 Advocates of surgical nerve decompression in a subset of patients with diabetic neuropathy have published some impressive outcomes, but critics of the procedure point to the conspicuous absence of randomized trials. The issue has become one of the most contentious in diabetes care.
Advocates of surgical nerve decompression in a subset of patients with diabetic neuropathy have published some impressive outcomes, but critics of the procedure point to the conspicuous absence of randomized trials. The issue has become one of the most contentious in diabetes care.
By Cary Groner
In April 1992, a paper appeared in the Journal of Plastic and Reconstructive Surgery describing the results of a prospective study of surgical nerve decompression (neurolysis) in patients with diabetic neuropathy.1 The outcomes were interesting but drew little attention at the time; 44% of patients had improved motor function and 67% showed improved sensory function, 10% did not improve, and 2% saw a decline in sensorimotor function. The author concluded that “symptoms of sensorimotor diabetic neuropathy may be partly due to compression of multiple peripheral nerves,” and that “surgical decompression of such nerves may result in symptomatic improvement.”
These were relatively modest claims, but over the years similar papers appeared, and many in the medical establishment came to view their assertions as unsupportable. Today the issue has become one of the most contentious in diabetes care.
The nerve decompression process involves first identifying cases in which nerves passing through a constricted area may experience compression. The usual suspects include the common peroneal nerve at the fibular neck, the superficial peroneal nerve in the leg, the deep peroneal nerve over the dorsum of the foot, and the tibial nerve and its branches at the medial ankle.2 The procedure itself essentially entails surgically releasing the nerve from entrapping tissues, which may include tendons or fascia, thus alleviating what is sometimes called the “napkin-ring effect.”
Skepticism
“The neurologists claim that I say every neuropathy patient should have a decompression, and that is absolutely not what I’ve ever said,” said A. Lee Dellon, MD, PhD, the author of the 1992 paper cited above. Dellon is a professor of plastic surgery and neurosurgery at Johns Hopkins in Baltimore, and over the phone he sounds like someone who’s been highly exasperated for a very long time. “I have said that if a patient with diabetes and neuropathy has a superimposed nerve compression, they should have a decompression.”

Figure 1. Illustrations depicting Dellon’s nerve decompression technique. (Reprinted with permission from Dellon.com)
For their part, “the neurologists” respectfully—and sometimes not so respectfully—disagree. In a practice advisory published in Neurology in 2006, the authors concluded the evidence supporting decompressive surgery for diabetic neuropathy was of poor quality and design. They recommended the treatment be classified as unproven.3
The next year, another group published a commentary in Diabetes Care outlining the authors’ concerns about Dellon’s approach.4 These included that surgical decompression of nerves was being advocated as “the treatment” for diabetic peripheral neuropathy (the charge Dellon denied above); that the diagnostic approach advocated by Dellon lacks sensitivity and specificity; and that the quality of clinical trials to that point had been poor. They concluded surgical decompression should not be recommended until better studies had been done.
The next year, in 2008, the authors of a Cochrane review assessed the studies related to nerve decompression, found eight they deemed relevant, and reported that they’d “failed to identify a single randomized controlled trial or any other well designed prospective study…that showed improvements in predefined endpoints after decompressive surgery.”5 If that statement sounds similar to the 2006 practice advisory, that may be because the lead author—Vinay Chaudhry, MD, a Johns Hopkins professor of neurology—was the same in both cases.
LER attempted to contact several of the authors of these papers for comment, including Chaudhry, but only one responded despite repeated emails. Andrew Boulton, MD, DSc, president of the European Association for the Study of Diabetes, a professor of medicine at the University of Manchester in the UK, and a visiting professor at the University of Miami, spoke briefly with the magazine.
“There still hasn’t been a randomized controlled trial, and until there is, there is no evidence to support [the procedure],” Boulton said. “Diabetic neuropathy is not a compression neuropathy; there may even be central nervous system involvement, and it’s characterized by a progressive loss of nerve fibers.”
