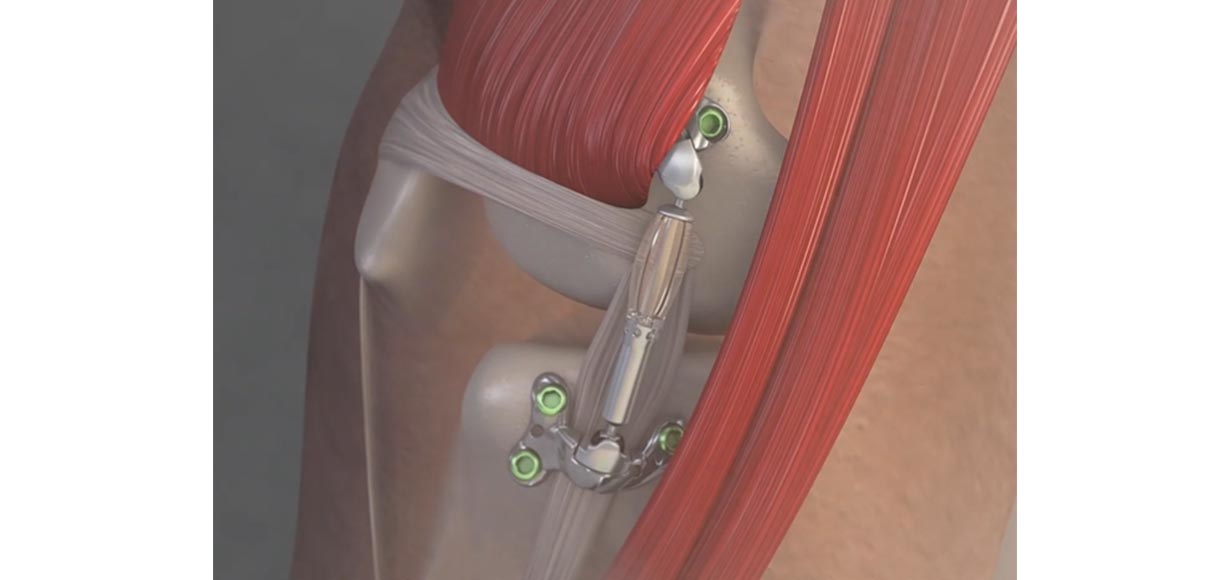
The US Food & Drug Administration has recently authorized marketing of an implantable shock absorber aimed at load reduction for people with refractory knee osteoarthritis (OA) who are not appropriate candidates for arthroplasty. Dubbed the MISHA Knee System, the device is specifically indicated for patients with medial knee OA whose pain continues to interfere with daily life despite prior treatment, including surgery or non-surgical approaches, and are ineligible for, or unwilling to undergo, joint replacement due to age or absence of advanced OA. Implanted subcutaneously in the medial extra-capsular space, the device is affixed to the tibia and femur with screws—a procedure that can be performed in the outpatient setting.
Moximed
510/887-3300