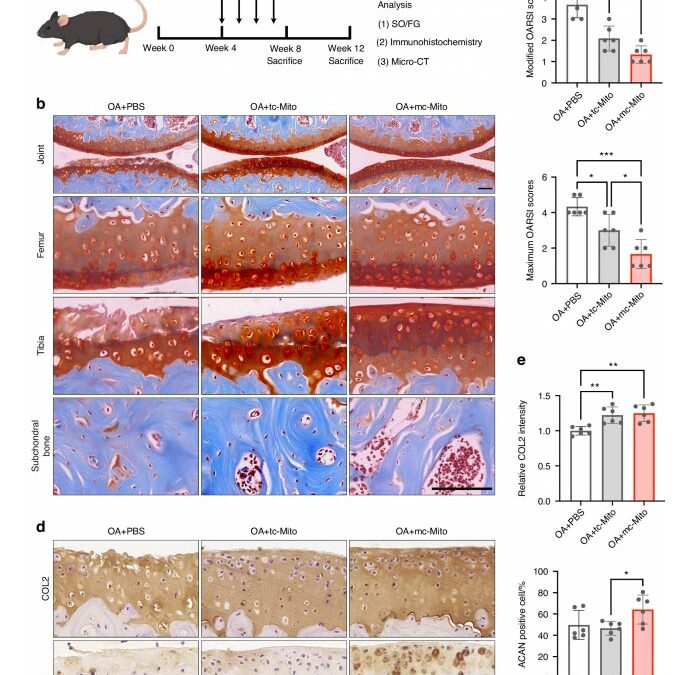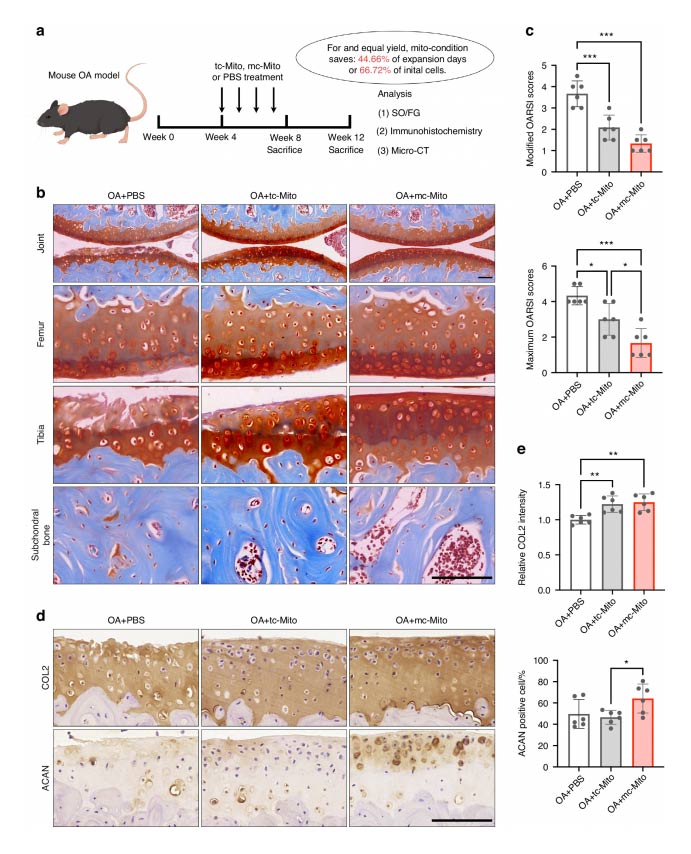
Mc—mitochondria exhibit superior performance for in vivo mitotherapy. (a) Schematic illustration of the establishment of the mouse OA model and the experimental design to evaluate the value of mitochondrial amplification for in vivo applications. (b) Safranin—O/Fast green staining of joint sections at 12 weeks. Scale bar, 50 μm. (c) Modified and maximum OARSI scoring system (sample = 6 for each group). (d) Immunohistochemical staining (COL2, ACAN) of joint sections at 12 weeks. Scale bar, 50 μm. (e) Quantification of COL2 and ACAN in cartilage tissues at 12 weeks (sample = 6 for each group). All data are presented as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001. P values were determined using one—way ANOVA (c, e).
Mitochondrial dysfunction is a common denominator in numerous diseases, including osteoarthritis (OA), heart failure, and metabolic disorders. While mitochondrial transplantation has emerged as a promising avenue for restoring tissue function, its clinical potential has been severely hampered by the scarcity of viable mitochondria. Thus, a team of researchers at Zhejiang University School of Medicine, Hangzhou, China, sought a sustainable and scalable solution for producing high-quality mitochondria suitable for clinical applications. The team has pioneered a stem cell–based system that functions as a “mitochondria factory.” By leveraging human mesenchymal stem cells and a specially designed culture medium dubbed “mito-condition,” the team achieved unparalleled increases in both mitochondrial quantity and quality. These mitochondria exhibited exceptional energy production and facilitated cartilage regeneration in OA models. This groundbreaking approach not only overcomes key barriers in mitochondrial transplantation but also deepens our understanding of cellular organelle regulation, opening new frontiers in regenerative medicine.
The innovative mito-condition culture medium integrates 9 essential components, including growth factors and human platelet lysate, to optimize mitochondrial production. Within just 15 days, this method generated 854 times more mitochondria than conventional approaches, all while preserving stem cell viability.
In OA models, transplantation of these enhanced mitochondria resulted in substantial cartilage repair over a 12-week period, surpassing the efficacy of traditional mitochondrial treatments. Furthermore, the mitochondria demonstrated impressive storage stability, retaining function for 24 hours at 4°C—a critical factor for real-world clinical applications. The concept of organelle tuning, as demonstrated in this study, could potentially be adapted to generate other cellular components, broadening the horizons of cell engineering and therapeutic applications.
The most immediate application of this technology lies in OA treatment, where it offers a promising regenerative solution. However, its impact extends far beyond joint disorders, with potential benefits for conditions such as heart disease, neurodegenerative disorders, and wound healing.