Asked if he thought the procedure had any validity at all, he replied, “No, nor do my friends in neurology.”
Of course, “no evidence” means one thing to some people and something else to others. The lack of randomized controlled trials (RCTs) is certainly a concern in this case. But, as LER has reported in the past, there are widely accepted medical practices for which there is little RCT support. One relevant example is the debridement of diabetic foot wounds, which is practiced universally but has never had its efficacy bolstered by randomized studies.6
Origins
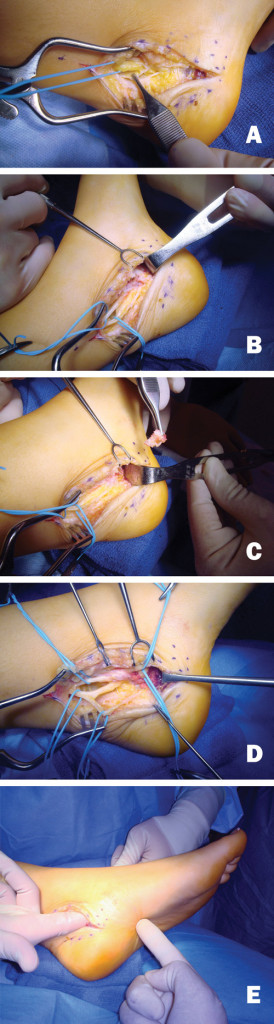
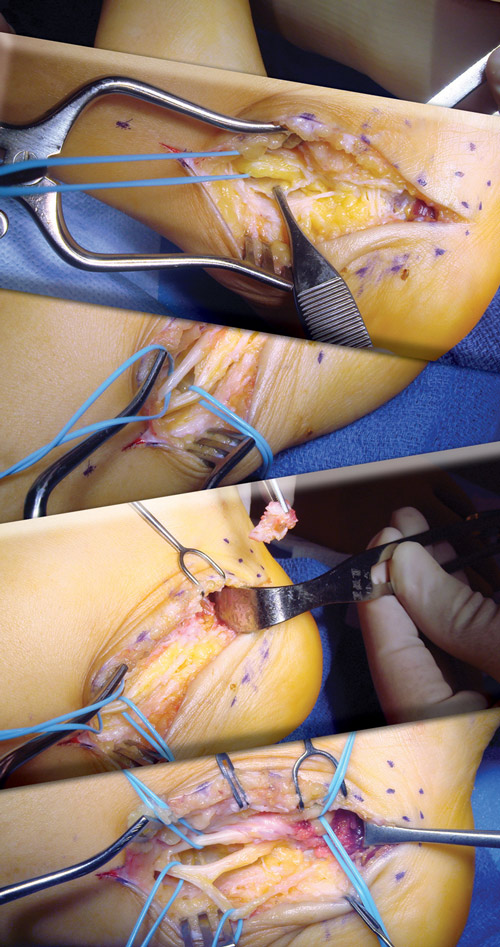
Figure 2. Dellon performed nerve decompression in a patient with a history of failed tarsal tunnel surgery performed by a different surgeon. A. The forceps holds the floor retinaculum, where there has been no previous surgery, and the tibial nerve is visible. B. The tibial nerve has been freed from scar tissue, and the medial and lateral plantar nerves are seen. C. The roofs of the medial and lateral plantar tunnels have been opened and the septum between the two tunnels is removed. D. The calcaneal nerve can be seen coming from the lateral plantar nerve. E. The surgeon’s finger can pass into the bottom of the foot. (Photos courtesy of A. Lee Dellon, MD, PhD).
As often happens when curious clinicians stray into new territory, Dellon began to develop his ideas almost by accident. In the early 1980s, he frequently operated on patients with carpal tunnel syndrome (CTS), a disproportionate number of whom were had diabetes. (Patients with diabetes develop CTS, an entrapment syndrome, at significantly higher rates than the general public—up to 20% vs 2.7%, according to some sources.7-9 After having median-nerve decompression, CTS patients reported symptoms in their thumbs and index and middle fingers had improved, but their ring and little fingers were still numb. So Dellon decompressed the ulnar nerve at the elbow, and those fingers got better too.
