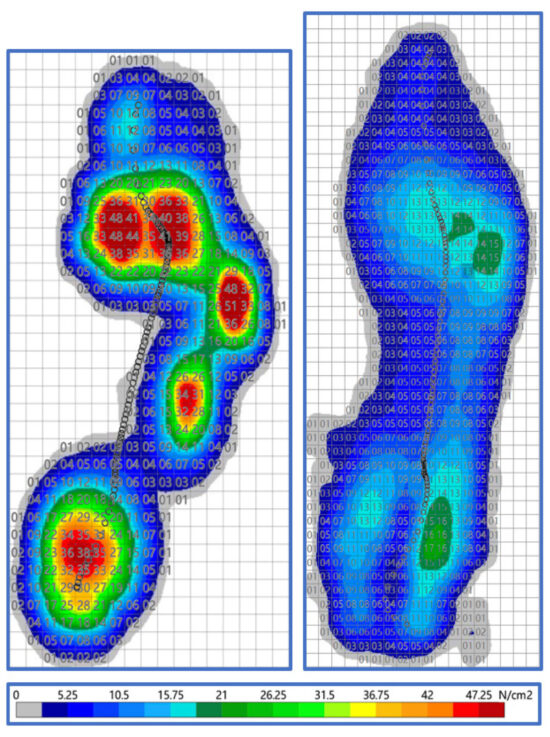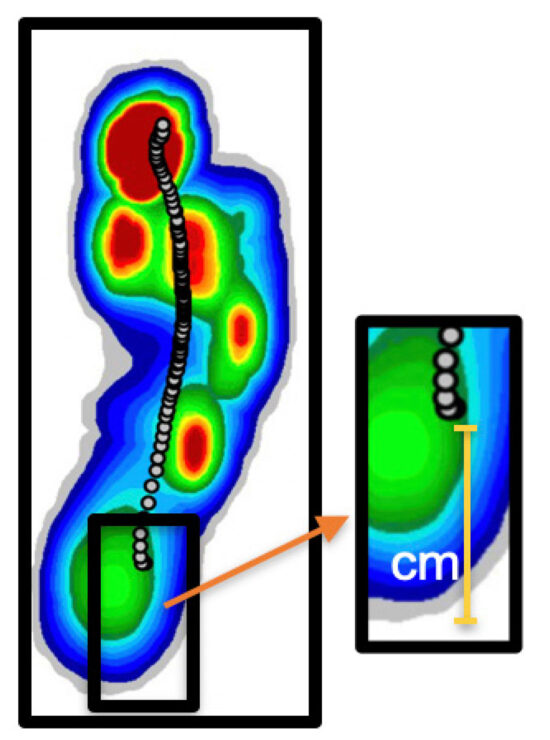
Figure 2: Johns Hopkins takes a multidisciplinary team approach using Computerized Gait Analysis (CAGA) to revolutionize care and recommendations.
By Jay Segel, DPM; Sally Crawford, MS; Mark Hopkins, PT, CPO, MBA; Brian Murray, PT; Tracy Winchester; Alexander Lakner, DPM; DeEtta McKnight, RN; Caitlin Hicks, MD, MPH-V; Christopher Abularrage, MD; Sanuja Bose, MD, MPH; Alana Keegan, MD; and Ronald Sherman, DPM, MBA
Diabetes mellitus has exploded in its prevalence worldwide, affecting millions with grave consequences, crippling both the patient and the medical system alike. The diabetic foot is a significant contributor to the global burden of disability.
“Diabetes is an epidemic.”
