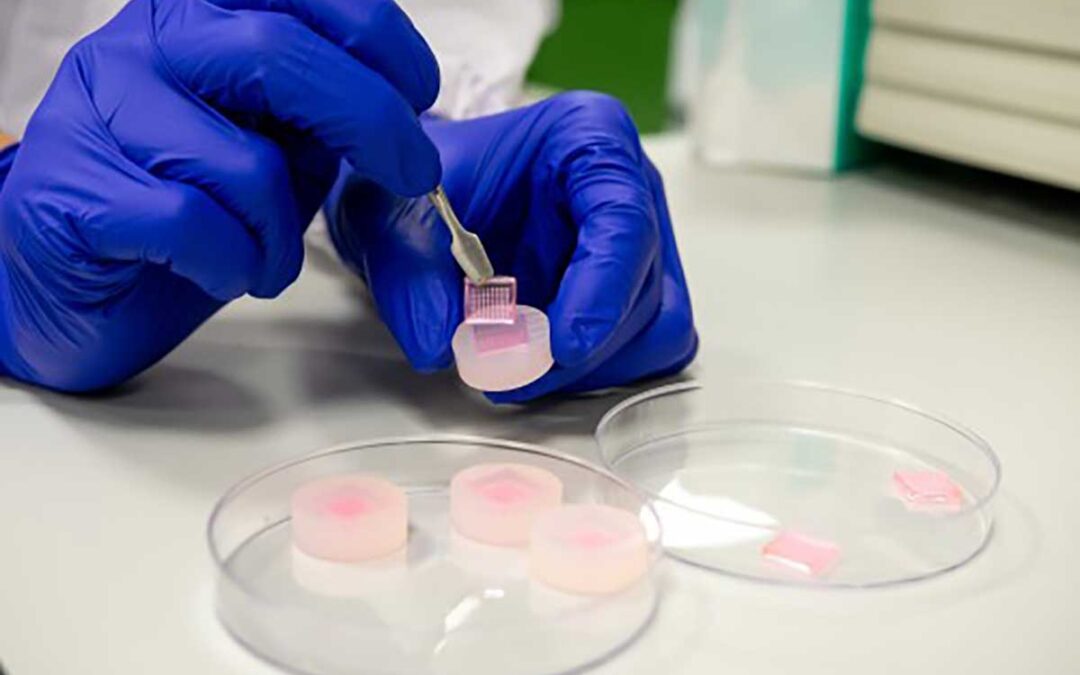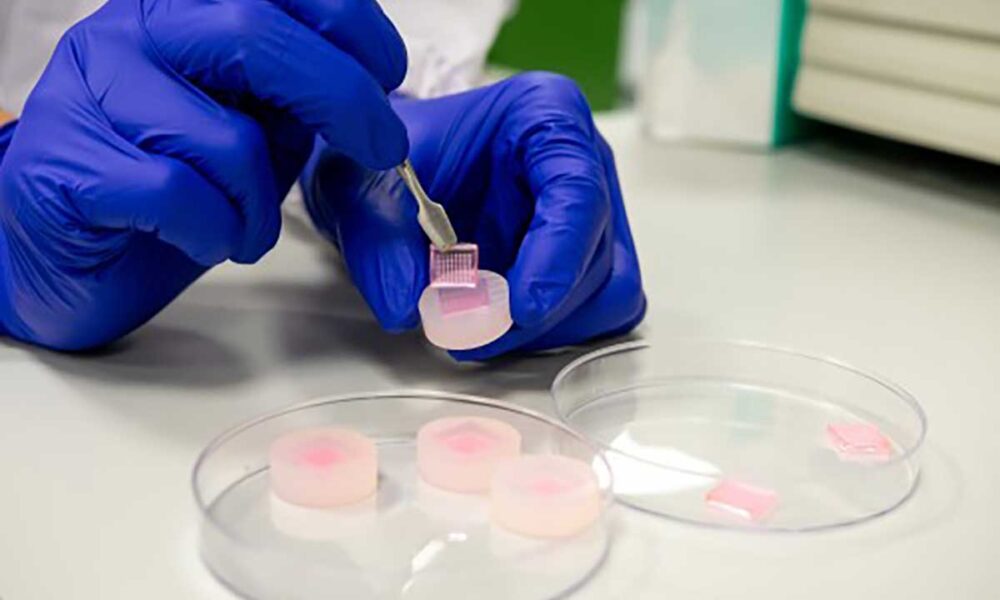
The microneedles were fabricated from poly lactic-co-glycolic acid (PLGA), an FDA-approved class of biodegradable polymers that are biocompatible and can be used in the sustained delivery of drugs and other molecules. The sponge-like PLGA microneedles, made using the moulds shown above, can help to alleviate inflammatory skin disorders.
Researchers from the National University of Singapore (NUS) have developed 2 microneedle technologies that have shown efficacy in accelerating diabetic wound healing in preclinical models by preserving the functions of proteins called growth factors, and removing undesirable inflammatory compounds.
In the first approach, instead of delivering the growth factors directly, the team first increased the production of growth factors within the wound by developing sucralfate microneedles (SUC-MN) to deliver an important immunomodulatory protein, interleukin-4 (IL-4), to stimulate the production of growth factors in diabetic tissues. IL-4 helps to regulate the immune response and promote tissue regeneration, while sucralfate protects growth factors from degradation. The microneedles dissolve in the wound, delivering IL-4 and sucralfate directly to the wound. This localized delivery system minimizes systemic side effects and avoids secondary damage to delicate, newly formed tissues caused by traditional adhesive dressing that is currently used clinically. The researchers found that SUC-MN significantly accelerated wound healing twice as fast when compared to traditional treatments.
For the second approach, the team explored the use of microneedles to extract undesirable pro-inflammatory proteins and immune cells. To do so, the team needed to find a suitable coating material—they screened and then used heparin-coated porous microneedles (HPMN)—that could act as a sponge to soak up pro-inflammatory compounds, known as chemokines, which are ‘messenger’ molecules that recruit and trap pro-inflammatory immune cells called monocytes in wound tissues. Based on previous studies, heparin has been found to bind readily to chemokines. The team demonstrated that HPMN could effectively deplete chemokines and monocytes from the wound site, leading to a 50% reduction in tissue inflammation as well as a 90% reduction in wound size by the fourteenth day of treatment.
The development of SUC-MN and HPMN represents a significant step forward in the field of wound healing and skin disease management. The team intends to conduct further studies to explore the potential of this technology and bring it to market. For extractive microneedles in particular, the team will fabricate microneedles with more controllable pore sizes using advanced technologies, such as 3D printing, and integrate antibacterial properties into the microneedles as clinical non-healing wounds often accompany infections. They are also designing flexible microneedle patches to ensure that they fit well to various tissue shapes.