 New research suggests that vasti muscle activation in women varies throughout the menstrual cycle, adding to the growing body of literature pointing to a connection between sex hormones and knee injury risk. The exact nature of that relationship, however, remains unclear.
New research suggests that vasti muscle activation in women varies throughout the menstrual cycle, adding to the growing body of literature pointing to a connection between sex hormones and knee injury risk. The exact nature of that relationship, however, remains unclear.
By Matthew S. Tenan, MA, ATC, and Lisa Griffin, PhD
Knee pain is the most commonly reported joint ailment in the US.1 Women are known to have higher incidence rates of both patellofemoral pain2 and anterior cruciate ligament (ACL) tears3 compared with men. The mechanisms underlying the higher incidence of knee pain and injury in women are likely multifactorial. Once mechanisms are identified, more effective treatments and preventive measures can be designed to decrease injury rates in women. Potential contributors to this sex discrepancy that have been explored in recent studies include the sex effects of the quadriceps angle (Q angle), changes in the material properties of muscles and ligaments attributed to sex hormones, and the effect of sex hormones on the nervous system controlling lower extremity movement.
The sex discrepancy in knee injury is commonly attributed to increased Q angle in women.4 The Q angle is formed by two frontal plane lines drawn from the anterior superior iliac spine to the center of the patella and a second line from the center of the patella to the center of the tibial tubercle. In theory, this increased Q angle increases tibial torsion and laterally vectors the patella.5 Simulation-based models have corroborated the increased lateral force vector of the patella6 and cadaver studies have shown that increasing Q angle results in an increased lateral patellar translation of approximately 4 mm to 6 mm along with increased patellar tilt.7 However, when patients are asked to contract their quadriceps in an magnetic resonance imaging8 or computed tomography scanner,9 researchers have found no relationship between Q angle and lateral patellar displacement or patellar tilt.
A high Q angle has also been shown to decrease knee abduction moments in running,10 making it unlikely to increase rates of patellofemoral pain. Furthermore, the Q angle has been shown to be a very poor predictor of knee injury in both large prospective studies11 and smaller cross-sectional studies.12-14 Despite common parlance attributing sex differences in knee injury and pain to the differences in Q angle, there is limited empirical support for this notion.
Hormonal factors
Instead of passive structural reasons for the injury rate discrepancy, newer research has tended to focus on the effects of sex hormones on the mechanical properties of tendons, ligaments, and muscles. Estrogen receptors have been identified within skeletal muscle,15 indicating a potential avenue for sex hormones to alter the contractile properties of the tissue. Bell et al16 found that women display decreases in musculotendinous stiffness with increases in estrogen, an effect that was not observed in men. Changes in musculotendinous stiffness of the hamstrings are important because this muscle group can limit the rate of loading and total loading on the ACL. The estrogen association, however, was observed during the early follicular phase of the menstrual cycle, when estrogen levels are at their lowest. It may not be a simple extrapolation to say that musculotendinous stiffness continues to decrease as estrogen levels increase fourfold over the menstrual cycle. Indeed, a second study by Bell et al17 found that muscle stiffness was not different during periods of high estrogen (luteal phase) compared with periods of low estrogen (early follicular phase). Furthermore, Blackburn et al18 showed that while women have lower levels of musculotendinous stiffness compared to men, this was likely related to differences in muscle cross-sectional area between the sexes. Therefore, preliminary reports suggest the effect of estrogen on the material properties of musculotendinous structures is minimal.
The ACL has estrogen receptors,19 and it has been demonstrated that stimulation of these receptors alters collagen RNA expression.20 The cellular finding has prompted a number of researchers to explore whether the mechanical properties of the ACL are altered by sex hormones. Clinical reports have produced conflicting results when examining anterior knee displacement across the menstrual cycle.21,22 However, highly invasive animal research conclusively demonstrates that knee ligament and tendon properties are not affected by estrogen in either rats23 or primates.24 At the present time, there is no conclusive evidence that sex hormones alter the mechanical properties of muscle, tendon, or ligament in vitro or in vivo.