“Then the patients would ask if I could do the same thing for their feet, and I said no, you have neuropathy in your feet; it’s a progressive, irreversible disease,” he said. “But then, I began to wonder about it. In the hand, the median and ulnar nerves serve an area that’s like a glove; in the feet, the peroneal and tibial nerves give you the distribution of a stocking. So if I could fix a glove distribution in the hand by nerve decompression, why couldn’t I do something similar in the foot?”
After publishing his 1992 article, Dellon began working with animal models to get a better understanding of what was going on. In a 1994 study of streptozotocin-induced diabetic rats, he and his colleagues reported that, when they performed tarsal tunnel decompression at the onset of diabetes, the rats didn’t develop the typical abnormal diabetic walking pattern.10 In a recent study, he found that decompression improved nociception in rats with surgically induced sciatic nerve compression.11
Other investigators have reached similar conclusions. Maria Siemionow, MD, PhD, DSc, a professor of surgery, director of plastic surgery research, and head of microsurgery training at the Cleveland Clinic in Ohio, reported in a 2007 paper that prophylactic decompression procedures prevented the deterioration of nerve function in diabetic rats compared with diabetic controls who did not have the surgery.12
Siemionow, who attended Dellon’s course and uses his technique, is a busy surgeon—she performed the first face transplant in the US, for example—and has done many nerve decompressions in humans. In a 2006 paper, she retrospectively reviewed 32 patients (12 with diabetes, 20 without) with lower extremity peripheral neuropathy who had undergone the procedure. At a mean of 7.7 months, 90% of patients showed significant improvement in clinical outcomes.13
“In the rat models, it was clear that there was an increase in volume, an edema, of the nerves, and after decompression, there was evidence of the return of microvessel circulation,” she told LER. “It’s the same in humans; in surgery I could see that in hindered tunnels, such as the tarsal tunnel or the medial and lateral plantar tunnels, the nerves were compressed by the ligament, which acted almost like a rubber band. I could see that the color of the nerve was different from healthy nerves. And after the opening of the ligament and decompression of the tunnel, there appeared to be a restoration of circulation from the vasa nervorum.”
In Siemionow’s experience, roughly 60% of patients with diabetes who have peripheral neuropathy also have superimposed compression neuropathy, a finding with clear clinical implications.14
Support
Several of Dellon’s papers support such conclusions and suggest physiological explanations for the problem. For example, in 2007, an article described metabolic abnormalities that may increase the risk of compression syndromes—e.g., CTS or lower extremity problems including tarsal tunnel syndrome—in patients with diabetes.15
According to this theory, conversion of glucose to sorbitol increases the intraneural water content, slowing of axoplasmic transport of proteins hinders structural repair, and glycosylation of endoneurial collagen reduces perineurial gliding. The nerves become swollen and more likely to be impinged at compression sites.
Other papers have elucidated the process in more detail, noting that, when intracellular glucose levels are elevated, the hexokinase enzyme becomes fully saturated, and excess glucose is converted to sorbitol by aldose reductase. The sorbitol then pulls extracellular fluid into the neuron, causing axonal and nerve trunk swelling.16 Ultrasound studies have shown that neuropathic diabetes patients often have nerve cross-sections 25% to 50% larger than those of healthy people, which would presumably affect the nerves in compression points such as the tarsal and carpal tunnels, and which might partly explain their higher prevalence of CTS.17
Dellon’s 2007 paper also reviewed 15 studies and concluded that decompression relieved pain and restored sensation in 88% and 79% of patients, respectively. The procedure was associated with a low ulcer prevalence in 665 patients at 2.5 years—.6% in patients without a previous history of ulceration, and 2.2% in those who did have such a history.
