Figure 1. The tendon to bone insertion site of mice shows the mineral gradient of the enthesis site. From Schwartz AG, Pasteris JD, Genin GM, et al. Mineral distributions at the developing tendon enthesis. PLoS One. 2012;7(11):e48630. Use is per Creative Commons Attribution License.
Formed during development, this efficient attachment system cannot be recreated during healing.
By Nicole Wetsman
Many common injuries, such as Achilles tendon and rotator cuff tears, require surgical procedures that reattach the affected tendon to the bone.
Stavros Thomopoulos, PhD, director of Carroll Laboratories for Orthopedic Surgery at Columbia University in New York, compares it to trying to attach a rope to a piece of cement. “You have tendon on one side, and bone on the other. They’re very different materials,” he says. “The body has evolved these really efficient attachment systems to solve this mechanical problem.”
In a healthy joint, the intersection between the two structures is referred to as the enthesis. That junction is extremely specialized, and—even with the most skillful of surgeries—it does not regrow if it’s been damaged. Surgeons can effectively reattach tendons and bone, Thomopoulos says, but they can’t recreate the conditions that build the enthesis. “All of these really great features that form during development, unfortunately, none of them get recreated during healing,” he says.
A unique structure creates a surgical challenge
The complex structure of the enthesis develops after birth, as the changing expression levels of various growth factors trigger the growth of the cell types that form the juncture. However, the specifics of enthesis development are still poorly understood—one reason that medically recreating it after damage is so challenging.
On the bone side, the enthesis forms an attachment over a wide surface area, which allows it to distribute force and tension through the joint. “It’s incredibly effective at transferring the load from muscle across the tendon,” Thomopoulos says. The enthesis itself has a gradient of cell tissue: at the point it intersects with tendon, it contains connective tissue and tendon cells. It transitions to uncalcified fibrocartilage cells, then to calcified fibrocartilage cells, and finally, to bone (see Figures). The mineral content of the structure increases as it moves from tendon to bone.1 “There’s a linear increase,” he says. “The two fit together like a puzzle.” 2
Molecular analysis of the cells in the enthesis and at the intersection between tendon and bone shows a wide range of cell types with varying levels of gene expression. In the tendon, for example, there was an increase in expression of genes involved in cell adhesion. Cells in the enthesis had enriched expression of genes involved in collagen organization.3
The enthesis is a robust structure, Thomopoulos notes, and it’s rarely itself damaged. “The healthy enthesis is really built to take a lot of stress,” he says. Injuries are far more likely to arise in the bone or in the tendon on either side of the junction. The surgical problem arises, he says, because there often isn’t enough tissue available to attach the tendon back to the enthesis. “There’s no way a surgeon would repair tendon-to-tendon in that case.” That’s why they have to bypass the enthesis and create a new link between the bone and the tendon.
To do so, surgeons use bolts and anchors to stitch the two structures together. They work to create a wide attachment point that spreads the mechanical load that the joint will need to hold, Thomopoulos says. Surgeons might use adhesives to spread out the forces, as well. “The problem is, the right kind of adhesives don’t exist: crazy glue is strong but brittle. Medical glues, they’re really stretchy, but they’re not strong enough.”
Surgeons are able to successfully recreate the attachment—but after the procedure, the junction heals as scar tissue, not the previously existing structure of the enthesis. The mineral gradient and gradually transitioning cell types do not return during the healing process.
“We used to think healed tendon to bone would match what was normally there,” says Augustus Mazzocca, MS, MD, professor in the department of orthopedic surgery and director of the musculoskeletal institute at the University of Connecticut. “If you cut your skin, a lot of times, it doesn’t form a scar. If you break your bone, it heals, and looks normal. The tendon-to-bone connection doesn’t do that.” Even when the joint heals, he says, and brings back normal strength and range of motion, the tendon-to-bone junction is never the same.
The challenges associated with tendon-to-bone surgical procedures, and the current inability to recreate the enthesis, are likely one of the reasons that failure rates for these procedures are so high. Rotator cuff repairs in the shoulder have particularly high failure rates—around 30% of repairs fail, Mazzocca says, and some estimates put the failure rate near 95%.2 The size of most rotator cuff tears can make them more likely to fail, says Robert LaPrade, MD, PhD, a complex orthopaedic knee and sports medicine surgeon in Colorado.
However, procedures in the lower extremity, like anterior cruciate ligament and patellar tendon repair, have high failure rates as well: rates of failure in the patellar tendon have been reported to be as high as 66%.4 Failures can result in unresolved pain, even years after a procedure, or, in some cases, might require additional surgical procedures.
