 Documenting how shock propagates through the leg and is attenuated by the soft tissues appears to be a critical step toward advancing practitioners’ and researchers’ understanding of lower extremity injury mechanisms related to running and landing activities.
Documenting how shock propagates through the leg and is attenuated by the soft tissues appears to be a critical step toward advancing practitioners’ and researchers’ understanding of lower extremity injury mechanisms related to running and landing activities.
By Timothy A. Burkhart, PhD, EIT, Alison Schinkel-Ivy, MHK, and David M. Andrews, PhD
The prevalence of lower extremity injuries associated with activities that expose participants to repetitive submaximal loading is high.1,2 For example, research has shown that individuals entering basic military training3-6 and those participating in competitive and recreational athletics3,7-9 all have an increased risk of lower extremity injury, with an estimated 20% to 75% of participants affected. Although these injuries can occur to soft tissues (e.g., muscle injuries), those that occur to bone (e.g., medial tibial stress syndrome and stress fractures) are of great interest given their relatively high occurrence (4% to 16% of all running-related injuries) 2 and lengthy recovery times (minimum of six weeks).10,11
A number of investigations have tried to identify the significant risk factors that lead to lower extremity bone injuries. These risk factors can be categorized as general (e.g., sex, age), physiological (e.g., diet), or biomechanical (e.g., impact force magnitude, anthropometrics).
Impact forces during running and jumping activities are produced as the feet come into contact with the ground, initiating a shock that propagates through the lower extremities (primarily through bone) toward the head. The propagating shock has the potential to produce microdamage to the bone,12 and when this process outpaces bone regeneration (i.e., through continuous exposure to impact with minimal rest) an injury may result.13

Figure 1: A schematic representation of an acceleration time curve showing important measures used previously to quantify the tibial response to impact (PA = peak acceleration; TPA = time to peak acceleration; AS = acceleration slope).
Although a number of extrinsic (e.g., shoe design) and intrinsic (e.g., joint angles) strategies have been developed that aim to minimize the magnitude of impact forces and attenuate the resultant shock, more recent attention has been given to the tissue composition of the lower extremities and the role this plays in shock attention and injury prevention.14
The primary tissues of the lower extremity are:
- muscle, or lean mass (LM),
- all intramuscular and subcutaneous adipose tissue, or fat mass (FM),
- bone tissue, or bone mineral content (BMC), and
- the combination of FM and LM, or wobbling mass (WM).
Background
There is general agreement that the tissues of the lower extremity are both affected by and can affect the propagating shock. With respect to impact forces (i.e., the initial force developed as the foot contacts the ground), Liu and Nigg15 and Pain and Challis16 determined that increased rigid (bone) mass in the lower extremity resulted in increased peak impact forces. However, when the role of the passive muscle mass was considered, the peak impact forces and loading rates17 were reduced significantly. Conversely, when the active muscle contributions were accounted for, the muscles increased the stiffness of the distal lower extremity, causing an increase in the peak forces, loading rates, and the time to peak force at the point of impact.18-20

Figure 2: DXA images of the thigh (a,b), leg (c,d), and foot segments (e,f) showing the skeletal (a,c,e) and soft (b,d,f) tissues. Also highlighted (blue boxes) are the manually selected regions of interest. (Reprinted with permission from Burkhart et al.32)
While impact forces provide an excellent measure of the response between the foot and the ground, accelerometers attached to bony anatomical landmarks can be used to examine the propagation of the shock through the tissues at locations proximal to the initial impact.21,22 Wosk and Voloshin23 determined the acceleration experienced at the tibial tuberosity was reduced by 70% by the time the shock wave reached the head. Similar to changes in the impact force variables, this attenuation can occur through either passive or active mechanisms; for example, movement of the soft tissues, or wobbling masses,24 relative to the underlying bone can occur either actively or passively.