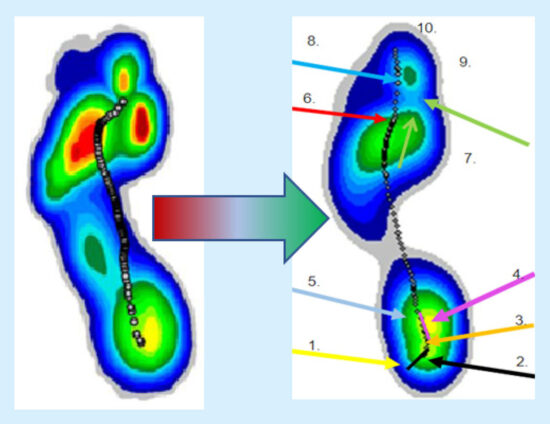
Three years ago, at a round table discussion, these words inspired an evocative and profound study centered at the Johns Hopkins Hospital. This discussion would be the commencement of an Institutional Review Board approved study to determine the efficacy of diabetic re-ulceration prevention through Computer Aided Gait Analysis (CAGA), custom prescription orthoses, and defined footwear.
The majority of diabetic ulcers develop as a result of everyday stresses, or repetitive micro-trauma at the foot’s plantar surface. These can occur during routine ambulation as patients go about their normal routine, or occurrence can be heightened with specialized activities.
The underlying contributory factors for this are biomechanical and structural deformities of the foot that directly impact areas of identifiable risks on the diabetic plantar foot. This is secondary to multifactorial diabetic co-morbidities including both sensory and motor neuropathy. Hence, as a sequela of the neuropathy and deformities, this patient population will demonstrate an abnormal walking pattern, including a less stable gait and truncated center of pressure, as well as the development of multiple abnormal temporospatial parameters in the multiple phases of gait. These, in turn, create altered gait kinematics and increased peak plantar pressures which overall impair the ability to heal (Figure 1). Herein, a three-dimensional foot of limited mass is colliding with a two-dimensional, relatively unyielding surface of relatively unlimited mass (the ground or support surface). This is a battle the foot encounters 4 million times per year, as this is the average number of steps an individual takes.
These repetitive deforming, shock-producing, and energy-dissipating events will opportunistically wreak havoc on areas at risk on the plantar surface of the foot. Calluses/ulcerations form with the repetitive trauma, and peripheral neuropathy desensitizes the patient to these problems. Furthermore, the presence of arterial disease, common in the diabetic population, makes it difficult to heal these areas. Unfortunately, it is a battle the foot faces repetitively, making it a battle it cannot win. Because of the additional musculoskeletal, neurological, integumentary, and circulatory complications, it is particularly devastating to the compromised diabetic foot which will eventually lose the war to deformity, tissue quality as well as circulatory, neurological complications, and the driving biomechanical forces of basic collision dynamics.
At Johns Hopkins, a multidisciplinary team approach was leveraged to provide the best care possible for these patients. Each contributor brings their own unique viewpoint and expertise of this devastating disease, including predominant specialties: Surgical Podiatry, Vascular Surgery, and Endocrinology. A further contribution is seen by physician assistants, certified prosthetists, physical therapists, occupational therapists, wound care nurses, biomechanist, and a data management team. Consultations from other specialists, including plastic surgery, orthopedic foot and ankle, and infectious disease specialists, are utilized as needed. Efforts by the team in the past were able to repetitively achieve high limb preservation rates for complex diabetic wounds, but the recurrence rates seen were similar to the nationwide rate: 40% ulcer recurrence within 12 months, 60% in 3 years, and 65% recurrence in 5 years. There had to be a better mousetrap, perhaps one that wasn’t in existence or utilized to its full potential. It is into this reality that CAGA was introduced into the treatment paradigm (Figure 2).
Modern-day technology has evolved; the applications and possibilities of today’s advancements are endless and constantly being developed. The progressing technology, CAGA, allows for the objective collection of data for this at-risk patient population. With CAGA technology, the understanding of diabetic gait and the identification of occult high plantar-foot pressures is possible, even prior to any apparent clinical signs or symptoms identified by patient and physician. These metrics are then able to be communicated to specialists, who interpret and apply the data to try to prevent future ulceration through footwear and gait modification.
