 Preliminary research suggests platelet-rich plasma (PRP) is a safe and effective means of treating hamstring injuries that do not respond to early conservative measures. As an adjuvant to physical therapy, PRP may help delay or obviate surgery for partial hamstring tears.
Preliminary research suggests platelet-rich plasma (PRP) is a safe and effective means of treating hamstring injuries that do not respond to early conservative measures. As an adjuvant to physical therapy, PRP may help delay or obviate surgery for partial hamstring tears.
By Frank B. Wydra, MD; Ryan R. Fader, MD; Omer Mei-Dan, MD; and Eric C. McCarty, MD
Hamstring injuries, acute and chronic, are common in long distance runners, sprinters, and jumpers. The hamstring muscle group extends the leg at the hip and flexes the leg at the knee; it consists of three muscles contained within the posterior compartment of the thigh.
The semimembranosus, semitendinosus, and the long head of the biceps femoris originate from the ischial tuberosity. The short head of the biceps femoris originates from the linea aspera on the posterior femur. Each inserts just distal to the knee joint. The semimembranosus inserts onto the posterior surface of the medial tibial condyle. The semitendinosus inserts onto the anterior proximal medial tibia at the pes anserine. The long and short heads of the biceps muscles merge and insert into the proximal fibula and proximal lateral tibia.1
All hamstring muscles are innervated by the tibial component of the sciatic nerve with the exception of the short head of the biceps, which is innervated by the peroneal division of the sciatic nerve.1
Injuries to the hamstrings are commonly seen in athletes at all levels of competition. They are usually referred to as pulled or strained hamstrings. The typical injury mechanism involves an athlete performing rapid and eccentric contractions of the hamstring complex. Examples include sprinting, jumping, and swift acceleration and deceleration.2-4 These types of actions can stem from kicking sports like soccer or other activities such as water-skiing.5

Figure 1. The PRP preparation rich in growth factors (PRGF) technique. The leukocyte-poor layer lies just above the white blood cell (WBC) layer and is considered pure PRP, without WBC. (Photo courtesy of Omer Mei-Dan, MD.)
There are a wide variety of injuries to the hamstring muscle-tendon complex. The majority of acute injuries occur at the myotendinous junction, an area prone to muscular strains. However, the myotendinous junction of the hamstring muscles, unlike other muscles, spans most of the muscle belly due to long proximal and distal tendons as described by Woodley and Mercer.1 Koulouris and Connell reviewed 179 acute hamstring injuries in athletes and found that nearly 90% of tears occur at the myotendinous junction, a finding that has been supported throughout the literature.6-11 Approximately 12% of the patients in the study suffered proximal injuries, either avulsion of the ischial tuberosity or a partial tear proximally. Fewer than 1% of the patients had injuries of the distal tendons. Taking into account all locations of hamstring injuries, the biceps femoris was most commonly injured, accounting for about 80% of the cases.6,12,13
Although the mechanism of injury and underlying pathology of hamstring injuries is well described, treatment options vary widely without an agreed-upon gold standard, and are affected by age, activity level, and the possible need for rapid return to play. The last variable can be the main determining factor in the decision-making process for professional athletes.3,5,14-16 As with other muscle-tendon injuries, early treatment options consist of mostly conservative measures, including rest, activity modification, nonsteroidal anti-inflammatory drugs, physiotherapy, corticosteroid injections, therapeutic ultrasound, laser therapy, orthoses, and topical glycerine.15,17-19
A meta-analysis of 56 studies recently evaluated the role of eccentric exercises for the treatment of various tendinopathies and muscular injuries.20 Although the results showed promise for Achilles tendinopathy and patellar tendinitis, their search yielded only one randomized controlled trial for partial tears of the hamstring myotendinous unit, not including avulsion or complete disruption injuries. The trial found that eccentric exercises had no difference in outcomes compared with a progressive running regimen for pain or timing of return to sport.21
A Cochrane Database search performed in 2012 identified only two randomized controlled trials looking at physical therapy for partial tears of the hamstrings.22 The first suggested that stretching could reduce time to return to sport in elite athletes.23 The second study looked at participants of varying athletic levels and found no difference in time to return to sport, pain, or participant satisfaction; however, the authors did see a decreased risk of reinjury in the individuals who performed physical therapy.24
More recently, in 2014, Askling et al performed a prospective randomized comparative study looking at a lengthening rehabilitation protocol versus conventional physical therapy in 56 Swedish sprinters with magnetic resonance imaging-confirmed acute hamstring tears. Sprinters who underwent the lengthening protocol had a significantly shorter return to sport compared with a conventional protocol; 49 days versus 86 days, respectively.25 This is the first insight into an injury-specific rehabilitation for hamstring injuries as there are no currently accepted standardized physical therapy protocols for partial tears of the hamstring myotendinous unit. Further randomized controlled studies are needed to validate the effectiveness of various proposed protocols.
