
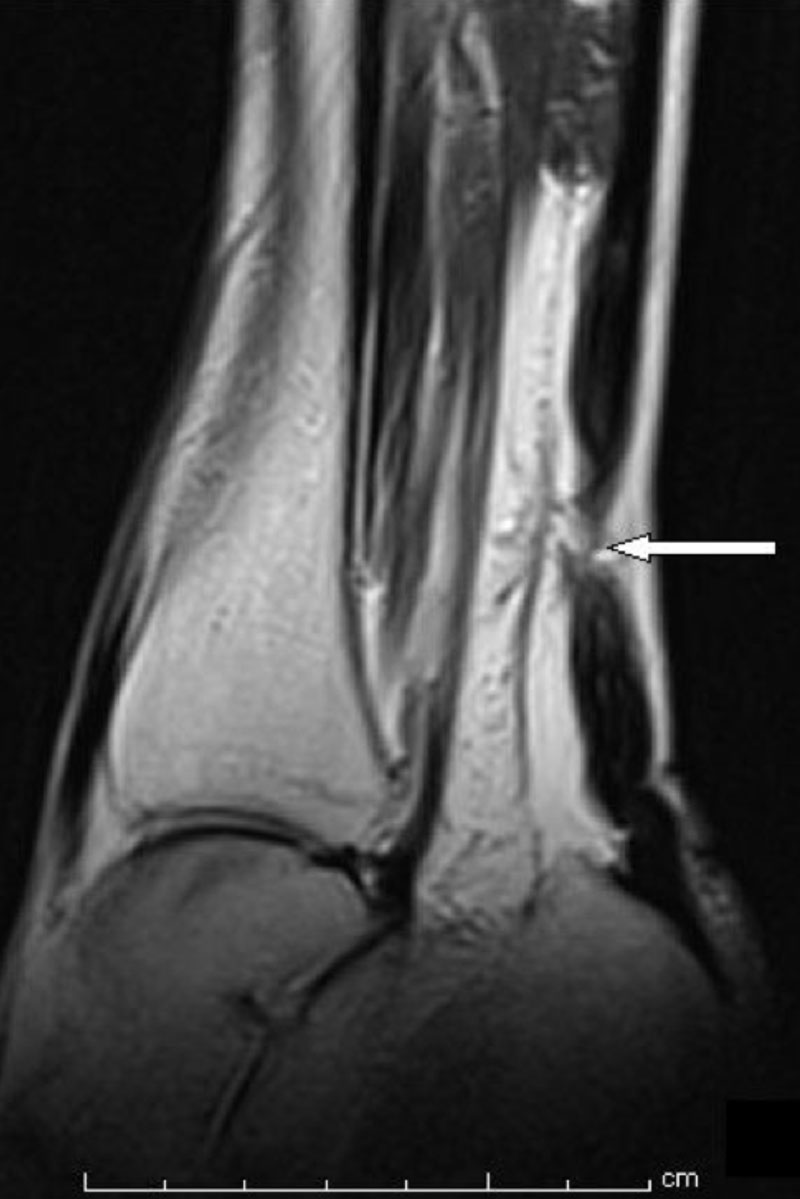
Right Achilles tendon rupture in a patient with bilateral, spontaneous Achilles rupture while prescribed steroids and fluoroquinolones.3 (Use is per Creative Commons Attribution License.)
Despite Black Box warning for risk of tendinitis and tendon rupture, clinicians continue to prescribe fluoroquinolones.
By Nicole Wetsman
Half of the musculoskeletal injuries in the United States each year involve tendons and ligaments, and of the tendon injuries that can occur, those that strike the Achilles tendon are the most common. Higher rates of participation in physical activity and sports, and participation in such activities later in life, have driven an increase in Achilles tendon injuries over the past decades.1 Recovery can be a long process, and for a full rupture, can take up to a year.
Many risk factors for Achilles injury or rupture come from physical factors—like ankle instability, excessive exercise, or calf muscle weakness. However, risk factors can also come from inside the medicine cabinet: in particular, data show that a class of antibiotics, called fluoroquinolones, increases the chances that a patient can injure their Achilles tendon.
“There’s a significant association between their use and tendinitis and tendon rupture,” said Nick Daneman, MD, FRCPC, MSc, a scientist studying infectious diseases and antibiotics at Sunnybrook Research Institute in Toronto, Canada.
While the reasons that these antibiotics are linked to tendon injury are still under investigation, experts say they present a significant enough risk that physicians should keep the association in mind when prescribing these drugs and working with patients who experience tendon injuries.
