in Cleveland, Ohio. She is a dedicated healthcare advocate with interests focused on medical education, diabetic foot care, wound care, limb salvage, clinical research and humanitarian efforts. Dr. Cole has published extensively on these topics and is a sought-after speaker both nationally and internationally. Dr. Cole also serves as a member of the Editorial Advisory Board for LER.
- Neidrauer M, Zubkov L, Weingarten MS, Pourrezaei K, Papazoglou ES. Near infrared wound monitor helps clinical assessment of diabetic foot ulcers. J Diabetes Sci Technol. 2010;4(4):792–798.
- Khaodhiar L, Dinh T, Schomacker KT, et al. The use of medical hyperspectral technology to evaluate microcirculatory changes in diabetic foot ulcers and predict clinical outcomes. Diabetes Care. 2007;30(4):903–910.
- Nouvong A, Hoogwerf B, Mohler E, Davis B, Tajaddini A, Medenilla E. Evaluation of diabetic foot ulcer healing with hyperspectral imaging of oxyhemoglobin and deoxyhemoglobin. Diabetes Care. 2009;32(11):2056–2061.
- Edwards R, Harding KG. Bacteria and wound healing. Curr Opin Infect Dis. 2004;17(2):91-96.
- Wu YC, Smith M, Chu A, et al. Handheld fluorescence imaging device detects subclinical wound infection in an asymptomatic patient with chronic diabetic foot ulcer: a case report. Int Wound J. 2016;13(4):449-453.
- Rennie MY, Dunham D, Lindvere-Teene L, Raizman R, Hill R, Linden R. Understanding real-time fluorescence signals from bacteria and wound tissues observed with the MolecuLight i:X™. Diagnostics (Basel). 2019;9(1):22.
- DaCosta RS, Andersson H, WilsonBC. Molecular fluorescence excitation-emission matrices relevant to tissue spectroscopy. Photchem Photobiol. 2003;78(4): 384-92.
- Cole W, Coe S. Use of a bacterial fluorescence imaging system to target wound debridement and accelerate healing: a pilot study. J Wound Care. 2020;29(Sup7);S44
- Bird HA, Ring EF. Thermography and radiology in the localization of infection. Rheumatol Rehabil. 1978;17(2):103-106.
- Fierheller M, Sibbald RG. A clinical investigation into the relationship between increased periwound skin temperature and local wound infection in patients with chronic leg ulcers. Adv Skin Wound Care. 2010;23(8):369-379.