In unrelenting cases of proximal hamstring tendinosis, surgical intervention may be necessary. This is the scenario more commonly seen in complete avulsion injuries of the proximal hamstrings or unhealed partial tears. Options include open or minimally invasive endoscopic surgical debridement of the hamstrings footprint, drilling of the ischium, and various types of repair or reconstruction.25
Recent literature shows successful results from surgical treatment of hamstring injuries, although one must be aware of the indications and the rehabilitation course.27,28 A literature review by Harris et al performed in 2011 found that individuals who underwent surgical treatment of hamstring injuries, mainly acute proximal tendon ruptures and proximal bony avulsion injuries, were more likely to return to their preinjury state, had higher levels of patient satisfaction, and demonstrated improved strength compared with those who received nonsurgical treatment.29 Although promising, this analysis does have its limitations, including heterogeneity among patients included, the use of various surgical techniques, and low numbers of nonsurgical participants.
Bowman et al published a case series of 17 patients who underwent operative treatment of partial proximal avulsion injuries.28 Outcomes show these patients have satisfactory functional results, however, surgery is best reserved for patients who have exhausted nonoperative management. Surgery comes with its own risks and can include a difficult rehabilitation protocol including splinting or bracing.28,30
There has been increasing interest in the therapeutic benefits of PRP for various soft tissue injuries. In the hamstrings, PRP may provide a reasonable alternative to surgery for partial tears that have not responded to the previously mentioned early conservative measures. Previous studies have reported varying results when using PRP for treatment of tendinosis.2,15,31,32 Although the popularity of PRP for various injuries is increasing, the literature remains controversial due to inconsistency and lack of standardization of the techniques, indications, and even the protocols studied.
Biology and physiology of PRP
The clinical potential of PRP for soft-tissue injury healing stems from its ability to promote chemotaxis and neoangiogenesis. It attempts to mimic the body’s natural healing response. The α-granules of the platelets are responsible for releasing inflammatory and neovascular factors such as tumor-growth factor ß (TGF-ß), platelet derived growth factor (PDGF), insulin-like growth factors 1 and 2 (ILGF), fibroblast growth factor (FGF), epidermal growth factor (EGF), vascular endothelial growth factor (VEGF), and endothelial cell growth factor (ECGF).15,32-38 These factors promote chemotaxis, cell differentiation, and angiogenesis, and are used therapeutically at a concentration that is two to 27 times that of human plasma, depending on the preparation used.
Various authors have suggested that lower concentrations may be below the effective range, while concentrations that are too high may have an inhibitory effect.15,34,39-45 Some authors believe the most biologically effective concentration lies between four and six times that of native plasma, while others believe the effective dose lies around 2.5-fold.45-47 Graziani et al performed an in vitro study of PRP and found that concentrations around fivefold had an inhibitory effect on fibroblast proliferation.45 Regardless of the ideal concentration, PRP has the potential to provide a healing environment that promotes regeneration of collagen and restoration of the fiber orientation, which is disrupted by microtears resulting from the inciting injury.38,48,49
De Mos et al performed a controlled laboratory study looking at the effects of PRP on cultured human tenocytes.50 The PRP effectively increased expression of cell proliferation, matrix degradation enzymes, endogenous growth factors, and collagen production. Although more research is needed, the current belief is that PRP promotes degradation of the damaged area of tendon and then promotes angiogenesis and collagen production to create a fibrovascular clot for healing. More recently, Alsousou et al published a laboratory study looking at in vivo effects of PRP on Achilles tendon ruptures.51 The researchers compared immunohistochemistry six weeks postinjection in individuals who received either a PRP injection or a placebo injection. The PRP group had significantly higher cellularity, glycosaminoglycan content, and type I collagen, which is believed to enhance the maturity of the healing tendon. Further studies are needed to look at long-term differences between groups, as well as the clinical implications of the findings.