Sex hormone oscillations in the blood can affect the central nervous system because hormones can easily traverse the blood-brain barrier due to their high lipid solubility.25 Estrogen binds to estrogen receptor α sites on γ-aminobutyric acid (GABA)-releasing neurons, resulting in decreased GABA transmission26 and enhanced cerebellar neuron discharge during rodent locomotion.27 Due to the inhibitory nature of GABA, it is unsurprising that multiple studies utilizing transcranial magnetic stimulation have found increased corticospinal tract excitability during the late follicular phase of the menstrual cycle, when estrogen levels are at their highest.28-30 Unlike estrogen, progesterone can act like GABA (by activating GABA receptors) and directly increase neural inhibition.31 Again, this finding in rodent models has been corroborated by human research with transcranial magnetic stimulation studies showing decreased corticospinal tract excitability in the mid-luteal phase of the menstrual cycle, when progesterone is at its peak.29 The basic research clearly indicates that sex hormones have profound effects on the nervous system, but does this translate to changes in the recruitment of muscles in the periphery?
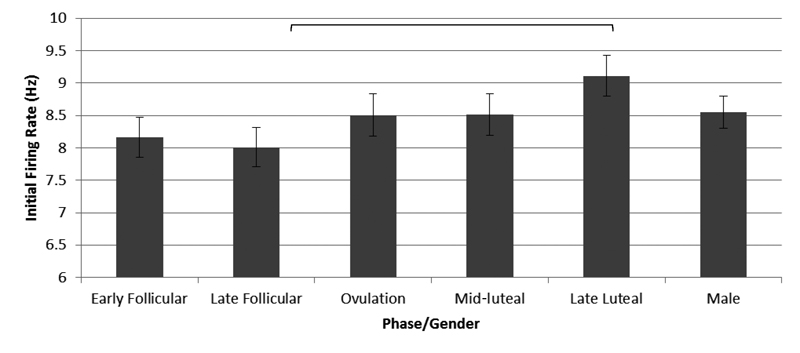
Figure 1. Motor unit firing rates at recruitment. The late luteal phase firing rate is significantly higher than firing rates at early follicular and late follicular (p < .05) after controlling for force generation. (Adapted using pooled data from Tenan et al.32)
Our research
Our research group recently investigated the effect of the menstrual cycle on changes in the recruitment and activation of the muscle fibers of the vastus medialis (VM) and the vastus medialis oblique (VMO).32 Based on muscle attachment sites, the VM predominately performs knee extension while the VMO is theorized to medialize the patella. Proper control of the patella is essential to limiting the occurrence of patella subluxation/dislocation and patellofemoral pain syndrome. To our knowledge, this is the first and only study
examining the recruitment and discharge patterns of muscle fibers across the menstrual cycle.
Eighteen people (seven women) participated in the study. All participants were free from current injury and reported no chronic musculoskeletal or nervous system ailments. All the women had a history of normal menstrual cycles and had not been on hormonal birth control for at least six months prior to the study.
The male participants took part in one study visit. The female participants attended five study visits at distinct time points in the menstrual cycle corresponding to the five menstrual phases: early follicular, late follicular, ovulation, mid-luteal, and late luteal. Our study used fine-wire electromyography to examine the electrical depolarization of individual motor units in the VM and VMO. These wires, about the thickness of a human hair, were inserted just medial to the patella (VMO) and 7 cm superior (VM) on the participant’s dominant leg. The participants were seated with their hip and knee angles at 90° and their dominant ankle affixed to a strain gauge to measure knee extensor force generation. The force was displayed on a computer monitor positioned in front of them. The participants were trained in performing a ramp contraction of the knee extensors by tracing a target line of force production up to 30% of maximal voluntary force.
Upon completion of data collection for all participants, the action potential discharges were identified for each motor unit. That motor unit was considered “recruited” when it had discharged four times at consecutive regular intervals. We quantified the rate of those first four discharges (in Hz) and the relative force at which the motor unit was recruited. Using this method, we were able to analyze a total of 510 individual motor units.
Due to the angle of pennation of the VMO fibers, the VMO acts medially on the patella, whereas the VM predominantly extends the knee.33 Therefore, our interest was in how the fibers of the two muscles are activated at the time of recruitment and how a participant’s sex, menstrual cycle phase, or both may affect this activation.