The prevalence and consequence of a diabetic foot ulcer (DFU) remains a devastating life altering event which continues to be a major contributor to our medical and public health challenges. These chronic wounds encompass complexities which have a long-term impact on patients’ morbidity, mortality, and quality of life. Globally, more than 425 million people are living with diabetes, and its prevalence is expected to increase at least 50% by 2045.1,2 Diabetes is, worldwide, the leading cause of blindness, non-traumatic lower extremity amputations, peripheral neuropathy, and end-stage kidney disease, which accounts for 43% of all diabetes-related deaths in people younger than 70 years of age.3-7 The American Diabetes Association estimates 10.5% of the US population is diagnosed with diabetes (32 million people) and is expected to be 36 million people affected by 2045.8 Diabetes is the 7th leading cause of death in the US as of 2017. More than 200,000 non-traumatic amputations linked to diabetes occur yearly. In the United States, 230 amputations per day occur in patients with diabetes. Worldwide it is estimated that every 30 seconds a leg is amputated from the manifestations of diabetes.9-10 Eighty-five percent of diabetes-associated amputations are preceded by the development of foot ulcers. It is estimated that 19–34% of patients with diabetes are likely to be affected with a diabetic foot ulcer in their lifetime, and the International Diabetes Federation reports that 9.1–26.1 million people will develop DFUs annually.11 Adding to the burden, even when a diabetic ulcer is finally healed, the risk of recurrence is rather commonplace with reported rates between 30% and 40% within the first 12 months.12-13 Lastly, the medical expenditure for the management of diabetic foot disease in the United States ranges from US $9.1 to $13.2 billion annually.14
Initially, the diabetic ulcer recurrence rate of the Johns Hopkins multidisciplinary team was similar to that of the nationwide data, as stated above. Our understanding of the literature found that preliminary protocols utilizing CAGA were creditable at improving outcomes. Therefore, CAGA had the potential to help us direct individuals into remission, thereby avoiding the development of recurrent foot ulceration and its associated morbidity, hospital admission, and subsequent amputation.
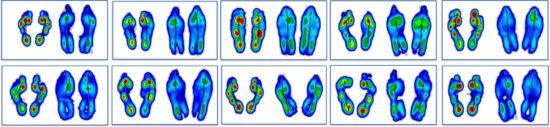
Figure 4: Sample of typical integrated round data comparisons characterizing baseline, test, re-test outcome tracking to monitor solution and care progress.
Methodology
Through the philanthropy of one patient, who personally realized our limb preservation success, initial resources were obtained to further comprehend the etiology and development of the diabetic foot ulceration. It was this initial impetus to construct the CAGA Diabetic Ulcer Reduction Study (CAGA DURS study). After approval by the Johns Hopkins Institutional Review Board (IRB), a nationwide, multidisciplinary committee was assembled and anchored at Johns Hopkins Hospital, utilizing the Noraxon MR3 software paired with a companion outfitted treadmill/video capture system, to perform computer-aided gait analysis.
The team continues work today and is comprised of notable and seasoned experts who are leaders in their fields. These specialists hold credentials and positions in podiatric and vascular surgery, podiatric biomechanics/orthotic prescription writing, biomedical engineering, pedorthics, physical therapy, orthotic lab management, medical writing, and statistical analysis.
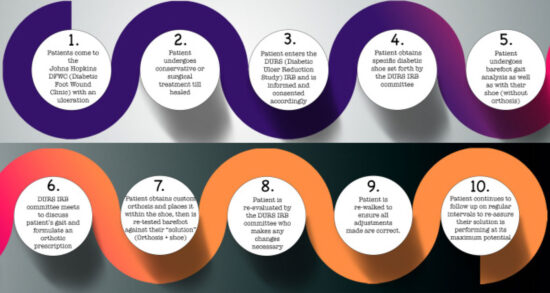
Patients are seen by principal providers of the service (vascular surgery, surgical podiatry, and endocrinology) at each inpatient admission and a weekly outpatient clinic visit. They are examined in an integrated fashion within a single location and followed up at frequent intervals for ongoing wound assessment and intervention (Figure 3). The service is designed to optimize wound healing in patients with DFUs. Patients undergo both conservative and surgical modalities to heal the ulcerations according to standard practice. Once healed, an explanation of the CAGA DURS study, its goals, parameters, and limitations/risks are presented to the patient. Each patient is informed and consented for participation within the study.