Passive mechanisms of shock attenuation are associated with either the compression of the soft tissues themselves (such as the heel pad, articular cartilage, or meniscus) or with movement of the soft tissues.25-27 Contracting muscles actively before and during impact can also affect the amount of shock attenuation that occurs as the shock travels through the segment. For example, when specific muscles of the distal lower extremity were fatigued (reduced muscle activation), Duquette and Andrews,28 Flynn et al,29 and Holmes and Andrews30 all reported a decrease in the peak acceleration experienced at the tibial tuberosity following heel impact. Conversely, Holmes and Andrews30 observed an increase in acceleration variables (peak acceleration, acceleration slope, time to peak acceleration; see Figure 1) when the stiffness of the distal lower extremity was increased through increased muscle activation.
Tissue mass measurement
Research has shown the passive motion of soft tissues relative to bone has a positive association with the magnitude of soft tissue in the leg.31 Quantifying the magnitude of segmental tissue masses (i.e., LM, FM, BMC, WM) in living people has most commonly been accomplished using dual energy x-ray absorptiometry (DXA), an imaging technique that uses two x-ray beams of different energies to penetrate the body’s tissues. The resultant scanned images can be segmented manually by tracing polygons around regions of interest (Figure 2) with accompanying software. The individual tissue masses within these areas can then be calculated.
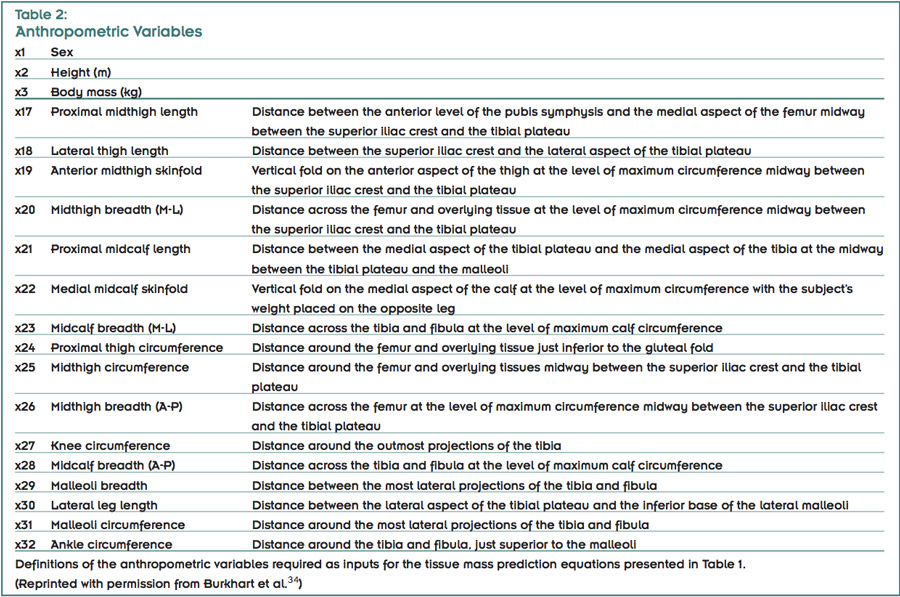
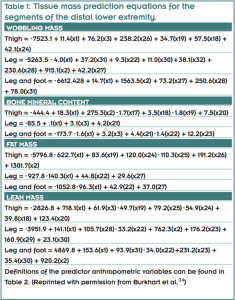 Although DXA is a reliable and accurate method for obtaining tissue mass estimates from living people,32 scanners are costly to purchase and operate, and are not always available for research purposes. Participants are also exposed to a small amount of x-ray radiation during every scan. In response to these limitations, Holmes et al33 developed regression equations (Table 1) that correlate a series of reliable anthropometric measurements (Table 2)34 to DXA-derived tissue masses (LM, FM, BMC, WM), providing a valid, cost effective, and easy-to-use method that quantifies the tissue masses in the thigh (hip to knee joint center), leg (knee joint center to ankle joint center), and foot (ankle joint center to distal phalanx) segments of healthy young adults.
Although DXA is a reliable and accurate method for obtaining tissue mass estimates from living people,32 scanners are costly to purchase and operate, and are not always available for research purposes. Participants are also exposed to a small amount of x-ray radiation during every scan. In response to these limitations, Holmes et al33 developed regression equations (Table 1) that correlate a series of reliable anthropometric measurements (Table 2)34 to DXA-derived tissue masses (LM, FM, BMC, WM), providing a valid, cost effective, and easy-to-use method that quantifies the tissue masses in the thigh (hip to knee joint center), leg (knee joint center to ankle joint center), and foot (ankle joint center to distal phalanx) segments of healthy young adults.
