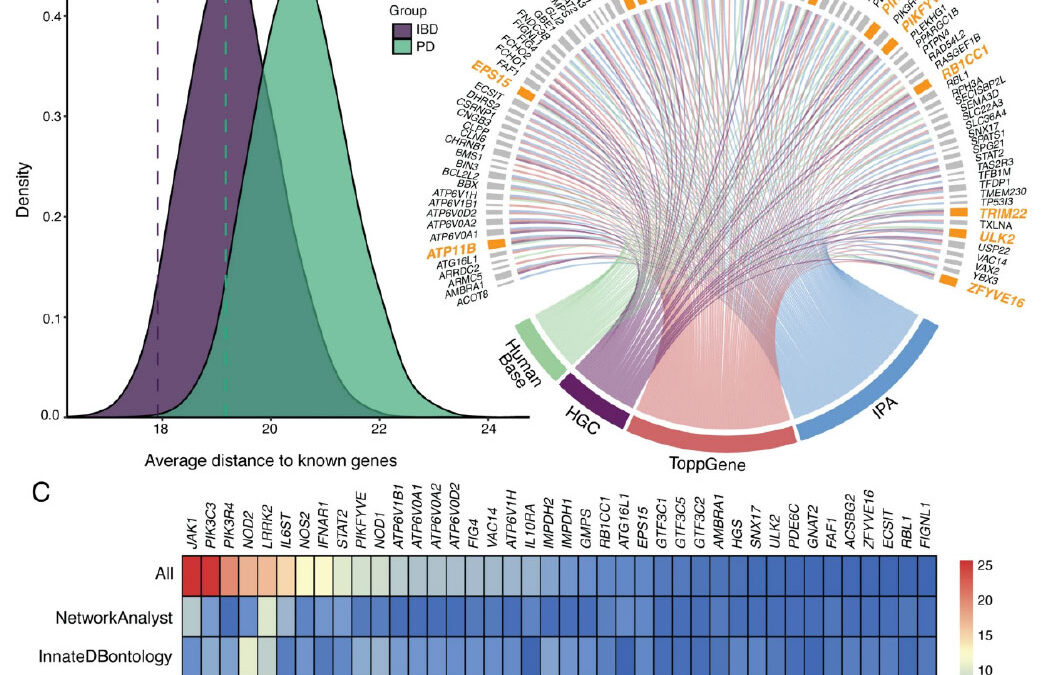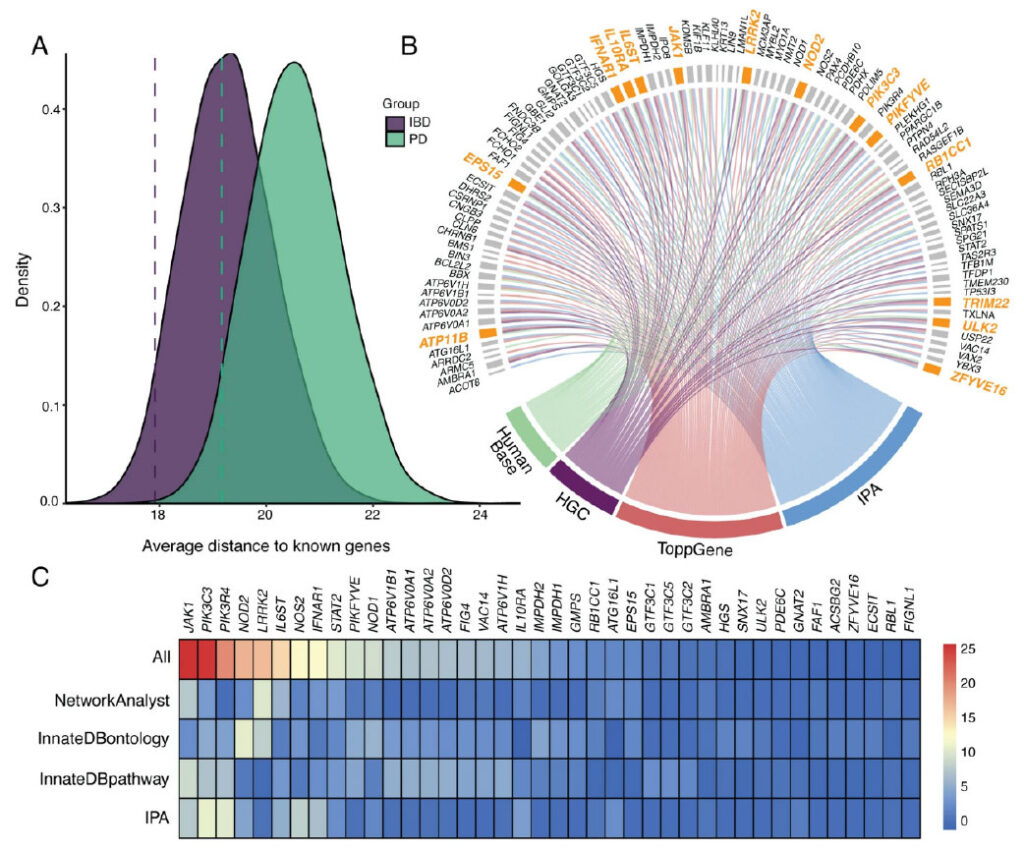
Figure. Candidate gene prioritization and validation using a phenotype-wide association study (PheWAS). A Density plots displaying the distribution of the average distances of 120 random genes (Drandom) to known inflammatory bowel disease (IBD, in purple) and known Parkinson’s disease (PD, in green) genes obtained from 10,000 resampling iterations. Dashed lines represent the average distance of the candidate gene set (Dcandidate, n = 120) to known IBD and PD genes. B Circos plot showing the candidate genes identified by four complementary pathway and biological relatedness approaches. Final candidate genes that were identified by all four methods are highlighted in orange. C Heatmap of the combined biological importance scores of the IBD-PD candidate genes. A higher score indicates a higher number of shared pathways, ontologies, or modules with known IBD and PD genes. The colors represent the magnitude of the scaled scores calculated by each method. The highest-scaled scores are depicted in red, whereas the lowest scores are shown in blue. The top 40 genes are shown in the plot.
Researchers at the Icahn School of Medicine at Mount Sinai have identified genetic connections between inflammatory bowel disease (IBD) and Parkinson’s disease (PD). The 2 chronic disorders have been suggested to share risk factors and underlying pathophysiology consistent with the “gut-brain axis” hypothesis. Previous studies, including a meta-analysis with 12 million patients, indicate that both Crohn’s Disease and Ulcerative Colitis (which make up IBD) are associated with an increased risk of PD diagnosis.
Using advanced genomic analysis techniques to investigate the genetic overlap between IBD and PD, their findings point to mutations in the LRRK2 gene as a common element linking both conditions and identify novel genes that are likely to be affected in people experiencing both IBD and PD.
“We’ve found that IBD and PD are caused by certain shared genetic factors, including variants in LRRK2 and other genes previously unknown for this combined condition. This could dramatically change our approach to these diseases, allowing for therapies that target both conditions simultaneously,” lead author Meltem Ece Kars, MD, PhD, said in a related press release.