Kajikawa et al demonstrated that PRP increases healing potential in tendons of rats.51 Rats expressing green fluorescent protein were used in this laboratory study to look at increased activity of circulation-derived cells that play a role in tendon healing. These circulation-derived cells mainly include the inflammatory and angiogenic mediators mentioned previously. The researchers induced perpendicular injuries to the patellar tendons of the rats and subsequently injected PRP into the wound. Compared with the control group, which received no PRP, the study group had significantly higher expression of circulation-derived cells at days three and seven. Immunologic analysis also showed elevated type I and III collagen in the PRP group compared with the control group, highlighting the increased healing potential in the PRP group.
Clinical benefits
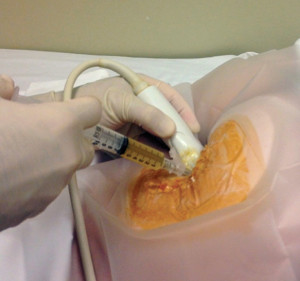
Figure 2. PRP preparation rich in growth factors (PRGF) injected under ultrasound guidance into a partial tear of the right hamstrings origin. (Photo courtesy of Omer Mei-Dan, MD.)
PRP has been used for a wide variety of tendinopathic clinical indications, including lateral epicondylitis, patellar tendinosis, rotator cuff tendinitis, and Achilles tendinitis. Its use has been mainly reserved for conditions that have failed conservative treatment modalities.
In a prospective clinical study, Kon et al injected three consecutive doses of PRP into the patellar tendon of athletes with patellar tendinosis who had failed conservative management.53 After six months, participants reported significant improvements in symptomatic as well as functional scores.
Wetzel and colleagues were the first to publish clinical results on PRP for partial proximal hamstring injuries.53 They retrospectively reviewed 12 injuries that failed conservative treatment modalities, including physical therapy, before undergoing one PRP injection into the hamstring origin. A separate control group of five participants continued with physical therapy alone. Although both groups showed improvements in subjective pain scales (ie, a visual analog scale [VAS] and the Nirschl Phase Rating Scale), only the PRP group had statistically significant results at an average follow-up of 4.5 months (p < .01 for the PRP group and p < .06 for the control group). All 17 participants went on to return to their previous level of competition, which included high-level play in nine cases. Only one participant required a second PRP injection. This study is limited by its small number of participants and retrospective nature, however, it shows promise that PRP is a safe and effective means of treating hamstring injuries that are refractory to conservative management.
Fader et al recently published a study highlighting the effectiveness of ultrasound-guided PRP injections for chronic hamstring tendinopathy in 18 patients who had failed conservative treatment modalities.2 All patients underwent a single ultrasound-guided PRP injection performed by a single radiologist. Six months after injection, there was an 80% or greater VAS improvement in 10 of 18 patients, while overall VAS improvement was 63% for the group. This study highlights the clinical benefits of PRP for hamstring injuries as an easy and alternate nonsurgical treatment in cases that do not respond to conservative measures.
A Hamid et al performed a randomized controlled prospective trial looking at 28 participants with acute (within one week of enrollment) hamstring injuries.55 The experimental group received one PRP injection along with a rehabilitation course, while the control group received rehabilitation alone. The time required to return to sports was significantly shorter in the group who received a PRP injection compared with the control group (p = .02; 26.7 ± 7 days vs 42.5 ± 20.6 days). Furthermore, secondary outcomes showed significantly lower subjectively reported pain scores in the PRP group than the control group throughout the study. This study demonstrated that PRP in conjunction with rehabilitation is a safe treatment option that may be more effective than rehabilitation alone. It also challenges the current practice of using conservative measures alone in the early period after hamstring injury.