Elevated Risk
In one case study published in 2018, a 71-year-old man receiving chemotherapy presented with fever and was diagnosed with cholangitis, an inflammation of the bile duct. He was prescribed a course of levofloxacin, which is a fluoroquinolone antibiotic, and experienced an Achilles tendon rupture 5 days later.2
“It’s a well-known complication,” Daneman said. The risk of fluoroquinolone antibiotics had not been identified during initial investigation of the drug before it was approved by the Food and Drug Administration (FDA) more than 30 years ago. However, once the drug was in clinical use, tendon rupture and tendinitis were reported by patients. In 2006, the FDA-watchdog group Public Citizen petitioned the agency to mandate warnings about the risk of the drug, and sued after the agency did not respond to its petition. In 2008, the FDA added a Black Box warning—the strictest warning that can be added to a drug—to fluoroquinolones, noting that they can cause tendinitis and tendon rupture.
“From November 1997 through December 2007, there have been 407 reported cases of tendon rupture and 341 cases of tendinitis in patients using fluoroquinolone antibiotics. Because only a small fraction of cases are typically reported to the FDA, the actual number of ruptures and other tendon injuries attributable to the antibiotic is much higher,” Public Citizen wrote in a statement in 2008.4
Since the first case study was published on tendon rupture and fluoroquinolones in 1983, research has consistently identified a relationship between the two. A 2002 study of lung cancer patients showed that fluoroquinolones carried nearly 4 times the risk of tendon rupture than other types of antibiotics,5 and a 2007 study showed that patients taking the drugs are 3 times more likely to rupture their tendon than patients not taking the drugs.6 However, despite the increased relative risk, tendon rupture—and tendon rupture associated with these antibiotics—is rare. In a 1996 study of 3 fluoroquinolones, the injuries occurred at a rate of 2.4 incidences per 10, 000 patient prescriptions (tendinitis) and 1.2 incidences per 10, 000 patients (tendon rupture).7 A new meta-analysis found that people taking fluoroquinolones had 2½ times the risk of a tendon rupture and nearly 4 times the risk of tendinitis.8
Despite the relative rarity, John Mozena, DPM, PC, FACFAS, a podiatrist and surgeon based on Oregon says that it’s something he looks for in patients. “One of the first things I think about when patients come in with tendinopathy is if they’ve been on antibiotics recently,” he said. “It’s surprising how many times the answer is yes.”
While the mechanisms by which fluoroquinolones disrupt tendons are not fully understood, research in cell lines and animal models indicate that they impact gene expression in cells that make up tendons. One study in rats, for example, found that treatment with the antibiotics led to cellular degeneration in the tendons;9 other research has found that the antibiotics increase the expression of certain enzymes that break down collagen—which makes up 70% of tendons.10
Because they likely impact collagen, fluoroquinolones might have an effect on other parts of the body with a large amount of collagen. “The aorta has the same collagen,” Daneman said. “We asked the question—is there an association between these antibiotics and aortic aneurysms?” Daneman analyzed data from more than 650,000 patients over age 65 who had been prescribed at least one fluoroquinolone. The study, published in 2015, confirmed that patients taking fluoroquinolones were more likely to have tendon ruptures than those who did not, and also found that patients taking those drugs were more likely to experience aortic aneurysms.11
Daneman said that other conditions that involve collagen might be a subject for future research on the effects of fluoroquinolones. “There are many serious problems that tie back to collagen,” he said.
John Mozena, DPM, PC, FACFAS
Applying the data in the clinic
While the Black Box warnings help inform physicians who prescribe antibiotics of the potential risk factors that come with fluoroquinolones, tendon injury or ruptures are still rare enough that clinicians might not recognize an antibiotic-related injury if they’re presented with one. “You could prescribe a lot of antibiotics without encounters with this,” Daneman said. “When something occurs on the order of 1 in 10,000, you might not see it. It becomes much more real when you see a case.”
Mozena thinks that tendon injuries related to antibiotics are underreported, which also contributes to lower counted case rates and less recognition from physicians. “What the mind doesn’t know, the eyes can’t see,” he said. “If you’re not aware of it, you wouldn’t recognize it or let patients know.”
For Mozena, pharmaceutical companies and the FDA should play a more active role in disclosing risks and tracking these complications. “There needs to be a better way for physicians to report them,” he said. “And how can you report if you’re not aware?”
Despite risks, though, fluoroquinolone antibiotics remain important tools for the treatment of infections, noted Daneman. “When prescribing antibiotics, you always have to weigh the benefits against the risks,” he said. One of the best ways to avoid the risks is good antibiotic stewardship and responsible antibiotic prescribing practices in general. “The main way to put this knowledge into practice is to avoid prescribing fluoroquinolones to patients who don’t need antibiotics,” he said. “The most upsetting thing would be to have a severe complication in a patient who didn’t need an antibiotic in the first place.”