Ulcer prevention is often perceived as a more objective outcome measure than those involving pain and sensation. In a 2012 paper, Dellon and his colleagues reported the results of a prospective multicenter trial of decompression involving 628 patients and 839 operated limbs.18 They reported that new ulcers occurred in just .2% of patients without an ulceration history, and in 3.8% of those with such a history, at two years. By contrast, statistics suggest that diabetes patients have roughly a 6.8% risk of developing foot wounds annually.19
The evidence
A further review of the evidence supporting nerve decompression is appropriate here.
In 2000, Austrian researchers (with Dellon as a coauthor) evaluated peripheral nerve decompression in one leg in 20 patients with diabetes and reported that, at a mean of two years, 69% of lower extremity nerves that underwent the procedure had improved sensibility, assessed as two-point discrimination. By contrast, 32% of nonoperated legs had a measurable progression of neuropathy.20
In 2003, researchers reported in the Journal of Foot & Ankle Surgery that, of 33 lower extremities treated with neurolysis of the common peroneal, deep peroneal, and tarsal tunnel nerves, there were good to excellent results in 90% of those with preoperative neuropathic pain, and good to excellent restoration of sensation in 66.7% of patients. Moreover, mean pain scores on a visual analog scale (VAS) were 9 preoperatively and 3.2 postoperatively.21
A 2004 retrospective analysis from the Austrian team followed 50 patients for a mean of 4.5 years after surgery and found no ulcers or amputations in the operative limbs, versus 12 ulcers and three amputations in the contralateral (nonsurgical) limbs in 15 patients.22
In 2011, researchers at Georgetown University Hospital in Washington, DC, reviewed the existing literature and concluded “the results of peripheral nerve surgery are promising for the prevention of limb loss in chronic diabetes mellitus, for diminishment of pain, and for restoration of sensory/motor function.”23
A number of papers on this topic were published in 2013. In one, coauthored by Dellon, researchers at the University of Michigan in Ann Arbor conducted a retrospective review that found 88% of patients with preoperative numbness reported improvement in sensation, and 81% reported improved balance.24 Moreover, 83% of those whose VAS pain scores were greater than 8 reported improvement.
In another, researchers in Shanghai performed neurolysis on 560 patients with neuropathy and tested various outcomes versus a control group of 40 diabetes patients without neuropathy. They found that nerve conduction velocity, quantitative sensory testing, and Toronto Clinical Scoring system data all improved significantly after decompression, though these measures remained worse than in the control group. Of the neuropathy patients, 208 (37%) had had a surface ulcer before surgery, but none had had an ulcer 18 months after surgery.25
And in yet another 2013 paper10—this one a meta-analysis— Johns Hopkins researchers reported that “observational data suggest that neurolysis significantly improves outcomes for diabetic patients with compressed nerves of the lower extremity.” (Dellon was not among the authors, nor was Chaudhry, which at least projected an impression of neutrality.) The authors noted that pain relief of more than four points on a VAS scale occurred in 91% of patients, that sensibility improved in 69%, and that postoperative ulceration and amputation rates were significantly reduced.26 And though they noted the absence of RCTs in their studies, they did clarify the record about one thing: “The approach first described by Dellon was proposed for diabetic patients with neuropathy and associated superimposed compressed nerves. Thus, the present study [limited] the population to diabetic patients with evidence…of superimposed nerve compression of the LEs.”
