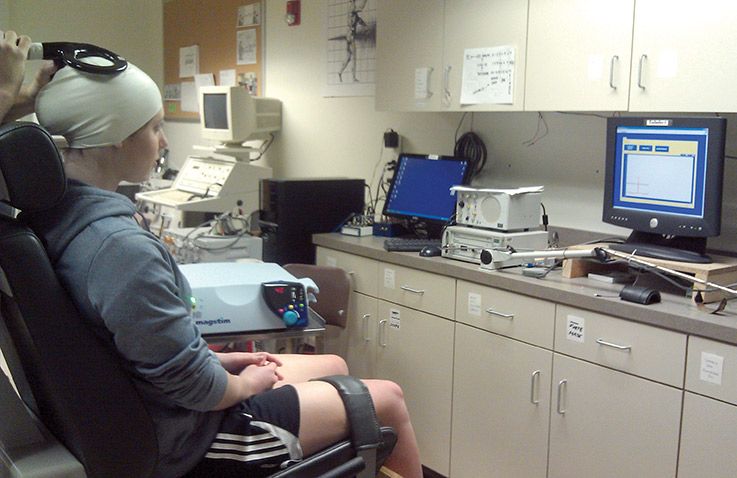
Study participant undergoes testing for cortical excitability and inhibition measures. (Image courtesy of Alan Needle, MS, ATC, CSCS.)
By Emily Delzell
A small study points to a “decoupling” effect between neurological and mechanical aspects of the ankle joint among some people with recurrent sprains and resultant functional ankle instability (FAI), reported investigators in a Journal of Sport Rehabilitation paper e-published on April 9.
To see how the brain’s regulation of dynamic joint restraint might change after ankle injury, investigators tested joint laxity with an ankle arthrometer and cortical excitability and inhibition with transcranial magnetic stimulation (TMS) in 24 adults.
Investigators sorted volunteers into two groups of 12 using the Cumberland Ankle Instability Tool (CAIT). One had a history of unilateral FAI and a CAIT score lower than 25 (lower scores indicate greater instability) while a control group had no reported sprains and a CAIT score higher than 27. Using TMS, investigators administered controlled pulses to the primary motor cortex through an elastic cap on participants’ heads and measured electromyographic response from the tibialis anterior (TA), peroneus longus (PL), and soleus muscles.
They found no significant between-group differences in laxity or cortical excitability or inhibition, but reported significant relationships between laxity and TA excitability in those with healthy ankles and between peroneal and soleus excitability and measures of laxity in individuals with FAI, said lead author Alan Needle, MS, ATC, CSCS.
The investigators quantified joint laxity as peak anterior displacement and inversion rotation and used active motor threshold, slope, and intensity at 50% of peak slope of TMS-derived recruitment curves to quantify cortical excitability from lower leg muscles. Intracortical inhibition was represented by the cortical silent period (CSP), a brief pause in voluntary muscle activity following magnetic pulse, from the PL.
In healthy ankles, Needle and colleagues found a relationship between less anterior displacement and greater TA excitability. This group also had a significant negative correlation between anterior displacement and the CSP of the PL, indicating a link between higher inhibition and greater laxity, he said.
“We saw a better link between the tibialis anterior and stiffness in our healthy ankles. The lack of this relationship in people with instability suggests a potential loss of cortical regulation of joint stiffness through the tibialis anterior,” said Needle, a doctoral candidate in the University of Delaware’s Department of Kinesiology and Applied Physiology.
Unstable ankles demonstrated significant correlations between increased anterior displacement and higher soleus excitability and between increased inversion rotation and higher peroneal excitability.
“Those with instability seem to be compensating by relying on a greater degree of excitability to stabilize the joint,” he said, noting that one of the difficulties in teasing out the etiology of FAI—and of treating those with unstable ankles—is the high variability among patients.
“What it’s really coming down to is that it’s an individualized change—and that’s where we’re missing the mark in treatment. Everybody gets textbook treatment, and we’re not really looking at where these individual differences are occurring,” he said. “Some changes may be mechanical, while some of our research suggests there’s something going on peripherally, such as decreased sensation, for which specific interventions like perturbation training would be beneficial. But if changes are occurring higher up in the cortex, like what we’re seeing here, it may be more important to work with interventions that require higher cortical processing, such as replicating complex tasks with your foot.”
Brian Pietrosimone, PhD, ATC, an assistant professor in the Department of Kinesiology at the University of Toledo in Ohio, who is also studying neuromuscular changes after joint injury, emphasized that research in this area is still in the early stages.
“The one thing I can say with any confidence is that there are different subgroups [among people with lower extremity joint injuries] in which people may have the same injury but a nervous system that responds completely differently,” said Pietrosimone, who along with his colleagues, is focusing on ways to treat these variable groups.
Potential interventions could involve using TMS to improve cortical excitability, he said.
“We’re also trying to determine if some deficits are due to cortical overfacilitation [excitability] versus inhibition—and we if we figure that out we might be able to eventually use pharmaceuticals to address changes,” he said.
Source:
Needle AR, Palmer JA, Kesar TM, et al. Brain regulation of muscle tone and laxity differs in functionally unstable ankles. J Sport Rehabil 2013 Apr 9. [Epub ahead of print]