While research on the risk of fluoroquinolones has largely been in the general population, Daneman said that physicians might be able to look for co-existing conditions or other risk factors when deciding if antibiotics are an appropriate choice for a particular patient. For example, if a patient was also taking steroids—which are a risk factor for tendon rupture—physicians may not want to add an additional risk factor by prescribing fluoroquinolones. One study, published in 2003, found that elderly patients also taking oral corticosteroids were at the highest risk of tendon rupture.12
Doctors could also consider if a tendon rupture would have an impact on a patient’s occupation, Daneman said, and people who regularly put strain on their tendons, like athletes, might not want to add the additional risk.
Mozena also said that physicians might think twice about prescribing these drugs to athletes. “In my experience, many clinicians overprescribe antibiotics for athletes to try and get them back to sports. In reality, it might be causing additional problems,” he said.
Researchers have proposed specific guidelines around the use of fluoroquinolones in athletes: 13
- fluoroquinolones should not be administered along with steroids,
- athletes and coaches should be made aware of the risks, and
- athletes should be monitored for one month after taking the antibiotics.
Continued research on fluoroquinolones would help physicians to understand which patients should take the antibiotics for various infections, and which should avoid them. Understanding the mechanisms by which the drugs act on tendons might also help reduce risk. “If people could determine the exact cellular mechanisms that cause this, we might be able to develop ways to prevent it, and ways to mitigate risk,” Daneman says. “It would be helpful to know if a subset of patients are at a particular risk, and where the risk-benefit ratio would shift in favor of not treating with these drugs.”
Nicole Wetsman is a freelance writer in New York City.
- Wu F, Nerlich M, Docheva D. Tendon injuries: Basic science and new repair proposals. EFORT Open Rev. 2017;2:332–342.
- Kitagawa S. Levofloxacin-induced Achilles tendon rupture. Intern Med. 2018;57:2921–2922.
- Khanzada Z, Rethnam U, Widdowson D, Mirza A. Bilateral spontaneous non-traumatic rupture of the Achilles tendon: a case report. J Med Case Rep. 2011;5:263.
- Tanne JH. FDA adds “Black Box” warning label to fluoroquinolone antibiotics. BMJ. 2008;337:a816.
- Chhajed PN, Plit ML, Hopkins PM, et al. Achilles tendon disease in lung transplant recipients: association with ciprofloxacin. Eur Respir J. 2002;19:469-71.
- Sode J, Obel N, Hallas J et al. Use of fluroquinolone and risk of Achilles tendon rupture: a population-based cohort study. Eur J Clin Pharmacol. 2007;63:499-503.
- Wilton LV, Pearce GL, Mann RD. A comparison of ciprofloxacin, norfloxacin, ofloxacin, azithromycin and cefixime examined by observational cohort studies. Br J Clin Pharmacol. 1996;41:277–284.
- Alves C, Mendes D, Marques F B. Fluoroquinolones and the risk of tendon injury: a systematic review and meta-analysis. Eur J Clin Pharmacol. 2019; 10.1007/s00228-019-02713-1.
- Shakibaei M, Stahlmann R. Ultrastructure of Achilles tendon from rats after treatment with fleroxacin. Arch Toxicol. 2001;75:97-102.
- Tsai WC, Hsu CC, Chen CP, et al. Ciprofloxacin up-regulates tendon cells to express matrix metalloproteinase-2 with degradation of type I collagen. J Orthop Res. 2011;29:67-73.
- Daneman N, Lu H, Redelmeier DA. Fluoroquinolones and collagen associated severe adverse events: a longitudinal cohort study. BMJ Open. 2015;5:e010077.
- van der Linden PD, Sturkenboom MC, Herings RM, et al. Increased risk of Achilles tendon rupture with quinolone antibacterial use, especially in elderly patients taking oral corticosteroids. Arch Intern Med. 2003;163:1801-7.
- Lewis T, Cook J. Fluoroquinolones and tendinopathy: a guide for athletes and sports clinicians and a systematic review of the literature. J Athl Train. 2014;49:422–427.








Uses recently prescribed doxycycline and prednisone. Is there risk. I have excruciating pain in my upper arm
I was prescribed these antibiotics for several weeks for a sinus and urinary tract infection. Then after several weeks came down with bursitis in my right hip along with bumps on my hands. I have quit these antibiotics after talking to the pharmacy. The pain in my hip is debilitating but does get better during the day. I am 71 and very active spending at least two hours per day working out. I think that this came from antibiotic use.