When we pooled the data for the women across the menstrual cycle, we found the muscle fibers of the VMO had a significantly lower discharge rate compared to the VM. This difference was not observed in our male cohort. The female data was then sectioned out by menstrual cycle phase. The results showed that, overall, the discharge rate of both the VM and VMO increased from the early follicular phase through the late luteal phase (pooled data are shown in Figure 1).
The VMO fiber discharge was significantly lower than the VM only in the ovulatory and mid-luteal phases of the menstrual cycle. Interestingly, Wojtys et al34,35 implicated the ovulatory phase as a time of increased risk of ACL tear compared with the follicular phases, though there is considerable disagreement in the field as to the accuracy of the reported increased risk in this phase.36,37
Clinical implications
The timing of the changes in muscle fiber discharge suggest that progesterone is the primary mediator in alterations to muscle fiber discharge across the menstrual cycle, though it does not preclude effects from estrogen or other sex hormones. The results of our study prompt as many questions as they answer. Is it more or less “desirable” to have differential activation of the VM and VMO? What are the possible repercussions of the increase in overall muscle discharge rate across the menstrual cycle? Perhaps more importantly, what are the clinical implications of the variability in muscle activation we observed across the menstrual cycle?
Cardiologists have known for years that variability, within normal ranges, indicates a healthy cardiovascular system.38 The menstrual cycle itself is a result of variations in the secretion of sex hormones in women, and the repeated occurrence of these variations as part of a regular menstrual cycle is indicative of a healthy reproductive system. The observed variability may be “good,” as indicated by our apparently healthy female cohort.
Conversely, gait stride time variability in the elderly is highly predictive of falls incidence.39 It’s possible that variability itself is the culprit predisposing women to higher rates of knee injury. Consider the analogy of continually landing on an unstable surface; the body may continually be adjusting to the changes in discharge pattern, decreasing the “stability” of the system. In the context of the present analogy, our study population may simply be the few women who have managed not to “fall on the unstable surface” (i.e., have knee pain as a result of variability in muscle recruitment). This proposal is, of course, hypothetical in nature and not supported by the current literature. Rather, the analogy is being used to stimulate conversation and possibly controversy with regard to the potential effects of sex hormones on motor control and injury incidence.
 Recall that both the male and female cohorts in our study included healthy individuals with no history of injury or immobilization. Our study also was not longitudinal in nature, meaning we have no intention of tracking our participants to determine if they become injured. Our study, using only women with normal menstrual cycles, may also not represent the majority of active young women. The effect of hormonal contraception on the nervous system is largely unknown. Hormonal contraception suppresses the natural sex hormones by introducing synthetic sex hormones that may or may not mimic their natural analogs’ effects on the nervous system. Additionally, some highly active women may be amenorrheic or oligomenorrheic, and the effect of those conditions on the nervous system is also unknown.
Recall that both the male and female cohorts in our study included healthy individuals with no history of injury or immobilization. Our study also was not longitudinal in nature, meaning we have no intention of tracking our participants to determine if they become injured. Our study, using only women with normal menstrual cycles, may also not represent the majority of active young women. The effect of hormonal contraception on the nervous system is largely unknown. Hormonal contraception suppresses the natural sex hormones by introducing synthetic sex hormones that may or may not mimic their natural analogs’ effects on the nervous system. Additionally, some highly active women may be amenorrheic or oligomenorrheic, and the effect of those conditions on the nervous system is also unknown.
In conclusion, considerable progress is being made toward understanding the apparent sex discrepancies in rates of knee injury. The underlying cause of the discrepancy is likely multifactorial. Our research provides an early glimpse into the effect of the menstrual cycle on muscle activation in healthy eumenorrheic women. Sex hormones may also affect the musculotendinous and ligamentous structures surrounding the knee, though the research in this area is still inconclusive. Clinicians are encouraged to inquire as to the hormonal status of their female patient base during the course of treatment as anecdotal reports and population surveys may be the most practical way to guide future research toward decreasing the sex discrepancy in knee injury.