Inclusion criteria include a diagnosis of diabetes and a foot ulceration where the wound had remained healed for at least 4 weeks. Excluded were those candidates with Charcot neuroarthropathy and compliance issues.
Consented patients are simultaneously referred to the study’s physical therapist (PT) and orthotist/prosthetist (Certified Prosthetist/Orthotist, CPO) for independent initial evaluations. Johns Hopkins has multiple academic locations and laboratories which were all made available to accommodate this patient population’s needs.
The initial evaluation is done by PT and includes measurements on limb length, muscle tone, flexibility, and other parameters affecting gait. Before the PT facilitates the patient walking on the specialized treadmill for their initial formal computerized gait analysis, a baseline scan of the patient’s bilateral feet is performed by the CPO with iPad scanning software.
CAGA testing is then performed. All testing for this study was completed on a medical grade treadmill where spatial, temporal, center of pressure, and variability parameters are obtained. This is done using the research grade Zebris FDM-T Treadmill (Zebris1 Medical GmbH, Germany) fitted with an under-belt platform consisting of 10,240 force sensors, each approximately 1 cm × 1 cm. Patients are initially walked at a comfortable gait speed (traditionally 0.7mph-1.2mph), while barefoot, to create a baseline gait assessment. During walking (dynamic) and standing (static) modalities, as well as reactive-normal forces are captured in the X, Y, Z directions by the sensors at a sampling rate of 120 Hz. Due to the high density of the sensors, the foot is mapped at a high resolution to facilitate even subtle changes in force distribution. Timing can also be monitored and standardized in a fashion allowing adequate repeat testing. Dedicated Noraxon MyoResearch Software 3.18 running dynamic hardware configurations in 10 zone Myopressure expert mode is integrated with the force signals and provides 2D/3D graphic representation. Additionally, synchronized to the pressure outfitted treadmill are 2 slow-motion high-definition cameras (Logitech C 920) to capture video for analysis.
Once the patient is tested by PT, the findings are reviewed by the CAGA DURS committee which meets weekly. The committee assesses the patient’s past medical and surgical history, as well as the PT physical exam findings (limb length, muscle tone, etc.). The committee scrutinizes the analytical components of the CAGA data, focusing on quantifying characteristics of the gait pattern and formulates a prescription for a custom orthotic and/or prosthetic based on the patient’s areas of risk and their unique biomechanics (Figure 4). The majority of DFUs arise from everyday wear-and-tear, the result of repetitive micro-trauma. In analyzing the pressure print generated by the CAGA, one can identify the areas experiencing a high N/cm2 (usually in excess of 40 N/cm2) which denote an area at heightened risk for future ulceration. This may be the area of former ulceration or an area of current stresses which may become a future ulceration without proper intervention. Furthermore, morphological assessment of the center of pressure (COP) line, indicative of the foot moving throughout stance phase of gait cycle, can denote progressive and/or regressive pronatory and supinatory events of the foot/ankle complex as well as initial contact point, propulsion throughout the gait cycle, and many other parameters. By observing the manner in which the patient walks and identifying overburdened areas based on pressure and timing abnormalities, the podiatric biomechanical gait specialist can devise a device to effectively offload areas of risk for ulceration and improve problematic gait characteristics. The initial committee session examining the CAGA findings ends with a coordination of care plan which starts by communicating our custom orthotic to the director of manufacturing of said device.
Orthotic prescription writing is both an art and a science, and for this research study, a choice is made between 3 shell types and a multitude of additions and modifications for further customization. The type of orthotic prescribed will determine the materials used. There are 3 types of devices employed:
Type 1 is a soft tri-laminate foam foot bed that is used mainly for cushioning and offloading of the insensate foot.
