Research suggests that plantar pressure measurement can help practitioners pinpoint problematic areas, compare different orthotic devices, and improve consistency in fabrication.
By Georgeanne Botek, DPM, and Tammy M. Owings, DEng.
Central to the lower extremity management of patients with diabetes is prescription footwear. To reduce potentially harmful tissue stresses on the plantar aspect of the foot, the most direct therapeutic approach is the use of custom made insoles, which provide an interface between the foot and the shoe. Custom made insoles are the standard of care for the diabetic patient who is “at risk” for foot ulceration secondary to conditions such as peripheral neuropathy, peripheral arterial disease, foot deformity, or elevated plantar pressures. These insoles are prescribed to cushion, protect, and redistribute pressures on the plantar aspect of the foot. Examples of “custom” features range from heat moldable flat insoles to devices that include arch fillers, metatarsal pads, and deep heel accommodations.
The practitioner’s personal experience, the preference of insole material, and the procedures utilized by the designated orthotic laboratory can influence the variability among custom made insoles. A case in point is the metatarsal pad, which is perhaps the most commonly made modification for off-loading high pressure regions under the bony prominences of the foot. Successful reduction of plantar pressures under the metatarsal heads using these devices has been reported by some researchers,1-5 while insignificant or even increased pressures at the metatarsal head region have been found by others.6-8 Different approaches to shape, size, material, and placement of the metatarsal pad can lead to varied outcomes.
In the clinical setting, health care providers commonly rely on information obtained from a physical examination to identify the location of elevated plantar pressures. One may presume, for example, that elevated plantar pressures exist when a callus is present, or when a claw toe or dorsiflexion deformity at the metatarsophalangeal joint exists. Yet, the level of agreement among various providers on the location of these pressures is limited and can add to the variability of insole effectiveness.9
A quantitative approach using barefoot plantar pressures to aid in the design of custom made insoles may minimize the variability. Bus et al10 investigated whether knowledge of barefoot plantar pressure could influence the pressure-relieving effect of custom made insoles in patients with diabetic neuropathy and foot deformities. Compared to flat insoles, the custom made insoles were significantly more successful at reducing pressures under the heel and first metatarsal regions, but results varied between subjects. The custom made insoles successfully reduced pressures at the first metatarsal head in one-third of patients (7 of 21), were moderately successful in another third, and failed to reduce pressures in the remaining third. These results led to the conclusion that, even though information on barefoot plantar pressure was provided, the custom made insoles were not consistent in their pressure-relieving effect at an individual level.10
In that study, pressure was measured only after the insoles had been fabricated; insole customization was based on the practitioner’s subjective clinical assessment. The inconsistent results point to the need for a more systematic, objective way of producing consistency within customized prescription footwear for the individual patient with diabetes.

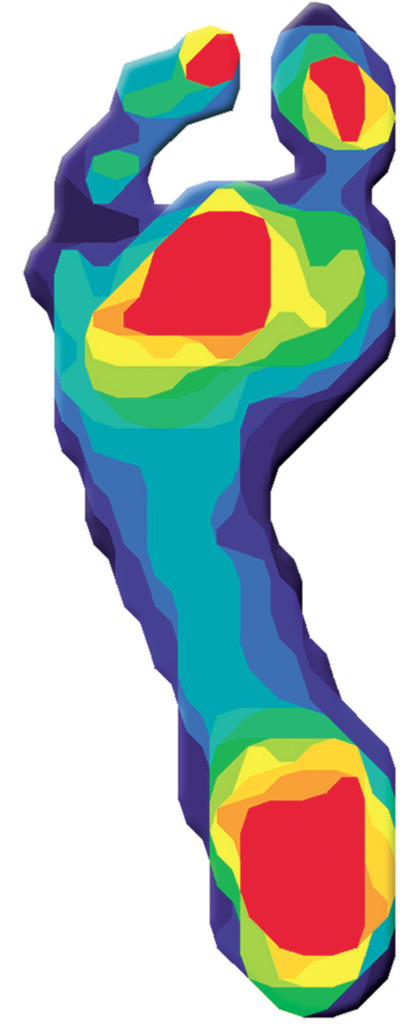
Figure 1
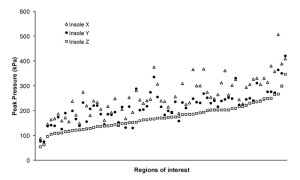
