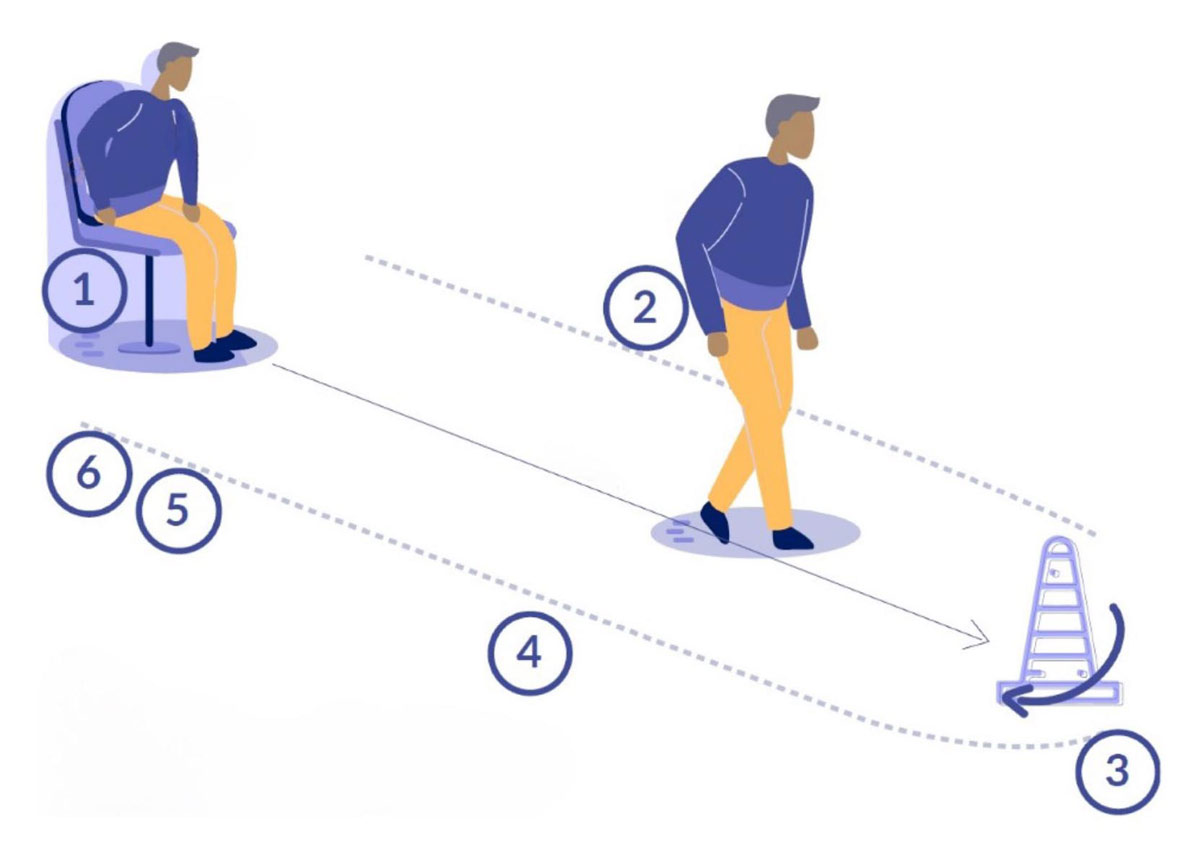
Timed Up and Go test with the different sub-phases after the most complete segmentation. (1) Standing. (2) Go Walking. (3) Three-meter turning. (4) Return Walking. (5) Pre-sitting turning. (6) Sitting. Image reprinted from Ortega-Bastidas P, Gómez B, Aqueveque P, Luarte-Martínez S, Cano-de-la-Cuerda R. Instrumented Timed Up and Go Test (iTUG)—More Than Assessing Time to Predict Falls: A Systematic Review. Sensors. 2023; 23(7):3426. https://doi.org/10.3390/s23073426. Use is per Creative Commons Attribution (CC BY) license.
While glatiramer acetate (GA; Copaxone®, Teva Neuroscience) has long been FDA-approved for relapsing forms of multiple sclerosis (MS), a new safety and efficacy snapshot study from Israel looked at its use in primary progressive MS (PPMS). Two GA depot doses (40 mg or 25 mg) were studied in a 12-week trial that included 30 patients age 18-65 years with documented PPMS and disease worsening in the year before screening at a rate of ≥1 point increase per year on Expanded Disability Status Scale (EDSS) score and baseline EDSS score of 2 to 6.5. At baseline, mean age was 50.3 years and there was an even distribution of men and women; mean EDSS score was 5.1
EDSS scores remained stable, with only one 12-week confirmed disability progression (CDP). Notably, 72.4% of patients (68.4% in the 40 mg dose group and 80% in the 25 mg dose group) showed no evidence of progression (NEP), defined by the absence of 12-week CDP (96.6%), 12-week Timed 25-Foot Walk (T25FW) progression (79.3%), and 12-week Nine-Hole Peg Test (9HPT) progression (93.1%). MRI analysis indicated low MRI activity. Injection site reactions were the most common adverse events (AEs) and no unexpected AEs were reported. Rate for AEs was lower in the 25 mg group compared to the 40 mg group.
The authors concluded that these interim findings suggest GA depot as a safe and effective treatment for patients with PPMS, with a remarkable 72.4% NEP, stable EDSS score, and high proportion of stable T25FW and 9HPT scores once treated.
Source: Fletcher S, Miller AE, Popper L, et al. (DMT21) Glatiramer Acetate Depot (Extended Release) Phase 2a Study in Patients With Primary Progressive Multiple Sclerosis: Safety and Efficacy Snapshot. Int J MS Care. 2024;26(S1):33.