Type 2 is a functional orthotic device with a thermoplastic shell and Poron (a polyurethane) arch reinforcement. The rigidity of the shell is determined by patient weight or the amount of control that is needed. These devices are covered full length with a layer of Poron padding and a high rebound ethylene vinyl acetate (EVA) as a top cover.
Type 3 is an Ankle Foot Orthosis (AFO) brace.
The most prescribed device is the Type 2. The casts are digitally corrected and milled on a Computerized Numerical Control (CNC) router. Materials are pressed into to the cast forms and then postings are added according to the prescription. The final step is finishing the devices, adding pads or accommodations that have been specified by the providers. They are quality checked and sent back to the CPO to dispense.
Patients are then instructed to obtain a designated diabetic shoe from a specifically recognized diabetic shoe manufacturer. The shoes are fitted and provided by 2 local medical pharmacies. Once the prescription is generated and the devices are authorized by the patient/funding source, the order is sent to the orthotic manufacturer for fabrication. The fabricated devices are then sent to the CPO who checks them for quality against the prescription and if accurate, then schedules the patient for dispensing and fitting of the device. At this appointment, the patient is expected to have acquired their specified diabetic shoes as it may be coordinated with re-testing of the patient’s gait with PT if that is possible and convenient for all parties.
The dispensing and fitting appointment involves a brief screening for any new issues, an assessment of the shoes for fit/size, fitting of the device to the shoes, and an education on use/care. This is followed by a full second gait analysis or Re-Test of the patient’s gait on the specialized treadmill with the custom orthotic plus shoe which is herein described as the “Solution.” The patient is re-tested barefoot against their solution (orthotic+shoe), and the results are reviewed by the CAGA DURS committee.
The CPO participates in the re-testing review meetings process and initiates any required follow-up visits for adjustments, repairs, re-fabrication, or replacement based on the outcome of the review process. These subsequent visits, at regular intervals with the CPO, are based on traditional wear patterns observed as well as any needs identified during the subsequent CAGA review meetings (Figure 5).
This process continues for the duration of the study at 1 month, 3 months, 6 months, 1-year, and 2 years per the rigors of the study guidelines. The plan is to maintain this process and support the patient ongoing—even beyond the study period, through regular and typical clinical activities and incorporating lessons learned from the larger study experience.
Biomechanical Analysis
Our primary goal, and consistent with our larger goal, is returning our patient population to their activities of daily living (ADLs). The decision-making around shell material is made considering the weight, foot length, and structural integrity of the wearer. For instance, there is an adage to be considered: the more flexible the foot, the more control it needs. Where length is concerned, the longer the foot, the longer the device, which makes the shell less rigid. When it comes to areas on the plantar surface deemed “at risk” for ulceration, the best techniques for offloading those regions are employed in accordance with the evidence base. Examples would include: arch height alteration, wells/cut-outs under the areas of high pressure, and/or domes underneath the adjacent metatarsal heads to reduce and divert forces away from the over-pressured forefoot regions.
Another well-utilized addition is that of a metatarsal pad to control forefoot frontal plane motion, also known as “the forefoot pronatory event.” This is particularly helpful for DFUs occurring under the first metatarsal head of a forefoot with a moderate- to high-varus component and calluses at the first interphalangeal joint. Indicators of the biomechanical deformity can be seen in the CAGA, with identification of regions of the foot and noting the segmental loading patterns and times of these various regions. CAGA also identifies pressure changes across each of the metatarsal bones as well as specific regions of the foot.
As an example, if the COP follows a “hockey stick” shaped morphology with a radical turn in the COP line in the vicinity of the metatarsal head region, a metatarsal pad is usually a good inclusion to most orthotic prescriptions (Figure 6). Each CAGA screen, with its graphs and temporospatial parameters, contribute to the discipline of writing a custom prescription orthotic and the above examples were but a few solutions employed in an ulcer prevention functional diabetic orthotic.
Preliminary Study Data
Through preliminary data assessment of those who have completed the study thus far, CAGA was instrumental in identifying peak plantar pressures representing areas at risk for diabetic foot ulceration, and analysis of those modifications implemented.