Matthew S. Tenan, MA, ATC, is a doctoral candidate at the University of Texas at Austin and a research neurophysiologist at the United States Army Research Laboratory in Aberdeen, MD. Lisa Griffin, PhD, is an associate professor in the Department of Kinesiology & Health Education at the University of Texas at Austin.
1. Centers for Disease Control and Prevention. QuickStats: Percentage of adults reporting joint pain or stiffness. MMWR 2008;57(17):467.
2. Boling M, Padua D, Marshall S, et al. Gender differences in the incidence and prevalence of patellofemoral pain syndrome. Scand J Med Sci Sports 2010;20(5):725-730.
3. Prodromos CC, Han Y, Rogowski J, et al. A meta-analysis of the incidence of anterior cruciate ligament tears as a function of gender, sport, and a knee injury-reduction regimen. Arthroscopy 2007;23(12):1320-1325.
4. Woodland LH, Francis RS. Parameters and comparisons of the quadriceps angle of college-aged men and women in the supine and standing positions. Am J Sports Med 1992;20(2):208-211.
5. Insall J, Falvo KA, Wise DW. Chondromalacia patellae. A prospective study. J Bone Joint Surg Am 1976;58(1):1-8.
6. Elias JJ, Bratton DR, Weinstein DM, Cosgarea AJ. Comparing two estimations of the quadriceps force distribution for use during patellofemoral simulation. J Biomech 2006;39(5):865-872.
7. Mizuno Y, Kumagai M, Mattessich SM, et al. Q-angle influences tibiofemoral and patellofemoral kinematics. J Orthop Res 2001;19(5):834-840.
8. Sheehan FT, Derasari A, Fine KM, et al. Q-angle and J-sign: indicative of maltracking subgroups in patellofemoral pain. Clin Orthop Relat Res 2010;468(1):266-275.
9. Biedert RM, Warnke K. Correlation between the Q angle and the patella position: a clinical and axial computed tomography evaluation. Arch Orthop Traum Surg 2001;121(6):346-349.
10. Park SK, Stefanyshyn DJ. Greater Q angle may not be a risk factor of patellofemoral pain syndrome. Clin Biomech 2011;26(4):392-396.
11. Boling MC, Padua DA, Marshall SW, et al. A prospective investigation of biomechanical risk factors for patellofemoral pain syndrome: the Joint Undertaking To Monitor And Prevent ACL Injury (JUMP-ACL) cohort. Am J Sports Med 2009;37(11):2108-2116.
12. Caylor D, Fites R, Worrell TW. The relationship between quadriceps angle and anterior knee pain syndrome. J Orthop Sports Phys Ther 1993;17(1):11-16.
13. Cooney AD, Kazi Z, Caplan N, et al. The relationship between quadriceps angle and tibial tuberosity–trochlear groove distance in patients with patellar instability. Knee Surg Sports Traumatol Arthrosc 2012;20(12):2399-2404.
14. Tsakoniti AE, Mandalidis DG, Athanasopoulos SI, Stoupis CA. Effect of Q-angle on patellar positioning and thickness of knee articular cartilages. Surg Radiol Anat 2011;33(2):97-104.
15. Lemoine S, Granier P, Tiffoche C, et al. Estrogen receptor alpha mRNA in human skeletal muscles. Med Sci Sports Exerc 2003;35(3):439-443.
16. Bell DR, Blackburn JT, Norcross MF, et al. Estrogen and muscle stiffness have a negative relationship in females. Knee Surg Sports Traumatol Arthrosc 2012;20(2):361-367.
17. Bell DR, Blackburn JT, Ondrak KS, et al. The effects of oral contraceptive use on muscle stiffness across the menstrual cycle. Clin J Sport Med 2011;21(6):467-473.
18. Blackburn JT, Bell DR, Norcross MF, et al. Comparison of hamstring neuromechanical properties between healthy males and females and the influence of musculotendinous stiffness. J Electromyogr Kinesiol 2009;19(5):e362-e369.
19. Liu SH, Al-Shaikh RA, Panossian V, et al. Estrogen affects the cellular metabolism of the anterior cruciate ligament: A potential explanation for female athletic injury. Am J Sports Med 1997;25(5):704-709.