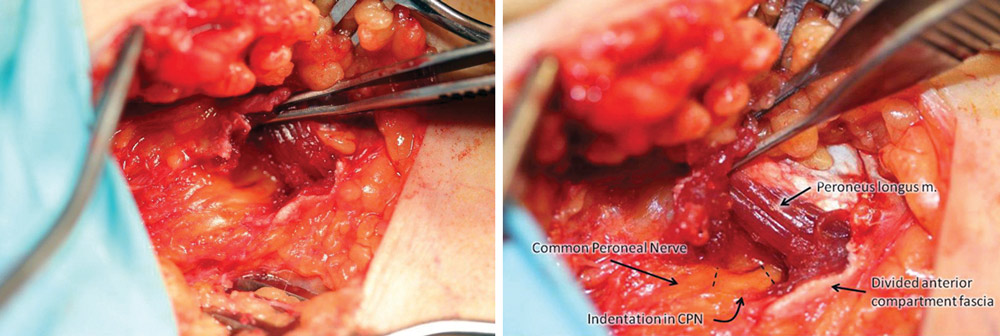
Figure 3. Photos illustrate release of common peroneal nerve. (Photographs courtesy of D. Scott Nickerson, MD.)
A review of Dellon’s papers supports this assertion—which, as noted, he made in his conversation with LER. For example, in a 2008 paper describing his approach,2 under the subhead “Surgical Indications / Contradictions,” number two on the list (an indication) is: “Physical findings of a compressed peripheral nerve at the known sites of anatomic narrowing.”
Personal experience
D. Scott Nickerson, MD, a retired orthopedic surgeon in Sheridan, WY, who suffers from diabetic neuropathy himself, found that Dellon’s theories matched his own experience. Many years ago, he was performing a transmetatarsal amputation on a patient with such complete neuropathy that no anesthesia was required. But then, as he began to remove the little toe, the patient reported pain. Nickerson puzzled over this until it dawned on him that the fifth toe was served by the sural nerve, which runs outside of the entrapment zone in the ankle. This impression was reinforced when his own right foot eventually went numb due to neuropathy, but not his left.
“These things were not consistent with length-dependent axonopathy,” he said. “They suggested individual nerve entrapment, and that made me think Dellon’s theory could make sense, that we needed to test it.”
Nickerson felt, though, that subjective outcome measures were insufficient, and was one of the first to measure the surgery’s effect on ulcer and amputation rates instead.
“Every new idea, anything demanding a change in paradigms, must justify itself with strong scientific evidence, and pain and sensibility aren’t strong enough,” he told LER.
In a recent paper, for example, Nickerson reported long-term follow-up data on 65 diabetes patients who had received decompression surgery in 75 limbs.27 Annual ulcer recurrence risk was 4.28% per patient-year at 2.49 years of follow-up. Moreover, of the 75 legs that received neurolysis, 12% developed an ulcer, whereas, of the 53 contralateral legs that were not operated on, 30% ulcerated and three were amputated. The sample size wasn’t large, but the statistics—a drop in ulcer risk of 60% with surgery—were compelling. In a report on long-term follow-up of the same cohort published in the Journal of the American Podiatric Medical Association (JAPMA)28 last fall, Nickerson and his colleagues reported that cumulative risk after an additional three years was 2.6% per patient year. In 42 patients who received unilateral lower extremity nerve decompressions, operated legs had a 1.6% per patient-year risk of reulceration, whereas the control legs had a 21% rate and required three amputations by 12 months’ follow-up.
Other practitioners have similar stories, even if they aren’t backed up by hard data.
“I’ve done about a thousand of these procedures in the past twelve years,” said Stephen Barrett, DPM, who practices in Phoenix and is an adjunct professor at the Arizona School of Podiatric Medicine. “I can tell you that, since 2004, eighty-five percent of patients have come back to have surgery on the other leg, and people don’t subject themselves to operations if they haven’t improved. That’s not evidence-based medicine—it’s Level IV from a research standpoint—but it’s significant from the point of view of the patient.”
Balancing the extremes
“You have the neuro-evangelists and the neuro-skeptics,” said David Armstrong, DPM, MD, PhD, a professor of surgery and codirector of the Southern Arizona Limb Salvage Alliance (SALSA) at the University of Arizona College of Medicine in Tucson. He added that he is a “neuro-agnostic.”
“Nerve entrapment isn’t the primary etiology of diabetic neuropathy, but I believe there are patients who may benefit from this procedure even if it doesn’t help everyone,” Armstrong said.