Analysis of the observed data was performed in the following manner:
Utilizing the Noroxon MyoResearch technology, the area in which a foot contacts the weight-bearing surface was mapped out indicating the maximum pressure values obtained throughout the gait cycle. These pressures, measured in N/cm2, were identified in a 1cm2 grid overlying each foot’s plantar pressure print.
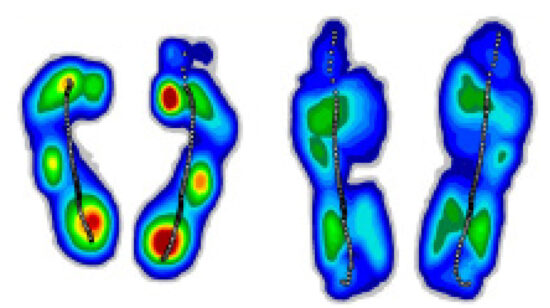
To obtain a baseline data point, the highest single pressure measurement value on each foot’s average contact, while barefoot, was then recorded (Figure 7). The corresponding osseous structure was also recorded, identifying these areas of highest risk: Submetatarsal 1: 33%; calcaneus: 24%; sub-metatarsal 3,4: 20%; cuboid 13%. The patient was then placed within their Solution (custom orthotic and specific diabetic shoe), and re-examined to identify the effective reduction in plantar foot pressures at the same identified zones. To decrease the chance incident of a single 1cm2 area measuring an abnormally high pressure, the identified 1cm2 highest single maximum value pressure reading was averaged along with the surrounding 8cm2 zones for a 9cm2 average.
A direct comparison and percent reduction between the barefoot and Solution values were then calculated and analyzed for both 1cm2 and 9cm2 metrics on the bilateral foot.
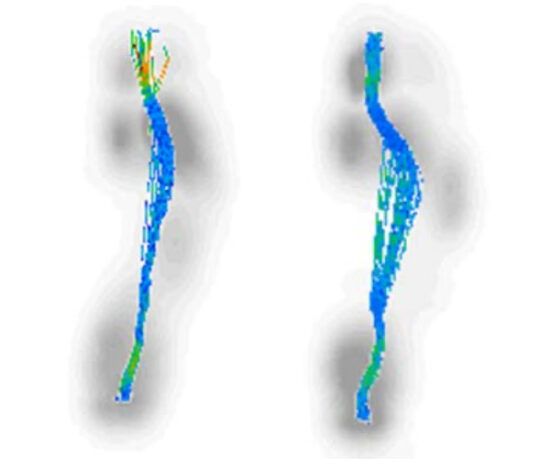
 Furthermore, the center line of pressure was identified as a useful tool for analyzing the subject’s gait. This identifies the initial contact point of the foot within the stance phase of the gait cycle as well as various pronatory, supinatory, transitional, and propulsion changes throughout the gait cycle. The initial contact point of the foot was compared between the initial barefoot gait and initial contact point once solution was employed (Figure 8).
Furthermore, the center line of pressure was identified as a useful tool for analyzing the subject’s gait. This identifies the initial contact point of the foot within the stance phase of the gait cycle as well as various pronatory, supinatory, transitional, and propulsion changes throughout the gait cycle. The initial contact point of the foot was compared between the initial barefoot gait and initial contact point once solution was employed (Figure 8).