Although instrumentation methods for measuring the magnitude of shock traveling through bone are well established,35 measuring the movement of the soft tissues with respect to the bone has not been studied widely. Given the demonstrated shock-altering capacity associated with soft tissue motion, quantifying this motion relative to bone is a primary interest of our research group. We have successfully used high-speed video-based motion tracking systems, which utilize surface markers that don’t interfere with the natural motion of the soft tissues (Figure 3), to quantify soft tissue motion using commercially available software programs (e.g., ProAnalyst,36 LabView37). Information regarding the propagation of the shock (Figure 3) can be obtained when markers in various regions (within zones) are followed throughout impact.
In a recent investigation, Brydges et al36 found this method to be highly reliable, such that analysts who were working independently were able to repeatedly select the same marker to within .5 cm, resulting in maximum tissue velocity errors of only 1 m/s. This work also confirmed the oscillatory movement patterns of the soft tissue, with the greatest displacement and velocity occurring in more proximal zones of the leg (nearer to the knee), where the quantity of soft tissue is greatest. Figure 4 illustrates this finding for soft tissue velocity in the proximal direction. It also highlights that regional differences in soft tissue motion exist between genders, likely due in part to differences in the quantity and composition of the soft tissues in these regions.
Current concepts
As suggested previously, the magnitude and proportion of tissues in the distal lower extremity may affect how impact-induced shock propagates through the leg, and how tissue damage and injury can potentially develop through cumulative mechanisms.

Figure 3: A single high-speed video frame showing the experimental set up for tracking skin markers in response to a pendulum heel strike (simulating landing during running). The numbers 1-10 represent the different regions corresponding to those shown in Figure 4 (page 50).
Schinkel-Ivy et al,35controlling for muscle activation, investigated the effects of whole body and local leg tissue composition on shock propagation. In this study, participants were subjected to consistent, nonweight-bearing (i.e., horizontally oriented), running-type impacts (velocity = 1 m/s–1.15 m/s; force = 1.8–2.8 times body weight) to the heel through a custom-designed human pendulum apparatus;28-30 the propagation of the resulting shock was measured at the tibial tuberosity.
The authors found higher magnitudes of lean and bone mass within the leg (calculated using the Holmes et al33 equations) resulted in decreased measures of shock. When normalized to LM, WM, and BMC, women experienced 50%, 62%, and 70% greater peak acceleration, respectively, than men as expressed per gram of tissue. These results showed clearly that tissue composition affects the ability of the leg to attenuate shock following impact, and provides insight into lower extremity overuse injury mechanisms and may help to explain why women are five times more likely than men to sustain a lower extremity impact related tibial bone injury.3
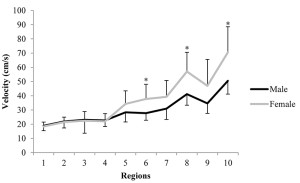
Figure 4: Comparison of the mean velocities of the soft tissue moving toward the head, across regions of the leg, and between sexes (*p < .05).
The work by Schinkel-Ivy et al35 highlighted a viable relationship between tissue mass composition and shock magnitude, but did not directly assess the link between composition and injury. In response, Burkhart et al38 conducted a preliminary investigation of 100 university athletes (participating in basketball, volleyball, soccer, and cross-country) to determine the variables most strongly associated with lower extremity bone tissue injuries. The purpose of this work was to develop multivariate injury risk prediction equations, focused on tissue mass composition, that could be used to identify athletes at the highest risk of sustaining a bone injury over the course of their respective seasons. The study found a significant difference in the FM:BMC ratio between those who reported an injury and those who did not, with the FM:BMC ratio being larger in individuals who sustained a tibial bone injury.38 Leg FM:BMC ratio and playing surface were also significant predictors of bone injury. The risk of injury was almost double for every unit increase in the FM:BMC ratio.
The finding that tissue composition affects the propagation of shock waves through the lower extremity (which, in turn, is thought to influence injury risk) has implications for athletes and coaches involved in sports characterized by repetitive impacts.
