New partnership puts streamlined manufacturing process in the podiatrist’s office
By Keith Loria
Three-dimensional (3D) printing has shown promise in improving medical device manufacture, but cost-effectiveness and seamless manufacturing processes have kept it from becoming mainstream. All that is about to change.
Now, HP Inc. (formerly Hewlett Packard) has partnered with Go 4-D, a visionary player in the medical orthosis industry, to make a difference in footwear and the way podiatrists, orthopedists, and other foot-care professionals deal with some of the most pressing problems involving the foot.
Goal: A Better Product Using Additive Manufacturing
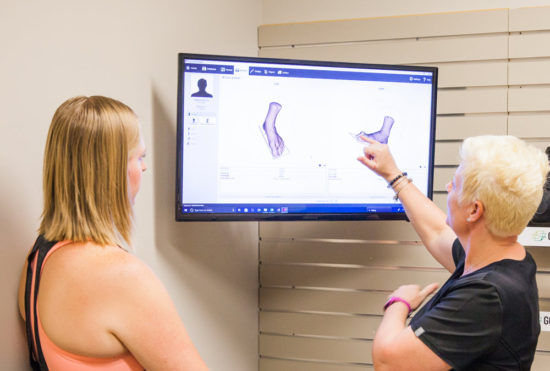
Go 4-D and HP Development Company have released the innovative FitStation, a dual hardware and software platform that captures 3D scans of the foot, collects foot-pressure measurements, performs gait analysis, and builds a user profile based on the input. The data are then utilized to create manufacturing instructions for 3D-printed custom foot orthoses, which are then printed by Go 4-D using HP Jet Fusion 3D printing systems.
Go 4-D was founded by Paul Linton, who helped thousands of health-care practitioners use gait-scanning technology to assess patients’ dynamic needs and order custom orthoses; Lori Yarrow, a chiropractor who practiced and dispensed custom orthoses for years; and Chris Patten, a tech expert who worked in the custom orthotics space for 20 years.
“All 3 of us have been in this space for a long time,” said Linton, who serves as Go 4-D chief executive officer. “We wanted to bring 3D scanning and 2D plantar-pressure measurement together in 1 platform, using the biometric data to customize the product, and then enhancing the product offering through additive manufacturing—something that couldn’t be done under traditional subtractive manufacturing.”
The process started long before the trio became involved. HP, RSscan International, and Materialise had been working together to determine how 3D scanning could translate to a printable orthotic product.
“RSscan is a research medical tool innovator, and they needed a go-to-market company. Our history and experience were well-aligned, and we became involved earlier this year [2018],” Linton said. “We run the HP FitStation and will service the medical channel here in North America.”
Taking 500 Precise Measurements per Second
Edward Ponomarev, general manager of commercial business development and strategic partnerships for FitStation, explained that dynamic gait analysis uses pressure-plate technology and the 3D foot scanner to take as many as 500 precise measurements per second—a game-changer in the orthotics and medical market.
“FitStation is a truly transformative platform that supports HP’s journey to reinvent how the world designs and manufactures with commercial 3D technologies,” he said. “The partnership underscores a push in the health-tech space to merge data with clothing, wearables, and, increasingly, footwear.”
In 2018, FitStation and HP partnered with the National Football League to provide customized cleats to players on all 32 teams using 3D scanning technology.
Having an Impact on the Clinical Environment
Patients interface with the FitStation when they take multiple steps across a pressure-mat system that comes in various lengths (0.5 meter, 1 meter, 1.5 meter, and 2 meter plates). A podiatrist or orthotics specialist then uses a 3D scanner to create the needed footwear.
“We’re using the Tiger scanner, a 9-camera laser scanner. We get true 3D data derived from this as it gets partnered with the 2D data,” Linton said. “Predictive software aids in the design of the orthotic, but the doctor has prescriptive control over what additions and modifications need to be designed. Or the doctor can tweak and alter what he believes is best for the patient. Then he clicks ‘send that order,’ and that order goes to us to be printed on a HP multi-jet fusion printer.”
This process, which is expected to reduce the amount of time spent waiting for the product to arrive, differs strikingly from the old standard of care: plaster of paris casting, a static, subjective, manual process developed in the 15th Century and associated with a great deal of waste.