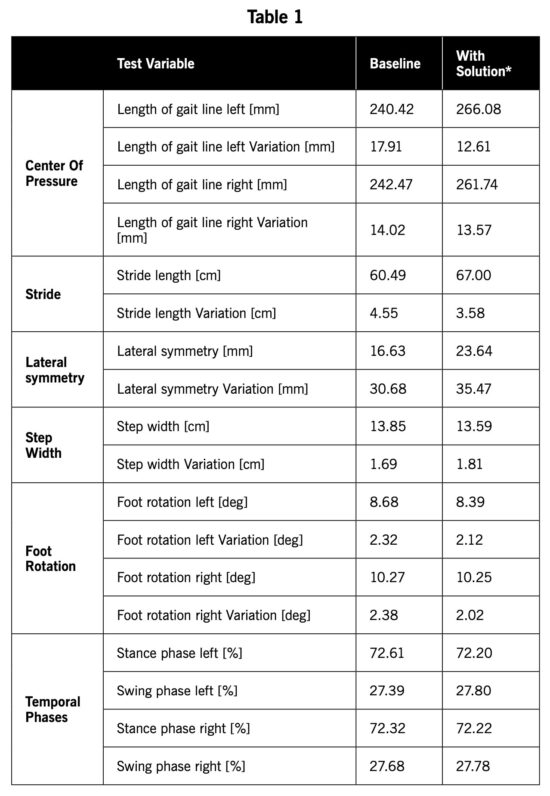
This yielded an average 66.18% decrease in pressure (Left: 47.79nm2 vs. 12.36nm2 and Right: 47.79nm2 vs. 13.46nm2, P<0.001) [Table 1]. It was also noted that the initial contact point of gait (Figure 8) was decreased (moved more proximally/posteriorly by an average of a 28.69% (Left: 2.67cm and Right: 2.42cm, p<0.001) [Table 1], elongating the gait line, and subsequently better dispersing the forces acting upon the foot. The most consequential parameters which are contributory to this success are being statistically analyzed and include: length and deviation of gait line, stride length, lateral symmetry, step width, foot abduction, and stance vs. swing ratio. The preliminary data utilizing CAGA shows that the length of gait line was elongated on average by 22.96mm (Left:26.66mm, Right: 19.26mm) and stride length increased by 6.5cm. Lateral symmetry (COP intersecting/ a balance measurement/center of mass) increased by 7.01cm, step width (heel to heel width) decreased by 0.25cm, and rotation/abduction of the foot decreased by 0.33 degrees (Left: 0.29 degrees, Right:0.37 degrees). Lastly, swing vs/stance phase was modified by 0.255% (Left: 0.41% more time spent in stance, Right: 0.1% more time spent in stance phase). These pressure and gait modifications are encouraging as they demonstrate overall changes in a positive direction. All parameters and data will be fully analyzed and published once full cohort (n=90) is completed.
Conclusion
Utilizing a disciplined, multidisciplinary approach that begins with a healed diabetic foot, we are able to use CAGA to analyze the temporospatial gait parameters of a patient with diabetes. Computer-assisted gait analysis allows practitioners to perform a detailed comparison between a patient’s gait while barefoot and with their unique solution (orthotic and specific shoe) to obtain the best quantitative data. This objective data allows users to analyze both the biomechanical deformity and structural deformity (eg, limb length, metadductus, cavus foot type, diminished fat pad), as well as the effectiveness of footwear modification. As a highly accurate, objective diagnostic tool, CAGA yields the ability to capture temporospatial parameters, both statically and dynamically, pinpointing plantar regions that are at risk. These areas can be identified even before they show signs of pre-ulceration. It allows a detailed evaluation of a patient’s gait, identifying areas of concern, success in the management of problematic areas, and areas that need further attention. Our preliminary data has validated our ability to provide increasing gait stability and off-loading, leading to a potential decrease in risk of ulceration. It is hopeful this is the impetus for dramatic change that yields a new and brighter future for this highly “at-risk” patient population.
Jay Segel, DPM, is a Podiatrist, Biomechanist, Orthotic and Gait Analysis Specialist, in Martha’s Vineyard, MA.
Sally Crawford, MS, is a Biomedical Engineer and data manager in Omaha, NE.
Mark Hopkins, PT, CPO, MBA, is a Certified Pedorthist at The Johns Hopkins Hospital in Baltimore, MD.
Brian Murray, PT, is a Physical Therapist at The Johns Hopkins Hospital in Baltimore, MD.
Tracy Winchester is Director of Orthotics Fabrication at Forward Motion Orthotics in St. George, UT.