In a recent study by Schinkel-Ivy et al,39 leg tissue masses were predicted for basketball, volleyball, soccer, and cross-country athletes, from which tissue mass ratios (LM:FM, LM:BMC, FM:BMC) were calculated. Cross-country athletes tended to have the lowest tissue mass magnitudes, while basketball (male) and volleyball (female) athletes had the highest. In addition, among the female athletes, cross-country runners had the highest WM:BMC and LM:BMC ratios. From the same cohort, more pain was reported by athletes who were categorized as having low LM:FM and LM:BMC ratios and high FM:BMC ratios.40
The differences in tissue mass ratios between the sexes and between athletes of different teams suggests that the ratios of soft to rigid tissues are optimized by the body for the typical loading patterns experienced by the athletes in each sport. Pain reporting may also be affected by these ratios, assuming that significant departures from optimal tissue mass ratios may put athletes at greater risk of injury.
This body of work suggests that even though individual tissues (e.g., bone mineral content) of the lower extremity are associated with injury risk, it may also be important to consider the relative contribution of each tissue type to the overall composition of the lower extremity (i.e., the ratios of tissues) when assessing injury risk, given the demonstrated importance of both soft and rigid tissues to impact force attenuation.
Conclusions
Taken together, the results detailed above highlight the importance of soft tissues in attenuating shock through the distal lower extremity during impacts consistent with those experienced during running and landing activities. They also illustrate the importance of monitoring the effects of impacts at locations more proximal to the impact site.
 Documenting how the shock propagates through the leg and is attenuated by the soft tissues appears to be a critical step toward advancing our understanding of lower extremity injury mechanisms. It is also important to consider the effects of tissue composition in athletes of different sports and genders to provide additional insights into the complexities of lower extremity musculoskeletal injury risk.
Documenting how the shock propagates through the leg and is attenuated by the soft tissues appears to be a critical step toward advancing our understanding of lower extremity injury mechanisms. It is also important to consider the effects of tissue composition in athletes of different sports and genders to provide additional insights into the complexities of lower extremity musculoskeletal injury risk.
Future directions
Biomechanical models of the human body can provide valuable information about the response of soft and rigid tissues to impacts, but many existing models do not account for tissue composition and motion differences reported in the literature. In part, this is due to the cost of developing appropriately sophisticated models.
Future work in this area should expand on preliminary efforts addressing this important issue using existing models (e.g., Schinkel-Ivy et al41),and by developing more anatomically biofidelic wobbling mass models that take tissue differences (e.g., between the sexes) into consideration. In vitro impact work, such as that documented by Burkhart42,43 for the upper extremity, needs to be carried out for the lower extremity to improve our understanding of injury mechanics and help build and validate detailed anatomical models. A comprehensive approach that utilizes both experimental (living participants and human cadaveric specimens) and modeling studies will facilitate the greatest gains in understanding, such that improved injury prevention and treatment strategies can be implemented by practitioners at all levels.
Timothy Burkhart, PhD, EIT, is a CIHR-Bisby postdoctoral fellow in the Jack McBain Biomechanics Testing Laboratory and the Hand and Upper Limb Centre at the University of Western Ontario in London, Canada. Alison Schinkel-Ivy, MHK, is a PhD candidate in the Spine Biomechanics Laboratory in the School of Kinesiology and Health Science at York University in Toronto. David Andrews, PhD, is a professor in the Department of Kinesiology and a research chair in the faculty of Human Kinetics at the University of Windsor.
Acknowledgements
We would like to thank the Natural Sciences and Engineering Research Council of Canada and the Canadian Society for Biomechanics for funding, and Don Clarke, Evan Brydges, Jennifer Stefanczyk, Robyn Bertram, Mike Angelidis, William Altenhof, Kathy Harvie, and Dave Stoute for their valuable contributions to this work.
1. Brubaker CE, James SL. Injuries to runners. J Sports Med 1974;2(4):189-198.
2. Bennell KL, Malcolm SA, Thomas SA, et al. The incidence and distribution of stress fractures in competitive track and field athletes. A twelve-month prospective study. Am J Sports Med 1996;24(6):211-217.