These studies focusing on the use of PRP for hamstring injuries indicate that PRP is a safe and effective means of treating hamstring injuries that have persisted despite early conservative measures. PRP can be an efficacious adjuvant to physical therapy and may serve to delay or obviate surgery for partial hamstring tears.
PRP preparations
Different commercial PRP preparations vary in their biologic makeup. There is a great deal of controversy surrounding the optimal consistency and ideal standardized preparation of PRP. Many researchers categorize PRP preparations based on concentration of white blood cells (WBCs) as leukocyte-rich versus leukocyte-poor concentrates. Leukocyte-poor preparations have WBCs intentionally eliminated, as some believe that WBCs will induce an exaggerated inflammatory response, leading to the destruction of healing tissue, and may contribute to a higher pain response postinjection.42,44,56-58 (Figure 1.)
Normal clots that form at soft tissue injuries consist of almost 94% red blood cells, which have limited healing capabilities, and only about 6% platelets. The goal of a PRP injection is to replace the natural clot with one that is highly concentrated in platelets. This highly concentrated clot provides elevated chemokine production and a higher healing potential. Additionally, the leukocyte-poor PRP preparations effectively skip the inflammatory stage of the acute response to injury, since the presence of WBCs in the area is limited. This decreases macrophage migration to the injured area, decreasing soft tissue destruction by the macrophages.42,47,56
PRP injection technique
To accurately inject hamstring pathology, one must navigate the needle deep into thick layers of subcutaneous fat and muscle, which can be difficult. Imaging has been shown to substantially improve the accuracy and results of these therapeutic injections; therefore most PRP injections are now being performed with ultrasound guidance.58,60 (Figure 2.) Ballaudière et al published results of 408 single ultrasound-guided PRP injections for various tendinopathies, showing significantly improved functional scores postinjection.61 This is reinforced by the previously mentioned findings of Fader et al regarding the use of ultrasound-guided PRP injections for hamstring injuries.2
Future directions
We are still in the early stages of understanding the indications and effectiveness of the use of PRP for soft tissue injuries. While PRP may allow elite level athletes to forgo definitive surgery until the end of the season, it may also benefit elderly individuals who desire pain relief and wish to avoid a surgical procedure. Future studies on the use of PRP for the various types of hamstring injuries will need to include larger prospective randomized control trials and to evaluate proper indications, potential candidates, injury patterns, techniques, preparations, volume, location of injection, and regimens including one or multiple injections.
Conclusion
While platelet-rich plasma therapy for hamstring injures shows promising preliminary results, there remains a paucity of literature regarding the specific types of injuries for which it is best suited and which standardized protocol is most effective.
The current understanding that PRP elicits an elevated inflammatory response, thus creating an advantageous environment for healing, has been demonstrated in vitro. Several laboratory analyses have demonstrated a significant response to PRP that involves promoting inflammation and neovascularization as well as degradation of damaged areas of tendon. Few clinical studies have demonstrated the benefit of PRP in various soft tissue injuries in general and hamstring injuries in particular. It is well accepted that the use of ultrasound guidance helps improve the accuracy of such injections and will likely be of benefit in PRP therapy as it evolves. Future research is needed to expand our understanding of the use of PRP as a therapeutic option for hamstring injuries to allow for improved symptomatic relief and earlier return to sport.
Frank B. Wydra, MD is a third-year orthopedic surgery resident at the University of Colorado School of Medicine in Aurora. Ryan R. Fader, MD, is a fifth-year orthopedic surgery resident at the University of Colorado School of Medicine. Omer Mei-Dan, MD, is an associate professor at the University of Colorado School of Medicine and a certified orthopedic surgeon who specializes in hip preservation. Eric C. McCarty, MD, is an associate professor at the University of Colorado School of Medicine and chief of Sports Medicine and Shoulder Surgery for the Department of Orthopedics at the University of Colorado.