Alexander Lakner, DPM, is Fellow in Limb Preservation and Wound Care at The Johns Hopkins Hospital, in Baltimore, MD.
DeEtta McKnight, RN, is a Wound Care Nurse at The Johns Hopkins Hospital in Baltimore, MD.
Caitlin Hicks, MD, MPH-V, is a Vascular Surgeon at The Johns Hopkins Hospital in Baltimore, MD.
Christopher Abularrage is a Vascular Surgeon at The Johns Hopkins Hospital and Director of the Diabetic Foot and Wound Care Service in Baltimore, MD.
Sanuja Bose, MD, MPH, is a statistical data analyst at The Johns Hopkins Hospital in Baltimore, MD.
Ronald Sherman, DPM, MBA, is Assistant Professor in the Department of Surgery and Program Director of the Limb Preservation and Wound Care Fellowship at The Johns Hopkins Hospital in Baltimore, MD. He also serves as the Principal Investigator for the DURS CAGA Study.
- International Diabetes Federation. IDF Diabetes Atlas. 8th ed. IDF: 2017.
- World Health Organization. Global Report on Diabetes. WHO: 2016
- Bourne RRA, Stevens GA, White RA, et al. Vision Loss Expert Group. Causes of vision loss worldwide, 1990-2010: a systematic analysis. Lancet Glob Health. 2013;1(6):e339-49.
- Moxey PW, Gogalniceanu P, Hinchliffe RJ, et al. Lower extremity amputations—a review of global variability in incidence. Diabet Med. 2011;28(10):1144-53.
- Dyck PJ, Kratz KM, Karnes JL, et al. The prevalence by staged severity of various types of diabetic neuropathy, retinopathy, and nephropathy in a population-based cohort: the Rochester Diabetic Neuropathy Study. Neurology. 1993;43(4):817-24.
- Saran R, Li Y, Robinson B, et al. US Renal Data System 2014 Annual Data Report: Epidemiology of Kidney Disease in the United States. Am J Kidney Dis. 2014;66(1 Suppl1):S188-210.
- Emerging Risk Factors Collaboration; Sarwar N, Gao P, Seshasai SR, et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet. 2010;375(9733):2215-22.
- International Diabetes Federation. IDF Diabetes Atlas. 8th ed. IDF: 2021.
- Mason J, O’Keeffe C, Hutchinson A, et al. A systematic review of foot ulcer in patients with type 2 diabetes mellitus. II: treatment. Diabet Med. 1999;16(11):889–909.
- Reiber GE, Lipsky BA, Gibbons GW. The burden of diabetic foot ulcers. Am J Surg 1998;176(2A Suppl):5S–10S.
- Armstrong DG, Boulton AJM, Bus SA. Diabetic foot ulcers and their recurrence. N Engl J Med. 2017; 376(24):2367–75.
- Prompers L, Huijberts M, Apelqvist J, et al. High prevalence of ischaemia, infection and serious comorbidity in patients with diabetic foot disease in Europe. Baseline results from the Eurodiale study. Diabetologia. 2007;50(1):18-25.
- Pound N, Chipchase S, Treece K, Game F, Jeffcoate W. Ulcer-free survival following management of foot ulcers in diabetes. Diabet Med. 2005;22(10):1306-9.
- Tennvall GR, Apelqvist J. Health–economic consequences of diabetic foot lesions. Clin Infect Dis. 2004;39(Suppl 2):S132-S139.
- Mathioudakis N, Hicks CW, Canner JK, et al. The Society for Vascular Surgery Wound, Ischemia, and foot Infection (WIfI) classification system predicts wound healing but not major amputation in patients with diabetic foot ulcers treated in a multidisciplinary setting. J Vasc Surg. 2017;65(6):1698-1705.e1.
- Faude O, Donath L, Roth R, Fricker L, Zahner L. Reliability of gait parameters during treadmill walking in community-dwelling healthy seniors. Gait Posture. 2012;36(3):444-8.