 Given the limitations of pharmacotherapy options for treating painful diabetic peripheral neuropathy, practitioners are also considering the merits of cognitive therapy, orthotic management, and combination therapies to relieve patients’ pain.
Given the limitations of pharmacotherapy options for treating painful diabetic peripheral neuropathy, practitioners are also considering the merits of cognitive therapy, orthotic management, and combination therapies to relieve patients’ pain.
By Larry Hand
Deciding how to treat a patient who suffers from peripheral neuropathic pain can be as complicated as the underlying disease from which the pain originates. Not only does the practitioner need to consider the symptom of pain, he or she also has to delve into comorbidities that often accompany the pain—including sleep deprivation, anxiety, and depression—and weigh the likelihood of noncompliance. Determining a treatment plan requires a head-to-toe analysis of the patient’s individual circumstances.
Recently published studies and reviews, however, offer an informative basis for treating the pain aspect of painful diabetic peripheral neuropathy (PDPN), a common complication of diabetes that produces pain in the hands and feet.
Neuropathic pain is prevalent among about 7% of the general US population, but about 20% of people with diabetes experience the condition,1 defined as “pain arising as a direct consequence of a lesion or disease affecting the somatosensory system.”2
“[PDPN] is a difficult-to-manage clinical problem, and patients with this condition are more apt to seek medical attention than those with other types of neuropathy,” wrote Aaron I. Vinik, MD, PhD, and Carolina M. Casellini, MD, of the Strelitz Diabetes Center for Endocrine and Metabolic Disorders at Eastern Virginia Medical School in Norfolk, VA, in their recent review of guidelines for diabetic nerve pain management.3 They also noted, “Early recognition of psychological problems is critical to the management of pain, and physicians need to go beyond the management of pain per se if they are to achieve success.”
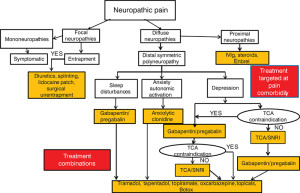
Figure 1. Treatment algorithm: neuropathic pain after exclusion of nondiabetic etiologies and stabilization of glycemic control. Abbreviations: IVIg, intravenous immunoglobulin; SNRI, serotonin-noradrenaline reuptake inhibitors; TCA, tricyclic antidepressants.
(Reprinted with permission from reference 3.)
A 2011 review described recent increases in prevalence rates for depression, anxiety, and diabetes complications, including PDPN, and examined associations among these conditions.4 While overall age-adjusted prevalence of anxiety amounts to 10.9% in people without diabetes, it is almost twice as high, at 19.5%, in people who have diabetes, according to findings from the 2006 Behavioral Risk Factor Surveillance System.5
Evidence is increasingly pointing to a circular relationship. Emotional stress, including depression and anxiety, can increase the risk of developing diabetes, and vice versa. Shared neurobiological systems that may contribute to these associations include the neuroendocrine system, autonomic system, and inflammatory cytokine system, as well as interrelated physiological pathways in the brain and the body.
Like the factors underlying PDPN, treatment of the condition is also multifaceted. Approaches to treating PDPN include pharmacotherapeutics, cognitive therapy, and orthotic management.
Pharmacotherapy approaches
Pharmaceutical management of PDPN is more complicated than many practitioners realize, according to Vinik.
“The single biggest factor is if you don’t consider the comorbidities, and if you don’t look at the underlying cause of pain, you use the wrong drugs in the wrong patients,” Vinik told LER in an interview.
Mistakes happen for many reasons, he added, with one major reason being misdiagnosis of the underlying cause of pain. For instance, patients with diabetes can develop pain as a result of heel spurs, Morton’s neuroma, ischemia, and other non-neuropathic causes.
“All of those produce pain, and if you try and use a drug that is known to relieve neuropathic pain, you’re making a big mistake, because most of those patients don’t respond to that,” Vinik explained.
Patients have described pain originating from peripheral nerve injury as burning or shooting with unusual tingling. “Some describe bees stinging through the socks while others talk of walking on hot coals,” wrote authors of a 2008 paper.2 The pain is often worse at night, which contributes to sleep deprivation.