1. Woodley SJ, Mercer SR. Hamstring muscles: Architecture and innervation. Cells Tissues Organs 2005;179(3):125-141.
2. Fader RR, Mitchell JJ, Traub S, et al. Platelet-rich plasma treatment improves outcomes for chronic proximal hamstring injuries in an athletic population. Muscles Ligaments Tendons J 2015;4(4):461-466.
3. Ahmad CS, Redler LH, Ciccotti MG, et al. Evaluation and management of hamstring injuries. Am J Sports Med 2013;41(12):2933-2947.
4. Andia I, Maffulli N. Platelet-rich plasma for managing pain and inflammation in osteoarthritis. Nat Rev Rheumatol 2013;9(12):721-730.
5. Clanton TO, Coupe KJ. Hamstring strains in athletes: Diagnosis and treatment. J Am Acad Orthop Surg 1998;6(4):237-248.
6. Koulouris G, Connell D. Evaluation of the hamstring muscle complex following acute injury. Skeletal Radiol 2003;32(10):582-589.
7. Garrett WE Jr, Rich FR, Nikolaou PK, Vogler JB 3rd. Computed tomography of hamstring muscle strains. Med Sci Sports Exerc 1989;21(5):506-514.
8. Pomeranz SJ, Heidt RS Jr. MR imaging in the prognostication of hamstring injury. Work in progress. Radiology 1993;189(3):897-900.
9. Brandser EA, el-Khoury GY, Kathol MH, et al. Hamstring injuries: Radiographic, conventional tomographic, CT, and MR imaging characteristics. Radiology 1995;197(1):257-262.
10. Speer KP, Lohnes J, Garrett WE Jr. Radiographic imaging of muscle strain injury. Am J Sports Med 1993;21(1):89-95.
11. De Smet AA, Best TM. MR imaging of the distribution and location of acute hamstring injuries in athletes. Am J Roentgenol 2000;174(2):393-399.
12. Garrett WE Jr, Califf JC, Bassett FH 3rd. Histochemical correlates of hamstring injuries. Am J Sports Med 1984;12(2):98-103.
13. Sallay PI, Friedman RL, Coogan PG, Garrett WE. Hamstring muscle injuries among water skiers. Functional outcome and prevention. Am J Sports Med 1996;24(2):130-136.
14. Lempainen L, Sarimo J, Mattila K, et al. Proximal hamstring tendinopathy: Results of surgical management and histopathologic findings. Am J Sports Med 2009;37(4):727-734.
15. Andia I, Latorre PM, Gomez MC, et al. Platelet-rich plasma in the conservative treatment of painful tendinopathy: A systematic review and meta-analysis of controlled studies. Br Med Bull 2014;110(1):99-115.
16. Cacchio A, Rompe JD, Furia JP, et al. Shockwave therapy for the treatment of chronic proximal hamstring tendinopathy in professional athletes. Am J Sports Med 2011;39(1):146-153.
17. Valent A, Frizziero A, Bressan S, et al. Insertional tendinopathy of the adductors and rectus abdominis in athletes: A review. Muscles Ligaments Tendons J 2012;2(2):142-148.
18. Rowe V, Hemmings S, Barton C, et al. Conservative management of midportion Achilles tendinopathy: A mixed methods study, integrating systematic review and clinical reasoning. Sports Med 2012;42(11):941-967.
19. Woodley BL, Newsham-West RJ, Baxter GD. Chronic tendinopathy: Effectiveness of eccentric exercise. Br J Sports Med 2007;41(4):188-198.
20. Frizziero A, Trainito S, Oliva F, et al. The role of eccentric exercise in sport injuries rehabilitation. Br Med Bull 2014;110(1):47-75.
21. Silder A, Sherry MA, Sanfilippo J, et al. Clinical and morphological changes following 2 rehabilitation programs for acute hamstring strain injuries: A randomized clinical trial. J Orthop Sports Phys Ther 2013;43(5):284-299.