Innovations in treatment

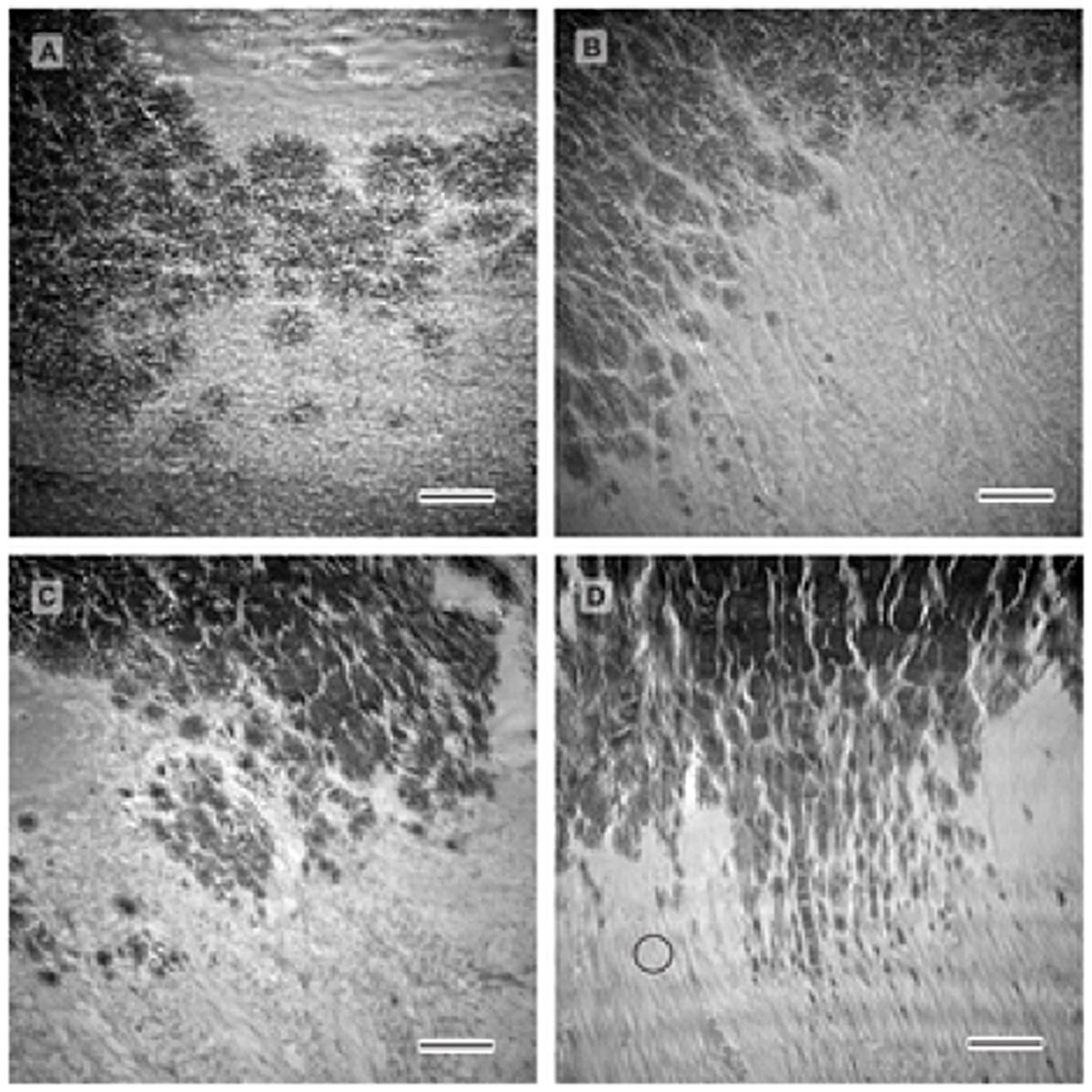
Figure 2. The stained cut of pig Achilles tendon and enthesis tissue shows the distinction between cells of the tendon, enthesis and bone. a) Enthesis cryocut section was stained for cells using SYTO® 13. Cells are depicted cyan, confocal reflection is depicted magenta. Scale bar corresponds to 150 μm. b) Scheme of cell arrangement observed at the enthesis. c) Cells within the interface are round shaped and are often arranged in pairs. Scale bar corresponds 50 μm. d) Cells residing within tendon are longitudinally arranged along the axis of tension in strings between tendon fibers. Scale bar corresponds 50 μm. From Kuntz LA, Rossetti L, Kunold E, et al. Biomarkers for tissue engineering of the tendon-bone interface. PLoS One. 2018;13(1):e0189668. Use is per Creative Commons Attribution License.
Because of the failure rates and the challenges associated with tendon-to-bone reattachments, efforts are underway to develop new tools to assist in these surgical procedures. Scientists are working to develop better adhesives, for example, Thomopoulos says. “We’re looking toward marine biology, to things like barnacles and mussels—they’ve formed these really neat systems for attaching themselves to surfaces,” he says. “We’re working with polymer chemists to come up with a mimic of that approach.”