Authors of a 2007 paper2 redefined neuropathic pain and established a grading system to help guide the degree of certainty regarding its diagnosis. Some combination of the four grading system criteria must be present for a diagnosis of definite, probable, or possible neuropathic pain. For example, a diagnosis of definite neuropathic pain requires the presence of all criteria (grades 1-4), while the presence of only grade 1 (defined as pain with an identifiable neuroanatomical distribution) plus grade 2 (defined as a history of a relevant lesion or disease affecting the peripheral central nervous system) indicates probable neuropathic pain.
Other methods have been developed to help with diagnosis of and differentiation between nociceptive and non-nociceptive pain, but there is no laboratory test that can quantify neuropathic pain.3
The Vinik-Casellini guidelines paper offers some important considerations for pharmacotherapy, including titrating a drug dose based on monitoring its effectiveness when first prescribing it for the patient, judging efficacy only after two to four weeks, possible advantages of drug combinations, and the need to guard against drug interactions.
Two drugs have been approved in the US for treatment of neuropathic pain: pregabalin (300-600 mg/day) and duloxetine (60-120 mg/day), and the American Academy of Neurology recom- mends only pregabalin as a Level A treatment (highest level of evidence) for PDPN.6 Neither, however, provides complete relief.
“Indeed, a sobering view is that few drugs achieve greater than 30% reduction in pain in >50% of patients, dictating a need to use more than one drug with different mechanisms of action,” Vinik and Casellini wrote.3
A number of other drugs have been tried and used for this indication, including tricyclics, selective serotonin reuptake inhibitors, serotonin norepinephrine reuptake inhibitors, anticonvulsants, opioids, topical treatments, and injections.3 The European Federation of Neurological Societies recommends a number of them as first-line treatments.7
“We don’t have any approved agents for treating the underlying diabetic pathophysiology of diabetic neuropathic pain,” Vinik said. “The real reason for that is that none has been shown to be effective in terms of the disease. We then need to resolve what is going to be the best Band Aid for the management of patients with neuropathic pain.”
Vinik explained one dilemma in choosing treatments.
“Take a comparison of amitriptyline, duloxetine, and pregabalin on sleep disturbance. If you’re relieving pain, then each of these drugs will actually improve sleep if sleep disturbance was the effect of the patient’s pain perception. Amitriptyline actually does nothing for sleep. Duloxetine actually fragments sleep and makes it worse. For a person who has sleep disturbance with pain you only really have one class of drugs, either gabapentin or pregabalin.”
Practitioners today most commonly prescribe the convulsant gabapentin and the tricyclic amitriptyline, Vinik said.
“They do that simply by cost or their familiarity with those drugs, which have been around for a long time. That ignores the fact that people who have significant pain do so in the environment of the patient’s predisposition to depression, anxiety, and sleep loss,” he said.
To gain evidence that might convince practitioners to do otherwise, he and his colleagues conducted a study on how pregabalin affects PDPN and postherpetic neuralgia.8
“The issue for us in this particular study is whether it was possible—using data based on the several studies that have been done on pregabalin and diabetic peripheral neuropathy and posterpertic neuralgia—to establish if the drug was doing so by direct effect on pain relief or doing so as a direct effect combined with an indirect effect. We concluded that there were two very clear pathways. One was the pathway of the impact on pain reduction by improvement of sleep, and the other was the direct pathway on pain relief,” he explained.
Other recent papers have compared PDPN treatments from various angles. In a cost-utility analysis, University of Utah researchers compared how pregabalin and duloxetine stack up from a payer’s perspective.9
“Initially we set out to compare the value of several drugs, but one of the goals of our study was really to incorporate real-world data regarding effectiveness and adherence and those kinds of things that weren’t used in previous pharmacoeconomic analyses,” Brandon K. Bellows, PharmD, research associate at the Pharmacotherapy Outcomes Research Center in Salt Lake City, told LER. “Due to limitations in the data availability, we ended up focusing primarily on pregabalin and duloxetine, which made sense at the time the article was published because they were both brand-name drugs. From a healthcare payer perspective, it’s important to decide which of these brand-name drugs are worth paying for.”
In the base-case pharmacoeconomic model (in which analysis results are expected to be better than in a conservative case scenario but worse than in an aggressive case scenario), the researchers concluded that duloxetine cost less after six months (-$187 in 2011 US dollars) and was more effective (+.011 quality adjusted life-years [QALYs]) than pregabalin. The base-case analysis was based on US Food and Drug Administration-approved doses of 300-mg pregabalin and 60-mg duloxetine.