22. Mason DL, Dickens VA, Vail A. Rehabilitation for hamstring injuries. Cochrane Database Syst Rev 2012;12:CD004575.
23. Malliaropoulos N, Papalexandris S, Papalada A, Papacostas E. The role of stretching in rehabilitation of hamstring injuries: 80 athletes follow-up. Med Sci Sports Exerc 2004;36(5):756-759.
24. Sherry MA, Best TM. A comparison of 2 rehabilitation programs in the treatment of acute hamstring strains. J Orthop Sports Phys Ther 2004;34(3):116-125.
25. Askling CM, Tengvar M, Tarassova O, Thorstensson A. Acute hamstring injuries in Swedish elite sprinters and jumpers: A prospective randomised controlled clinical trial comparing two rehabilitation protocols. Br J Sports Med 2014;48(7):532-539.
26. Rust DA, Giveans MR, Stone RM, et al. Functional outcomes and return to sports after acute repair, chronic repair, and allograft reconstruction for proximal hamstring ruptures. Am J Sports Med 2014;42(6):1377-1383.
27. Cohen SB, Rangavajjula A, Vyas D, Bradley JP. Functional results and outcomes after repair of proximal hamstring avulsions. Am J Sports Med 2012;40(9):2092-2098.
28. Bowman KF Jr, Cohen SB, Bradley JP. Operative management of partial-thickness tears of the proximal hamstring muscles in athletes. Am J Sports Med 2013;41(6):1363-1371.
29. Harris JD, Griesser MJ, Best TM, Ellis TJ. Treatment of proximal hamstring ruptures – a systematic review. Int J Sports Med 2011;32(7):490-495.
30. Chakravarthy J, Ramisetty N, Pimpalnerkar A, Mohtadi N. Surgical repair of complete proximal hamstring tendon ruptures in water skiers and bull riders: A report of four cases and review of the literature. Br J Sports Med 2005;39(8):569-572.
31. Mei-Dan O, Carmont MR. The role of platelet-rich plasma in rotator cuff repair. Sports Med Arthrosc 2011;19(3):244-250.
32. Mei-Dan O, Lippi G, Sanchez M, et al. Autologous platelet-rich plasma: A revolution in soft tissue sports injury management? Phys Sportsmed 2010;38(4):127-135.
33. Abate M, Schiavone C, Salini V, Andia I. Occurrence of tendon pathologies in metabolic disorders. Rheumatology 2013;52(4):599-608.
34. Engebretsen L, Steffen K, Alsousou J, et al. IOC consensus paper on the use of platelet-rich plasma in sports medicine. Br J Sports Med 2010;44(15):1072-1081.
35. Yeaman MR. The role of platelets in antimicrobial host defense. Clin Infect Dis 1997;25(5):951-968.
36. Dragoo JL, Braun HJ, Durham JL, et al. Comparison of the acute inflammatory response of two commercial platelet-rich plasma systems in healthy rabbit tendons. Am J Sports Med 2012;40(6):1274-1281.
37. Zhang J, Middleton KK, Fu FH, et al. HGF mediates the anti-inflammatory effects of PRP on injured tendons. PLoS One 2013;8(6):e67303.
38. Fredberg U, Stengaard-Pedersen K. Chronic tendinopathy tissue pathology, pain mechanisms, and etiology with a special focus on inflammation. Scand J Med Sci Sports 2008;18(1):3-15.
39. Abate M, Silbernagel KG, Siljeholm C, et al. Pathogenesis of tendinopathies: Inflammation or degeneration? Arthritis Res Ther 2009;11(3):235.
40. Andia I, Sanchez M, Maffulli N. Tendon healing and platelet-rich plasma therapies. Expert Opin Biol Ther 2010;10(10):1415-1426.
41. Dohan Ehrenfest DM, Rasmusson L, Albrektsson T. Classification of platelet concentrates: From pure platelet-rich plasma (P-PRP) to leucocyte- and platelet-rich fibrin (L-PRF). Trends Biotechnol 2009;27(3):158-167.