One option is to integrate the barefoot plantar pressure information into a computer algorithm used to design custom made insoles (Figure 1).11 This method of combining the shape of the foot with quantitative values obtained from barefoot pressure measurements to fabricate custom made insoles was compared to conventional custom insoles.12 Three sets of foam impressions were made for each of 20 diabetic patients selected on the basis of having high barefoot plantar pressures (>750 kPa) in the metatarsal head region of each patient’s foot. Two orthotic supply companies fabricated insoles based solely on the shape of the foot, while the third company was also supplied with plantar pressure data, which were integrated into its computer algorithms for fabrication. In 64 of 70 regions of interest at the metatarsal head (sites where barefoot pressure ≥450 kPa at baseline), the shape and pressure insole produced significantly better unloading than either of the two shape-based insoles (Figure 2). Overall, peak in-shoe pressure in the pressure-based insole was reduced by 32% compared to one shape-based insole and by 21% compared to the other. The reduced pressure was attained by transferring the load to the midfoot, which typically can bear load safely and effectively, rather than to other forefoot structures or adjacent metatarsal heads. This study shows the considerable differences in the offloading capabilities of custom made insoles obtained from different orthotic companies and demonstrates the offloading ability of a custom insole created by using a quantitative measure for insole fabrication.12
Clinical trials are needed to look at the short-term and long-term effectiveness of molded insoles customized based on barefoot plantar pressures. Do these quantitative insoles provide increased comfort and improve gait? Is the transference from the metatarsal head pressure to the midfoot tolerable in the short term for patient adherence purposes? Are there musculoskeletal or skin complications over time? Is the redistribution of pressures maintained after three months, six months, and 12 months of daily usage? And, importantly, does better unloading translate into reductions in ulceration or re-ulceration rates?
Although barefoot plantar pressure measurements can influence the fabrication of an orthotic for redistributing peak pressures, people with diabetes are generally advised against walking barefoot. Assuming that patients adhere to this advice and are compliant with wearing their prescribed footwear, the more important measurement is in-shoe plantar pressure. Such data can provide information about the effectiveness of the insoles in offloading the plantar aspect of the foot, which becomes crucial when dealing with a neuropathic patient who is at even higher risk for recurrent foot complications. Ideally, a known peak pressure threshold would be established as a guideline for effective offloading. Mean in-shoe pressure values of 134 kPa to 275 kPa have been reported in the forefoot region of 93 ulcer free patients with diabetes during level walking.13 In another study investigating the in-shoe plantar pressures of 49 diabetic patients with neuropathy and a prior ulcer (currently healed), the authors suggest a conservative goal of approximately 200 kPa that could be used as a guideline for forefoot metatarsal head pressures.14 This value is similar to a clinically derived maximum mean peak pressure threshold of 207 kPa used by Mueller et al.15
Burnfield et al16 used the above mentioned 207 kPa in-shoe pressure threshold to influence modifications of custom inserts for patients with diabetes mellitus at risk for ulceration. In-shoe pressure measurements were obtained from 39 custom inserts (from 21 patients) and revealed that 15 of the inserts were associated with a peak pressure that initially exceeded the 207 kPa threshold. Insert modifications reduced the peak pressure in 12 of those cases, such that eight additional inserts were associated with peak pressures values below the threshold. While this study shows that about 80% of the custom inserts could be fabricated/modified to produce peak pressure values below 207 kPa, follow-up information on these patients will be needed to determine the appropriateness of this threshold for ulcer prevention.