They also conducted “real-world” sensitivity analyses, in which a range of doses that may actually be prescribed in clinical practice were examined. In these analyses investigators found duloxetine cost more but was still more effective than pregabalin. Duloxetine had an incremental cost-effectiveness ratio of $16,300/QALY, meaning treatment with duloxetine would cost $16,500 more for each additional QALY gained compared with pregabalin. In the US, $50,000/QALY is commonly used as the willingness-to-pay threshold.
“We included real-world data,” Bellows said. “We included the adherence portion of the equation. And we also looked at strategies that aren’t necessarily recommended by the guidelines but might be used by practitioners in the real world. Additionally, we examined concomitant medication use. No prior studies had actually looked at that cost effectiveness of maybe adding on a therapy in patients with diabetic neuropathy.”
The “real-world” analysis is a strength of their study, Bellows said.
“When a payer looks at randomized trial, they can say, ‘OK, this drug can work in an ideal population, but does it work in the population that I’m covering and I represent?’ Observational data bridges that gap. In a real-world study, a payer can look at that study and say, ‘All right, this drug does work in a population that I represent,’” he said.
The recent Vinik study also found a flexible-dose benefit.
“If you look at people who have neuropathic pain and ask them about it, it’s not the same from day to day. It’s different,” Vinik explained. “It’s different at different times of day, and for most people who have diabetic neuropathic pain it’s much worse in the evening.”
In a systematic review and meta-analysis published online in March, researchers analyzed 58 studies that involved 29 interventions in 11,883 patients with PDPN.10 They found that pain reduction with a intervention versus placebo on an 11-point numeric rating scale ranged from -3.29 for anticonvulsant sodium valproate to 1.67 for nabiximol, a cannabinoid mouth spray. Pregabalin came in at
-1.08 and duloxetine at -1.06.
“Of the 29 different interventions studied, those studied in larger trials were similar to one another in their ability to reduce pain, and several were associated with no more adverse events than placebo,” explained Sonya Snedecor, PhD, a biomathematician and director of health economics and operations at Pharmerit International, in an email to LER.
However, the studies varied by length of study, number of participants, and methods of judging pain reduction. In their review, Snedecor and colleagues cited a need for closer examination of how these factors influence treatment effects.
Snedecor recommends that practitioners follow guidelines when making treatment decisions.
“These guidelines are typically created based on examination of the quality and results of individual studies. This analysis complements the guidelines by providing a way to synthesize the data from individual studies into a single analysis to provide a measure of comparative effectiveness,” she wrote. “Mathematics and clinical scientists have formed lots of collaborations to conduct analyses and create models to evaluate evidence which is difficult to obtain through clinical means. In this case, through statistics we have simulated a ‘29-arm clinical trial’ to compare different treatments for PDPN that would not have been possible otherwise.”
Cognitive therapy approaches
Results from a recent randomized controlled pilot study suggest that cognitive behavioral therapy (CBT) may provide PDPN patients with skills to become more active, which could lead to less pain.11 Researchers at the VA Boston Healthcare System in Massachusetts conducted a four-month trial in which veterans with PDPN were randomized (nonblinded) to receive either CBT or treatment as usual (TAU). The eight CBT participants included in the final analysis reported significant decreases in pain severity and pain interference from pretreatment to four-month follow-up, but no differences occurred in the eight-person TAU group.
TAU participants received no treatment for pain other than treatment by their primary care physician or other healthcare providers. CBT participants took part in 11 weekly individual sessions with a PhD-level clinical psychologist or a master’s-level therapist. Each session lasted an hour, during which they discussed the impact and cycle of pain and established goals for treatment. Participants learned relaxation techniques and how to identify and challenge negative thoughts, as well as a time-based pacing technique and the importance of scheduling pleasant activities.
Several participants receiving CBT experienced a substantial reduction in the severity of their pain; two reported a reduction of 50% or more in the severity of pain and three participants reported experiencing at least a 20% reduction in their pain.
Orthotic management approaches
The primary goal of using footwear and foot orthoses to offload plantar pressures in patients with diabetic neuropathy is to reduce the risk of future ulcerations, but this treatment option can also help manage pain associated with PDPN.