In casting, Linton explained, the clinician plastered the cast and sent it to a laboratory, where it was scanned and thrown out. Then, a positive orthosis was milled.
“What we’ve changed,” Linton said, “is no longer requiring a cutting away of materials. It’s a pure, simple process. From scanning the foot, to then having a 3D-printed orthotic made simply from the powder that’s required for that foot.”
Additionally, all the excess powder is recycled and able to be used again, making it user-friendly and better for the environment.
The Transformation Is Ongoing
“We constantly want to innovate the [orthotic] product,” said Lori Yarrow, chief customer officer at Go 4-D. “What’s changed right now is the intrinsic properties of the shell. We can make a device more flexible in one area, and in another area adjacent to that, make it more rigid, just by printing more material. This is not possible with subtractive manufacturing, because you can’t change the thickness of the plastic—whereas we’re not changing the thickness of the plastic at all, we’re just changing the amount of content of the plastic.”
Moreover, the unique lattice design of the product allows doctors to create motion control, or stiffness, within the device. So, if the podiatrist wants the device to resist motion in a certain plane, he can prescribe or design that feature in without adding bulk to the device.
“And at the end of the day, it comes down to compliance,” Yarrow said. “We can control the foot in a lot of ways that weren’t possible before, especially without adding that bulk. So now we can have a thinner, a more controlling, device that fits in more shoes.”
Ongoing changes in printer technology have affected the amount of material used and the time that a print job takes, resulting in how many devices or items can be done in 1 print session. Current multi-jet fusion technology prints 10 times faster than selective laser sintering technology of the first generation.
“Therefore, we can print a lot more product in a shorter period of time and bring the cost down to a level more in line with the industry-accepted range of prices for a custom-made product,” Linton said. “Before that, it would have been too expensive, and it wouldn’t have been disruptive if the price point was that high. We’re now at the point where we can be disruptive. We can do a better product than what exists in the market at a competitive price.”
Making a Difference
The benefits of 3D-printed orthoses are game-changing, Linton said, noting the ability to produce a lighter weight, more controlling, yet flexible device and an ability to control parameters that weren’t controllable under subtractive manufacturing.
And then there’s all that quantitative, objective, dynamic data that practitioners have been working with that can be combined to make things better for all involved.
“Podiatrists don’t really share their data very often, but if we are able to use this data to better enhance our product, and not only in orthotics but in footwear,” Linton said, “we can not only make a better product for that patient, but [for] every prospective patient with similar conditions and similar foot types. We can decrease the learning time to treat these patients, so there’s tremendous benefit. This will change orthotic therapy.”
The ultimate goal, Linton said, is to have a custom-made product that’s 3D-printed for the individual patient in a name-brand shoe, such as a Brooks Sports. The shoe can, for example, be produced in Size 9.23 US for one foot and Size 8.67 US for the other—fully unique, designed for that patient.
Identifying Customers of the Future
Becoming a major force in the orthotics market is a major goal of Go 4-D, although the need to have practitioners invest in the equipment presents a barrier to entry: Linton noted that the Tiger 9-camera scanning system and the pressure plate itself are expensive, but necessary tools to make the change happen.
“We want precise data, and it requires equipment that costs a bit of money. We also want doctors to get on board with what we’re trying to do in the market, so we’re giving the equipment to a practitioner based on a volume requirement,” he said. “The most likely candidates for this are established clinics that realize that the future is changing, and all labs eventually will go to 3D printing, and they want to be one of those early adopters and help lead this innovation.”
That means targeting high-volume podiatric clinics, for the most part, but also certain foot-care specialists.
“As costs of hardware come down and costs of printing come down, it may open up additional clinics that maybe do lower volume than those,” Linton said. “While it’s completely different technology, it’s not changing the model in which [podiatrists] prescribe: It’s just giving them extra abilities to prescribe different characteristics. It’s really just tweaking their existing process and bringing them on board to not only what we can do today, but what we can do in the future.”
Keith Loria is a Washington, DC-based freelance writer. Manufacturers cited here did not create or approve this copy.