20. Lee CY, Liu X, Smith CL, et al. The combined regulation of estrogen and cyclic tension on fibroblast biosynthesis derived from anterior cruciate ligament. Matrix Biol 2004;23(5):323-329.
21. Deie M, Sakamaki Y, Sumen Y, et al. Anterior knee laxity in young women varies with their menstrual cycle. Int Orthop 2002;26(3):154-156.
22. Park SK, Stefanyshyn DJ, Loitz-Ramage B, et al. Changing hormone levels during the menstrual cycle affect knee laxity and stiffness in healthy female subjects. Am J Sports Med 2009;37(3):588-598.
23. Warden SJ, Saxon LK, Castillo AB, Turner CH. Knee ligament mechanical properties are not influenced by estrogen or its receptors. Am J Physiol Endocrinol Metab 2006;290(5):E1034-E1040.
24. Wentorf FA, Sudoh K, Moses C, et al. The effects of estrogen on material and mechanical properties of the intra-and extra-articular knee structures. Am J Sports Med 2006;34(12):1948-1952.
25. Stoffel-Wagner B. Neurosteroid metabolism in the human brain. Eur J Endocrinol 2001;145(6):669-679.
26. Schultz KN, von Esenwein SA, Hu M, et al. Viral vector-mediated overexpression of estrogen receptor-alpha in striatum enhances the estradiol-induced motor activity in female rats and estradiol-modulated GABA release. J Neurosci 2009;29(6):1897-1903.
27. Smith SS, Woodward DJ, Chapin JK. Sex steroids modulate motor-correlated increases in cerebellar discharge. Brain Res 1989;476(2):307-316.
28. Smith MJ, Keel JC, Greenberg BD, et al. Menstrual cycle effects on cortical excitability. Neurology 1999;53(9):2069-2072.
29. Smith MJ, Adams LF, Schmidt PJ, et al. Effects of ovarian hormones on human cortical excitability. Ann Neurol 2002;51(2):599-603.
30. Inghilleri M, Conte A, Currà A, et al. Ovarian hormones and cortical excitability. An rTMS study in humans. Clin Neurophysiol 2004;115(5):1063-1068.
31. Callachan H, Cottrell GA, Hather NY, et al. Modulation of the GABAA receptor by progesterone metabolites. Proc R Soc Lond B Biol Sci 1987;231(1264):359-369.
32. Tenan MS, Peng Y-L, Hackney AC, Griffin L. Menstrual cycle mediates vastus medialis and vastus medialis oblique muscle activity. Med Sci Sports Exerc 2013 May 7. [Epub ahead of print]
33. Farahmand F, Senavongse W, Amis AA. Quantitative study of the quadriceps muscles and trochlear groove geometry related to instability of the patellofemoral joint. J Orthop Res 1998;16(1):163-143.
34. Wojtys EM, Huston LJ, Lindenfeld TN, et al. Association between the menstrual cycle and anterior cruciate ligament injuries in female athletes. Am J Sports Med 1998;26(5):614-619.
35. Wojtys EM, Huston LJ, Boynton MD, et al. The effect of the menstrual cycle on anterior cruciate ligament injuries in women as determined by hormone levels. Am J Sports Med 2002;30(2):182-188.
36. Vescovi JD. The menstrual cycle and anterior cruciate ligament injury risk: implications of menstrual cycle variability. Sports Med 2011;41(2):91-101.
37. Hewett TE, Zazulak BT, Myer GD. Effects of the menstrual cycle on anterior cruciate ligament injury risk: a systematic review. Am J Sports Med 2007;35(4):659-668.
38. Tsuji H, Larson MG, Venditti FJ, et al. Impact of reduced heart rate variability on risk for cardiac events. The Framingham Heart Study. Circulation 1996;94(11):2850-2855.
39. Hausdorff JM, Rios DA, Edelberg HK. Gait variability and fall risk in community-living older adults: a 1-year prospective study. Arch Phys Med Rehabil 2001;82(8):1050-1056.