In-shoe plantar pressures can also be utilized to determine if foot deformities can be accommodated with custom made insoles instead of additional bracing. Cavanagh et al17 have noted that patients in the quiescent phase of Charcot neuroarthropathy at the midfoot could be successfully managed with a semi-rigid custom made in-shoe orthosis with a rocker sole athletic shoe versus a more restrictive above-the-ankle bracing device. For these six diabetic patients with a history of Charcot arthropathy, barefoot plantar pressures, as expected, demonstrated large peaks at the midfoot region (mean of 600 ± 395 kPa). In comparison, in-shoe pressures using the semi-rigid orthosis and rocker sole athletic shoe showed that large reductions of pressure at the midfoot region had been achieved. On average, the mean peak value at the midfoot was 173 kPa, which did not differ significantly from the 165 kPa in-shoe pressure measured with the above-the-ankle bracing device. At a six month follow-up, none of the patients reported any ulcers or adverse events. Although in this study pressure measurement was not part of the orthosis fabrication process, the results demonstrate the use of plantar pressure measurements for evaluating the effectiveness of a truly customized orthotic device. With this evaluation tool, practitioners need not rely solely on past experience or guess work to determine which braces or footwear will maintain a plantigrade foot with optimum skin integrity.
Although the foot is exposed to three-dimensional force vectors during weight-bearing activities, analysis is generally limited to the vertical (or normal) component. Plantar shear is also known to be a factor in callus formation and has previously been associated with elevated ulcer incidence.18,19 Technological advances are being made that will facilitate investigation of the relationship between peak pressure and shear stress for barefoot20,21 and in-shoe measurements.22,23 Lott et al22 have noted that, compared to peak plantar pressure alone, peak maximum shear stress and peak pressure gradient can provide additional information for discriminating between groups of patients at risk for ulceration. Progressive studies are still needed to determine if these measurements can be used to predict ulceration risk, and questions remain as to whether various orthotic devices can reduce plantar shear.
Currently, Medicare reimburses the same amount for custom made insoles regardless of the methods used for custom fabrication, which means that the cost of performing plantar pressure measurement is not reflected in reimbursement rates. This leads us to the question whether different products should be reimbursed at different rates. But first we must ask whether certain orthotic management techniques reduce complication rates more effectively than others, and therefore prevent additional medical costs associated with skin breakdown. Do shape-and-pressure based insoles reduce ulceration rates over the shaped-based orthotic? Does measuring plantar pressure affect the quality of the product or the prescription written? Should Medicare reimburse for the measurement of plantar pressures? If so, can specialty clinics widely implement this measuring system into everyday practice to create a custom, efficacious device that is proven with in-shoe pressure measurements at time of dispensing? These questions may be sufficiently answered only when solid evidence exists to support the practice of measuring plantar pressures, since such measurement is critical to the making of a better product to preserve the foot at risk for skin breakdown and eventual amputation.
In summary, elevated plantar pressures have been implicated as a risk factor for ulceration in the neuropathic diabetic foot. Plantar pressure measurement, while not uncommon, is not a uniformly obtained measurement. However, barefoot plantar pressure measurements can be used to determine biomechanical pathology and to assist in the fabrication of “custom” insoles designed to offload the foot. In-shoe pressure measurement offers an objective, mechanical dimension that can allow comparison of foot insoles and shoe gear in terms of their efficacy for offloading at-risk areas of the foot.
The goal of any prescription footwear or orthotic device should be to produce a product that is predictable and efficacious. Considerable differences between devices in terms of offloading capabilities have been found using pressure measurement. The challenge now is to prove whether that ability to differentiate between orthotic management strategies will reduce complication rates. Once evidence supports a reduction in complication rates in the superior offloading device, plantar pressure measurement has the potential to become the standard of care.
Georgeanne Botek, DPM, FACFAS, is a podiatrist in the Orthopaedic Rheumatologic Institute and medical director of the Diabetic Foot Clinic at the Cleveland Clinic in Cleveland, OH. Tammy M. Owings, Deng is a research associate in the department of biomedical engineering at the Cleveland Clinic.
Disclosure: The authors have no conflicts of interest to declare.
References
1. Albert S, Rinoie C. Effect of custom orthotics on plantar pressure distribution in the pronated diabetic foot. J Foot Ankle Surg 1994;33(6):598-604.
2. Brown M, Rudicel S, Esquenazi A. Measurement of dynamic pressures at the shoe-foot interface during normal walking with various foot orthoses using the FSCAN system. Foot Ankle Int 1996;17(3):152-156.