The study analyzed data from the Mount Sinai BioMe BioBank, the UK Biobank, and a cohort of 67 patients diagnosed with both IBD and PD from the Danish National Biobank. This combined dataset enabled the researchers to explore high-impact rare genetic variants and identify new genes and biological pathways that contribute to the IBD-PD comorbidity.
“Our research not only links these two diseases genetically but also sets the stage for new forms of treatment, and potentially prevention strategies, that could lessen the burden of these diseases on patients,” Kars said.
The researchers used a variety of computational methods to uncover significant associations between the LRRK2 gene variants and the co-occurrence of IBD and PD, including the network-based heterogeneity clustering approach, which they have demonstrated to be highly effective for gene discoveries in small cohorts that cannot be analyzed by traditional gene association methods. Their analysis also revealed several pathways related to immunity, inflammation, and autophagy, the body’s cellular recycling system, that are involved in both conditions.
These insights have potential implications across multiple areas of medicine, suggesting that understanding genetic factors could lead to better-targeted therapies. The study underscores the importance of genetic research in developing personalized medicine approaches that could improve treatment for patients with both IBD and PD.
The promise of these findings extends beyond current treatment paradigms: “By pinpointing the genetic underpinnings common to both IBD and PD, we pave the way for innovative treatments, whether through the development of novel drug targets or the repurposing of existing drugs, that could potentially tackle the root causes of these conditions,” Kars said.
The results of this study could also influence future research directions, encouraging a more integrated approach to studying diseases that may appear unrelated but share common genetic pathways.
Source: Kars ME, Wu Y, Stenson PD, et al. The landscape of rare genetic variation associated with inflammatory bowel disease and Parkinson’s disease comorbidity. Genome Med. 2024 May 14;16(1):66. doi: 10.1186/s13073-024-01335-2.