3. Jones BH, Thacker SB, Gilchrist J, et al. Prevention of lower extremity stress fractures in athletes and soldiers: A systematic review. Epidemiol Rev 2002;24(2):228-247.
4. Shaffer RA, Rauh MJ, Brodine SK, et al. Predictors of stress fracture susceptibility in young female recruits. Am J Sports Med 2006;34(1):108-115.
5. Davidson PL, Wilson SJ, Chalmers DJ, et al. Examination of interventions to prevent common lower-limb injuries in the New Zealand Defense Force. Mil Med 2009;174(11):1196-1202.
6. Knapik JJ, Trone DW, Swedler DI, et al. Injury reduction effectiveness of assigning running shoes based on plantar shape in Marine Corps basic training. Am J Sports Med 2010;38(9):1759-1767.
7. Hreljac A, Marshall RN, Hume PA. Evaluation of lower extremity overuse injury potential in runners. Med Sci Sports Exerc 2000;32(9):1635-1641.
8. Milner CE, Ferber R, Pollard CD, et al. Biomechanical factors associated with tibial stress fracture in female runners. Med Sci Sports Exerc 2006;38(2):323-328.
9. Buist I, Bredeweg SW, Lemmink KA, et al. Predictors of running-related injuries in novice runners enrolled in a systematic training program: a prospective cohort study. Am J Sports Med 2010;38(2):273-280.
10. Beck TJ, Ruff CB, Shaffer RA, et al. Stress fracture in military recruits: gender differences in muscle and bone susceptibility factors. Bone 2000;27(3):437-444.
11. Franklyn M, Oakes B, Field B, et al. Section modulus is the optimum geometric predictor for stress fractures and medial tibial stress syndrome in both males and females. Am J Sports Med 2008;36(6):1179-1189.
12. Radin EL, Parker HG, Pugh JW, et al. Response of the joints to impact loading. 3. Relationship between trabecular microfractures and cartilage degeneration. J Biomech 1973;6(1):51-57.
13. Bennell KL, Brukner P. Preventing and managing stress fracture in athletes. Phys Ther Sport 2005;6(4):171-180.
14. Nigg BM, Liu W. The effect of muscle stiffness and damping on simulated impact force peaks during running. J Biomech 1999;32(8):849-856.
15. Liu W, Nigg BM. A mechanical model to determine the influence of masses and mass distribution on the impact force during running. J Biomech 2000;33(2):219-224.
16. Pain MTG, Challis JH. The influence of soft tissue movement on ground reaction forces, joint torques and joint reaction forces in drop landings. J Biomech 2006;39(1):119-124.
17. Gerritsen KG, van den Bogert AJ, Nigg BM. Direct dynamics simulation of the impact phase in heel-toe running. J Biomech 1995;28(6):661-668.
18. Butler RJ, Crowell 3rd HP, Davis IM. Lower extremity stiffness: implications for performance and injury. Clin Biomech 2003;18(6):511-517.
19. Nigg BM, Cole GK, Bruggeman GP. Impact forces during heel-toe running. J Appl Biomech 1995;11(4):407-432.
20. Pain MTG, Challis JH. Soft tissue motion during impacts: Their potential contribution to energy dissipation. J Appl Biomech 2002;18(3):231-242.
21. Burkhart TA, Andrews DM. The effectiveness of wrist guards for reducing wrist and elbow accelerations resulting from simulated forward falls. J Appl Biomech 2010;26(3):281-289.
22. Burkhart TA, Andrews DM. Activation level of extensor carpi ulnaris affects wrist and elbow acceleration response following simulated forward falls. J Electromyogr Kinesiol 2010;20(6):1203-1210.
23. Wosk J, Voloshin A. Wave attenuation in skeletons of young healthy persons. J Biomech 1981;14(4):261-267.
24. Gruber K, Ruder H, Denoth J, Schneider K. A comparative study of impact dynamics: wobbling mass model versus rigid body models. J Biomech 1998;31(5):439-444.
25. Derrick TR, Hamill J, Caldwell GE. Energy absorption of impacts during running at various stride lengths. Med Sci Sports Exerc 1998;30(1):128-135.
26. Fukuda Y, Takai S, Yohino N, et al. Impact load transmission of the knee joint-influence of leg alignment and the role of meniscus and articular cartilage. Clin Biomech 2000;15(7):516-521.