In patellar tendon repairs, research has shown that reducing the length of post-surgical immobilization and allowing for early motion at the joint can improve tendon healing and prevent complications. As such, novel surgical techniques that allow for patients to move earlier have been used in clinical practice.4
Other innovative strategies have included the use of scaffolding and grafts at the tendon-to-bone connection, which are seeded with protein growth factor and mesenchymal stem cells, which are typically isolated from bone marrow and can differentiate into bone and cartilage. However, these strategies have had limited success. “We really do not have enough stem cells present in these bone marrow treatments to make a difference in most circumstances, so we are relying on a concentration of growth factors, which may or may not help a specific healing response,” LaPrade says.
Long-term solutions, Thomopoulos says, will most likely result from the use of biologic materials created from cells and proteins. “If we can push the healing process toward regeneration and away from scar—that’s the idea,” he says. “We have to suppress the scar response, and at the same time, we have to promote regeneration.”
Adding platelet-rich plasma to and around the repair is another promising approach. The substance is a platelet and growth factor-heavy plasma isolated from the patient’s own blood—which helps ensure its safety—and it’s been used clinically to treat injuries within tendon and ligaments. Platelet-rich plasma contains growth factors like platelet-derived growth factor and insulin-like growth factor-I, which are involved in tendon and ligament healing.5
Preclinical studies in animal models have shown that platelet-rich plasma can improve healing in Achilles tendon repairs. For example, a 2015 study in rabbits found that animals treated with injections of the plasma healed with more collagen fibers and better arrangement of fibers than those who did not receive injections.6
Mazzocca has had success using platelet-rich plasma and thrombin, an enzyme important for clotting, to create a clot underneath and around the point of repair during surgical procedures in the rotator cuff, he says. “Clinically, it’s shown some encouraging results,” he says.
However, reviews of the clinical literature find that there is not sufficient evidence to recommend platelet-rich plasma during surgical procedures.7 Clinical evidence to date on Achilles tendon repairs, for example, is mixed. One study in 30 patients, published in 2016, found that a group treated with platelet-rich plasma during and after surgery did not have significantly different outcomes than a group that did not receive the plasma.8 However, a second 2016 study of 36 patients found that a group treated with the plasma had improved functional outcomes after surgery.9
Mixed results, Thomopoulos says, are due to the limited research that has been done to date.
“As it stands now, there’s not that much evidence of efficacy of any of these for tendon-to-bone repairs,” Thomopoulos says. “Part of the issue is that they’re very much first generation: platelet-rich plasma, for example, is a little bomb of growth factors. But we don’t know exactly what’s in their yet.”
Further research to refine the use of biologics will hopefully push surgical procedures forward. “We all hope that biologics significantly improve the ability for us to both speed healing and improve healing of torn tendons over time,” LaPrade says. “I believe that biologics are the next great advancement in sports medicine after the arthroscope. However, we are still in our infancy in the use of biologics.”
Continued basic science research, in the laboratory and in animal models, will be key to understanding the underlying biology of the enthesis. That work will help continue to drive innovation in treatment approaches, Thomopoulos says. “If we can define how the body builds the enthesis from scratch, we’ll have the instruction manual to get it back in the adult.”
Nicole Wetsman is a freelance writer in New York City.
- Schwartz AG, Pasteris JD, Genin GM, Daulton TL, Thomopoulos S. Mineral distributions at the developing tendon enthesis. PLoS One. 2012;7(11):e48630.
- Apostolakos J, Durant TJ, Dwyer CR, et al. The enthesis: a review of the tendon-to-bone insertion. Muscles Ligaments Tendons J. 2014;4(3):333–342.
- Kuntz LA, Rossetti L, Kunold E, et al. Biomarkers for tissue engineering of the tendon-bone interface. PLoS One. 2018;13(1):e0189668.
- Black JC, Ricci WM, Gardner MJ, et al. Novel augmentation technique for patellar tendon repair improves strength and decreases gap formation: a cadaveric study. Clin Orthop Relat Res. 2016;474(12):2611–2618.
- Yuan T, Zhang CQ, Wang JH. Augmenting tendon and ligament repair with platelet-rich plasma (PRP). Muscles Ligaments Tendons J. 2013;3(3):139–149.
- Allahverdi A, Sharifi D, Takhtfooladi MA, Hesaraki S, Khansari M, Dorbeh SS.. Evaluation of low-level laser therapy, platelet-rich plasma, and their combination on the healing of Achilles tendon in rabbits. Lasers Med Sci. 2015;30(4):1305.
- Le ADK, Enweze L, DeBaun MR, Dragoo JL. Current clinical recommendations for use of platelet-rich plasma. Curr Rev Musculoskelet Med. 2018;11(4):624–634.
- De Carli, A., Lanzetti, R.M., Ciompi, A. et al. Can platelet-rich plasma have a role in Achilles tendon surgical repair? Knee Surg Sports Traumatol Arthrosc. 2016;24(7): 2231–2237.
- Zou J, Mo X, Shi Z, et al. A prospective study of platelet-rich plasma as biological augmentation for acute Achilles tendon rupture repair. Biomed Res Int. 2016;2016:9364170.