3. Lord M, Hosein R. Pressure redistribution by molded inserts in diabetic footwear: a pilot study. J Rehabil Res Dev 1994;31(3):214-221.
4. Novick A, Stone J, Birke JA, et al. Reduction of plantar pressure with the rigid relief orthosis. J Am Podiatr Med Assoc 1993;83(3):115-122.
5. Postema K, Burm PE, van der Zande ME, van Limbeek J. Primary metatarsalgia: the influence of a custom moulded insole and a rockerbar on plantar pressure. Prosthet Orthot Int 1998;22(1):35-44.
6. Ashry HR, Lavery LA, Murdoch DP, et al. Effectiveness of diabetic insoles to reduce foot pressures. J Foot Ankle Surg 1997;36(4):268-271.
7. Uccioli L, Toffolo M, Volpe A, et al. Efficacy of different shoes and insoles in reducing plantar pressures in diabetic neuropathic patients (Abstract). Diabetologia 1997;40(Suppl 1):A-489.
8. Hewitt FG Jr., Ulbrecht JS, Becker MB, et al. The effect of molded insoles on in-shoe plantar pressures in rockered footwear (Abstract). Diabetes 1993;42(Suppl 1):194A.
9. Guldemond NA, Leffers P, Nieman FH, et al. Testing the proficiency to distinguish locations with elevated plantar pressure within and between professional groups of foot therapists. BMC Musculoskelet Disord 2006;7:93.
10. Bus SA, Ulbrecht JS, Cavanagh PR. Pressure relief and load redistribution by custom-made insoles in diabetic patients with neuropathy and foot deformity. Clin Biomech 2004;19(6):629-638.
11. Cavanagh PR, Owings TM. Nonsurgical strategies for healing and preventing recurrence of diabetic foot ulcers. Foot Ankle Clin 2006;11(4):735-743.
12. Owings TM, Woerner JL, Frampton JD, et al. Custom therapeutic insoles based on both foot shape and plantar pressure measurement provide enhanced pressure relief. Diabetes Care 2008;31(5):839-844.
13. Guldemond NA, Leffers P, Sanders AP, et al. Daily-life activities and in-shoe forefoot plantar pressure in patients with diabetes. Diabetes Res Clin Pract 2007;77(2):203-209.
14. Owings TM, Apelqvist J, Stenström A, et al. Plantar pressures in diabetic foot ulcer patients who have remained healed. Diabet Med (in press).
15. Mueller MJ, Smith KE, Commean PK, et al. Use of computed tomography and plantar pressure measurement for management of neuropathic ulcers in patients with diabetes. Phys Ther 1999;79(3):296-307.
16. Burnfield JM, Few CD, Whitney JA, et al. Influence of PEDAR-based insert modifications on walking plantar pressures in persons with diabetes mellitus. Presented at Ninth Annual Gait and Clinical Movement Analysis Society Meeting, Lexington, KY, April 2004.
17. Cavanagh PR, Botek G, Owings TM. Biomechanical factors in Charcot’s neuroarthropathy. In: Frykberg RG, ed. The Charcot Foot and Ankle. Towson, MD: Data Trace Publishing Company (in press).
18. Murray HJ, Young MJ, Hollis S, Boulton AJ. The association between callus formation, high pressures and neuropathy in diabetic foot ulceration. Diabet Med 1996;13(11):979-982.
19. Mackenzie IC. The effects of frictional stimulation on mouse ear epidermis. I. Cell proliferation. J Invest Dermatol 1974;62(2):80-85.
20. Mueller MJ, Zou D, Bohnert KL, et al. Plantar stresses on the neuropathic foot during barefoot walking. Phys Ther 2008;88(11):1375-1384.
21. Yavuz M, Erdemir A, Botek G, et al. Peak plantar pressure and shear locations: relevance to diabetic patients. Diabetes Care 2007;30(10):2643-2645.
22. Lott DJ, Zou D, Mueller MJ. Pressure gradient and subsurface shear stress on the neuropathic forefoot. Clin Biomech 2008;23(3):342-348.
23. Zou D, Mueller MJ, Lott DJ. Effect of peak pressure and pressure gradient on subsurface shear stresses in the neuropathic foot. J Biomech 2007;40(4):883-890.