Armstrong acknowledged that patients with diabetic neuropathy may be at greater risk for compression syndromes than people without diabetes, particularly in the lower extremities.
“Because of that [neuropathy], any degree of anatomic disease or compression will be highlighted clinically in these patients. There is clearly pathology associated with both edema and axonal loss, and both of those likely put people at greater risk for peripheral entrapments. But that entrapment is the peripheral manifestation of a systemic problem. That means you may be able to ameliorate the manifestation of the problem to some degree, but you will not cure it,” he said.
Credibility
Although none of the studies cited here are RCTs (the Shanghai study comes closest), it strains credibility to suggest that, taken together, they offer no evidence of efficacy. In any case, three randomized clinical trials of the procedure are well underway, and researchers may soon clarify the question. Investigators at the University of Texas Southwestern Medical Center in Dallas and at the University of Sault St. Marie in Canada have completed their data collection and will presumably publish results within a year or two (they declined to divulge any preliminary information to LER). Another study in the Netherlands continues, and SALSA will participate in a US-based multicenter trial along with five private practices.
“Ultimately we want to enroll one hundred twenty patients, of which our center will provide forty,” said Tim Rankin, MD, a fourth-year University of Arizona surgical resident who will manage SALSA’s participation. The primary outcome measure will be diabetic foot ulcer recurrence.
“I think the operation has shown promising results,” said Rankin. “Hopefully this trial will provide the necessary data to clarify morbidity, mortality, and associated costs.”
The future
When the results of the randomized trials are reported, things may settle down. On the other hand, they may not. By his own account, Dellon has trained 350 surgeons in the technique, which is offered in 44 states and 23 countries. This has led to grumbling in the field about the relationship between science and commerce. For example, in the Diabetes Care commentary noted earlier, the authors groused that Dellon’s hypotheses had “spawned an entire industry,” and one wonders what will happen to that industry if the results of the clinical trials are negative.
“People used to say that Dellon was just in it to make a lot of money,” said Nickerson. “But I think people are skeptical mainly because this calls for a different paradigm, and that’s why we need more objective studies.”
It’s also entirely possible that the randomized trials will produce positive results, of course. In any event, David Armstrong expressed a convincing formula for success.
“Let’s understand,” he said. “If the passionate do the surgery, the dispassionate have to assess the outcomes.”
Cary Groner is a freelance writer based in the San Francisco Bay area.
1. Dellon A. Treatment of symptomatic diabetic neuropathy by surgical decompression of multiple peripheral nerves. Plast Reconstr Surg 1992;89(4):689-697.
2. Dellon A. The Dellon approach to neurolysis in the neuropathy patient with chronic nerve compression. Handchir Mikrochir Plas Chir 2008;40:1-10.
3. Chaudhry V, Stevens J, Kincaid J, So Y. Practice advisory: utility of surgical decompression for treatment of diabetic neuropathy: report of the therapeutics and technology assessment subcommittee of the American Academy of Neurology. Neurology 2006;66(12):1805-1808.
4. Cornblath D et al. Surgical decompression for diabetic sensorimotor polyneuropathy. Diabetes Care 2007;30(2):421-422.
5. Chaudhry V, Russell J, Belzberg A. Decompressive surgery of lower limbs for symmetrical diabetic peripheral neuropathy. Cochrane Database Sys Rev 2008;16(3):CD006152.
6. Groner C. Despite few solid studies, ulcer debridement thrives. LER 2012;4(10):51-62.
7. Vinik A. Diabetic neuropathy: pathogenesis and therapy. Am J Med 1999;107(2B):17S-26S.
8. Atroshi I, Gummesson C, Johnsson R, et al. Prevalence of carpal tunnel syndrome in a general population. JAMA 1999;281(2):153-158.
9. Ramchurn N, Mashamba C, Leitch E, et al. Upper limb musculoskeletal abnormalities and poor metabolic control in diabetes. Eur J Int Med 2009;20(7):718-721.