“Where I practice, patients with diabetes and peripheral neuropathy are mostly eligible for diabetic shoes. These shoes include multiple-depth plastazote insoles, which are conformant and can reduce plantar pressures,” said David Shofler, DPM, assistant professor in the Department of Podiatric Medicine, Surgery, and Biomechanics at the Western University of Health Sciences College of Podiatric Medicine in Pomona, CA, in an email to LER. “These shoes can be a nice option for patients with neuropathic pain, as well. For a good deal of patients with diabetic nerve pain, shoe gear won’t affect their pain level at all. These patients would be just as uncomfortable in sandals as they would be in custom diabetic shoes. That being said, there are patients whose discomfort can be exacerbated with certain shoes. This can occur, for example, when shoe gear has insufficient padding under the metatarsal heads, an insufficient arch support, or an inappropriate fit. Diabetic shoes can help reduce their overall discomfort, though persistence of neuropathic symptoms is likely.”
Shofler explained the other aspects of the approach he uses when treating a first-time patient with PDPN.
“When a patient first presents, the initial concern is glycemic control. If the patient has high blood sugar levels, I try to address this before considering any medications. If the blood sugars are well-controlled but the patient is having persistent neuropathic pain, I then encourage physical activity and appropriate nutritional support. I also consider the antioxidant alpha lipoic acid,” he said. “If the patient is still having neuropathic pain, I will then start either gabapentin or pregabalin. Beyond this, there are several variables that influence how I would proceed further. Each patient presents a little differently. In some cases I end up treating the pain without needing to address nutritional issues, while in others I can avoid any pharmacotherapy. ”
Shofler said published guidelines are helpful, to a point.
“In my experience, though, practitioners are sometimes not as well-read on this topic as they could be,” he said.
Vinik agrees. Too many practitioners, he said, gloss over the evidence in favor of the cheapest or most common drug.
“We should be educating [patients] on the different mechanisms of neuropathic pain and the different approaches,” Vinik said. “That’s a very big undertaking, but it’s what we should be doing.”
Larry Hand is a writer in Massachusetts.
1. Vinik A. The approach to the management of the patient with neuropathic pain. J Clin Endocrinol Metab 2010;95(11):4802-4811.
2. Treede RD, Jensen TS, Campbell JN, et al. Neuropathic pain: redefinition and a grading system for clinical and research purposes. Neurology 2008;70(18):1630-1635.
3. Vinik AI, Casellini CM. Guidelines in the management of diabetic nerve pain: clinical utility of pregabalin. Diabetes Metab Syndr Obesity 2013;6:57-78.
4. Jain R, Jain S, Raison CL, Maletic V. Painful diabetic neuropathy is more than pain alone: examining the role of anxiety and depression as mediators and complicators. Curr Diab Rep 2011;11(4): 275-284.
5. Li C, Ford ES, Zhang X, et al. Diabetes and anxiety in US adults: findings from the 2006 Behavioral Risk Factor Surveillance System. Diabet Metab 2008;25(7):878-881.
6. Tesfaye S, Vileikyte L, Rayman G et al. Painful diabetic peripheral neuropathy: consensus recommendations on diagnosis, assessment and management. Diabetes Metab Res Rev 2011 Jun 21. [Epub ahead of print]
7. Attal N, Cruccu G, Baron R et al. EFNS guidelines on the pharmacological treatment of neuropathic pain: 2010 revision. Eur J Neurol 2010;17(9):1113-e88.
8. Vinik A, Emir B, Cheung R, Whalen E. Relationship between pain relief and improvements in patient function/quality of life in patients with painful diabetic peripheral neuropathy or postherpetic neuralgia treated with pregabalin. Clin Ther 2013;35(5):612-623.
9. Bellows BK, Dahal A, Jiao T, Biskupiak J. A cost-utility analysis of pregabalin versu duloxetine for the treatment of painful diabetic neuropathy. J Pain Pall Care Pharmacother 2012;26(2):153-164.
10. Snedecor SJ, Sudharshan MD, Cappelleri JC, et al. Systematic review and meta-analysis of pharmacological therapies for painful diabetic peripheral neuropathy. 2013 Mar 28 [Epub ahead of print]
11. Otis JD, Sanderson K, Hardway C, et al. A randomized controlled pilot study of a cognitive behavioral therapy approach for painful diabetic peripheral neuropathy. J Pain 2013;14(5):475-482.