42. Sanchez M, Anitua E, Orive G, et al. Platelet-rich therapies in the treatment of orthopaedic sport injuries. Sports Med 2009;39(5):345-354.
43. Mazzucco L, Balbo V, Cattana E, et al. Not every PRP-gel is born equal. Evaluation of growth factor availability for tissues through four PRP-gel preparations: Fibrinet, RegenPRP-kit, Plateltex and one manual procedure. Vox Sang 2009;97(2):110-118.
44. Anitua E, Sanchez M, Nurden AT, et al. New insights into and novel applications for platelet-rich fibrin therapies. Trends Biotechnol 2006;24(5):227-234.
45. Graziani F, Ivanovski S, Cei S, et al. The in vitro effect of different PRP concentrations on osteoblasts and fibroblasts. Clin Oral Implants Res 2006;17(2):212-219.
46. Weibrich G, Hansen T, Kleis W, et al. Effect of platelet concentration in platelet-rich plasma on peri-implant bone regeneration. Bone 2004;34(4):665-671.
47. Marx RE. Platelet-rich plasma: Evidence to support its use. J Oral Maxillofac Surg 2004;62(4):489-496.
48. Anitua E, Sanchez M, Nurden AT, et al. Reciprocal actions of platelet-secreted TGF-beta1 on the production of VEGF and HGF by human tendon cells. Plast Reconstr Surg 2007;119(3):950-959.
49. Anitua E, Sanchez M, Nurden AT, et al. Autologous fibrin matrices: A potential source of biological mediators that modulate tendon cell activities. J Biomed Mater Res A 2006;77(2):285-293.
50. de Mos M, van der Windt AE, Jahr H, et al. Can platelet-rich plasma enhance tendon repair? A cell culture study. Am J Sports Med 2008;36(6):1171-1178.
51. Alsousou J, Thompson M, Harrison P, et al. Effect of platelet-rich plasma on healing tissues in acute ruptured Achilles tendon: A human immunohistochemistry study. Lancet 2015;385(Suppl 1):S19.
52. Kajikawa Y, Morihara T, Sakamoto H, et al. Platelet-rich plasma enhances the initial mobilization of circulation-derived cells for tendon healing. J Cell Physiol 2008;215(3):837-845.
53. Kon E, Filardo G, Delcogliano M, et al. Platelet-rich plasma: New clinical application: A pilot study for treatment of jumper’s knee. Injury 2009;40(6):598-603.
54. Wetzel RJ, Patel RM, Terry MA. Platelet-rich plasma as an effective treatment for proximal hamstring injuries. Orthopedics 2013;36(1):e64-e70.
55. A Hamid MS, Mohamed Ali MR, Yusof A, et al. Platelet-rich plasma injections for the treatment of hamstring injuries: A randomized controlled trial. Am J Sports Med 2014;42(10):2410-2418.
56. Mei-Dan O, Mann G, Maffulli N. Platelet-rich plasma: Any substance into it? Br J Sports Med 2010;44(9):618-619.
57. Tidball JG. Inflammatory processes in muscle injury and repair. Am J Physiol Regul Integr Comp Physiol 2005;288(2):R345-R353.
58. Asfaha S, Cenac N, Houle S, et al. Protease-activated receptor-4: A novel mechanism of inflammatory pain modulation. Br J Pharmacol 2007;150(2):176-185.
59. Finnoff JT, Hurdle MF, Smith J. Accuracy of ultrasound-guided versus fluoroscopically guided contrast-controlled piriformis injections: A cadaveric study. J Ultrasound Med 2008;27(8):1157-1163.
60. Zissen MH, Wallace G, Stevens KJ, et al. High hamstring tendinopathy: MRI and ultrasound imaging and therapeutic efficacy of percutaneous corticosteroid injection. Am J Roentgenol 2010;195(4):993-998.
61. Dallaudiere B, Pesquer L, Meyer P, et al. Intratendinous injection of platelet-rich plasma under US guidance to treat tendinopathy: A long-term pilot study. J Vasc Interv Radiol 2014;25(5):717-723.