27. Dufek JS, Mercer JA, Griffin FR. The effects of speed and surface compliance on shock attenuation characteristics for male and female runners. J Appl Biomech 2009;25(3):219-228.
28. Duquette AM, Andrews DM. Tibialis anterior fatigue leads to changes in tibial response to impact when joint angles are controlled. Hum Move Sci 2010;29(4):567-577.
29. Flynn JM, Holmes JD, Andrews DM. The effect of localized leg muscle fatigue on tibial impact acceleration. Clin Biomech 2004;19(7):726-732.
30. Holmes AM, Andrews DM. The effect of leg muscle activation state and localized muscle fatigue on tibial response during impact. J Appl Biomech 2006;22(4):275-284
31. Brydges E. The effects of sex, leg region and impact technique on leg soft tissue motion and energy dissipation following heel impacts. [master’s thesis]. Windsor, Canada: University of Windsor; 2013.
32. Burkhart TA, Arthurs KL, Andrews DM. Manual segmentation of DXA scan images results in reliable upper and lower extremity soft and rigid tissue mass estimates. J Biomech 2009;42(8):1138-1142.
33. Holmes JD, Andrews DM, Durkin JL, Dowling JJ. Predicting in-vivo soft tissue masses of the lower extremity using segment anthropometric measurements and DXA. J Appl Biomech 2005;21(4):371-382.
34. Burkhart TA, Arthurs KL, Andrews DM. Reliability of upper and lower extremity anthropometric measurements and the effect on tissue mass predictions. J Biomech 2008;41(7):1604-1619.
35. Schinkel-Ivy A, Burkhart TA, Andrews DM. Leg tissue mass composition affects tibial acceleration response following impact. J Appl Biomech 2012;28(1):29-40.
36. Brydges EA, Burkhart TA, Altenhof WJ, Andrews DM. Reliability of leg soft tissue marker motion following manual digitization. Presented at the 17th Canadian Society for Biomechanics Conference, Vancouver, British Columbia, June 2012.
37. Reeves JM, Burkhart TA, McLachlin SD, Dunning CE. A colour-thresholding technique to quantify high-speed planar motion of isolated distal radii specimens during impact. Presented at the 10th Ontario Biomechanics Conference, Barrie, Ontario, March 2013.
38. Burkhart TA, Schinkel-Ivy A, Andrews DM. Tissue mass ratios and the reporting of distal lower extremity injuries in varsity athletes at a Canadian University. J Sports Sci 2013;31(6):684-687.
39. Schinkel-Ivy A, Burkhart TA, Andrews DM. Difference in distal lower extremity tissue masses and mass ratios exist in athletes of sports involving repetitive impacts. J Sports Sci 2013. In press.
40. Angelidis M. Association between injury and reported pain and lower extremity tissue mass ratios in varsity athletes. [master’s thesis]. Windsor, Canada: University of Windsor; 2011.
41. Schinkel-Ivy A, Altenhof WJ, Andrews DM. Validation of a full body finite element model (THUMS) for running-type impacts to the lower extremity. Comput Methods Biomech Biomed Engin 2012 Apr 23. [Epub ahead of print]
42. Burkhart TA, Andrews DM, Dunning CE. Failure characteristics of the isolated distal radius in response to dynamic impact loading. J Orthop Res 2012;30(6):885-892.
43. Burkhart TA, Andrews DM, Dunning CE. Multivariate injury risk criteria and injury probability scores for fractures to the distal radius. J Biomech 2013;46(5):973-978.









EXCELLENT ARTICLE…I HAVE FOUND THAT THE AMOUNT OF WOBBLE IN RUNNERS DIRECTLY CORRELATES WITH INJURY. THE LACK OF ATTENTION UNTIL RECENTLY OF THE ROLE OF SOFT TISSUE PATHOLOGY TO INJURY PREVENTION IS STARTLING AS A PHYSICIAN, RUNNER, BIOMECHANIST IT IS ENCOURAGING THAT WE ARE NOW OBTAINING SCIENTIFIC PROOF TO DOCUMENT THIS…..