FIGURE LEGENDS
Figure 1: Example of an insole based on a design algorithm that identified a pressure contour, along which a metatarsal bar was created. An area of the insole underneath a metatarsal head was removed in regions of excessive local pressure (>1000 kPa) (Reprinted from Owings et al., Diabetes Care 2008;31(5):839-844; Copyright © 2008 by the American Diabetes Association).
Figure 2: Individual peak pressures (ranked by peak pressure of Insole Z, which was shape and pressure based) at the 70 regions of interest for insoles in flexible shoes. Open triangles, Insole X; filled circles, Insole Y; open squares, Insole Z (Reprinted from Owings et al., Diabetes Care 2008;31(5):839-844; Copyright © 2008 by the American Diabetes Association).









I wish to comment on the article by G. Botek, DPM & TM Ownings, DEng, “Diabetes: Pressure Data Guide offloading efforts”, LER, Sept 2009.
I am in agreement with the authors’ statement, “Once evidence supports a reduction in complication rates in the superior offloading device, plantar pressure measurement has the potential to become the standard of care.” I would not specify “superior offloading device” as it is only necessary to define the desired result and document that result – no matter what shoe or shoe-orthotic combination is used.. Shoes as well as soft and even hard orthoics can be “off loading” but are not specific and are certainly not predictable in a practical sense. These products can also INCREASE in-shoe gaited peak plantar foot pressures either at the original peak pressure site or “transfer” peak pressures to another area on the plantar foot – not a desirable result.
Plantar foot pressure, force-time integrals and barefoot pressure determinations are interesting but do not specifically address the main driver of foot ulceration in the patient with diabetes and “high risk” insensate feet – in-shoe peak shear pressures. (authors’ ref. 22 and 23).
In a 2 year unpublished pilot study in California on 10 high risk diabetic patients who had recently healed their insensate plantar foot ulcer (Risk Category III), not a single recurrent foot ulcer developed during the study period. In this study, specific peak pressure redistributing orthotics were fabricated based on in-shoe collection of peak pressures throughout the plantar foot.during gait. Sequential post fit gaited in-shoe peak plantar foot pressure determinations were significantly and consistently reduced over the study period (vertical and shear). This is in contrast with the current foot ulcer recurrence rate of 22% in the best controlled protocols and 76% otherwise.
There is no question, that CMS policy should be changed to stipulate reimbursement for in-shoe gait force analysis to document (confirm) and monitor reduction of peak plantar foot pressures before and following TSB insert fitting. I would go even further to state that this should be a condition for reimbursement under the Medicare Part B Therapeutic Shoe Bill program. Unfortunately, current CMS policy re TSB is analogous to a physician’s management of hypertension without obtaining blood pressure determinations or caring for the patient with diabetes without blood glucose determinations. In all fairness to CMS, at the time of the TSB funding decision (May 1993) reliable technology and studies were not available to give guidance to CMS’s current policy.
Background. During March, 2010, I plan to initiate a Level i, RCT study. This study has received an IRB approval and is designed to confirm the pilot study results. My goal is to get multiple centers involved in this project. This will be done in conjunction with opening my initial AcuStep Foot Center, LLC in Springfield, MO. in November. As a semi-retired orthopedic surgeon, my goal is to address preventive care of the LE in patients with a number of maladies including diabetes, obesity, pre-diabetes, neuropathy and peripheral arterial disease using the latest technologies. Further my “standard of care” within the Medicare TSB program is to initially prescribe only 1 pair of A-5513 pressure redistributing custom inserts and do a follow-up in-shoe GFA within 1-2 weeks, then at 4 months, 8 months and annually depending upon the risk level.. New orthotics would only be provided based on GFA confirmation.
The only way to see improvement in care of the diabetic foot is to do the confirmatory preventive research, publish and address these new realities with CMS. The authors are to be commended for their efforts with this publication – much more is needed.
Thanks.
Jeryl G. Fullen, MD
888 No. 22 Street
Ozark, MO 67721
jg******@*****ep.com
785-826-0184
.