10. Dellon A, Dellon E, Seiler W. Effect of tarsal tunnel decompression in the streptozotocin induced diabetic rat. Microsurgery 1994;15(4):265-268.
11. Barac S, Jiga LP, Barac B, et al. Hindpaw withdrawal from a painful thermal stimulus after sciatic nerve compression and decompression in the diabetic rat. J Reconstr Microsurg 2013;29(1):63-66.
12. Siemionow M, Sari A, Demir Y. Effect of early nerve release on the progression of neuropathy in diabetic rats. Ann Plast Surg 2007;59(1):102-108.
13. Siemionow M, Alghoul M, Molski M, Agaoglu G. Clinical outcome of peripheral nerve decompression in diabetic and non-diabetic peripheral neuropathy. Ann Plast Surg 2006;57(4):385-390.
14. Siemionow M, Zielinski M, Sari A. Comparison of clinical evaluation and neurosensory testing in the early diagnosis of superimposed entrapment neuropathy in diabetic patients. Ann Plast Surg 2006;57(1):41-49.
15. Dellon A. Neurosurgical prevention of ulceration and amputation by decompression of lower extremity peripheral nerves in diabetic neuropathy: update 2006. Acta Neurochir Suppl 2007;100:149-151.
16. Sessions J, Nickerson D. Biologic basis for nerve decompression surgery in diabetic focal entrapment peripheral neuropathy. J Diabetes Sci Technol 2014;8(1): in press.
17. Watanabe T, Ito H, Morita A, et al. Sonographic evaluation of the median nerve in diabetic patients: comparison with nerve conduction studies. J Ultrasound Med 2009;28(6):727-734.
18. Dellon AL, Muse VL, Nickerson DS, et al. Prevention of ulceration, amputation, and reduction of hospitalization: outcomes of a prospective multicenter trial of tibial neurolysis in patients with diabetic neuropathy. J Reconstr Microsurg 2012;28(4):241-246.
19. Wu S, Armstrong D. Risk assessment of the diabetic foot and wound. Int Would J 2005;2(1):17-24.
20. Aszmann O, Kress K, Dellon A. Results of decompression of peripheral nerves and diabetics: a prospective, blinded study. Plast Reconstr Surg 2000;106(4):816-822.
21. Wood W, Wood M. Decompression of peripheral nerves for diabetic neuropathy in the lower extremity. J Foot Ankle Surg 2003;42(5):268-275.
22. Aszmann O, Tassler P, Dellon A. Changing the natural history of diabetic neuropathy: incidence of ulcer/amputation in the contralateral limb of patients with a unilateral nerve decompression procedure. Ann Plast Surg 2004;53(6):517-522.
23. Ducic I, Felder J, Iorio M. The role of peripheral nerve surgery in diabetic limb salvage. Plast Reconstr Surg 2011;127 (Suppl 1):259S-269S.
24. Valdivia JM, Weinand M, Maloney CT Jr, et al. Surgical treatment is superimposed, lower extremity, peripheral nerve entrapment for diabetic and idiopathic neuropathy. Ann Plast Surg 2013;70(6):675-679.
25. Zhang W, Li S, Zheng X. Evaluation of the clinical efficacy of multiple lower extremity nerve decompression in diabetic peripheral neuropathy. J Neurol Surg A Cent Eur Neurosurg 2013; 74(2):96-100.
26. Baltodano PA, Basdag B, Bailey CR, et al. The positive effect of neurolysis on diabetic patients with compressed nerves of lower extremities: a systematic review and meta-analysis. Plast Reconstr Surg Glob Open 2013;1:e24.
27. Nickerson DS. Low recurrence rate of diabetic foot ulcer after nerve decompression. J Am Podiatr Med Assoc 2010;100(2):111-115.
28. Nickerson D, Rader A. Low long-term risk of foot ulcer recurrence after nerve decompression and a diabetes neuropathy cohort. J Am Podiatr Med Assoc 2013;103(5):380-386.








