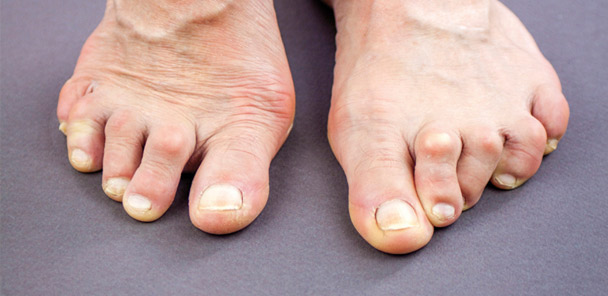
Research has identified ankle-brachial index, loss of sensation, and deformity as predictors of foot ulceration in patients with rheumatoid arthritis, and also suggests that ulceration influences health-related quality of life across physical, social, and psychological domains.
Research has identified ankle-brachial index, loss of sensation, and deformity as predictors of foot ulceration in patients with rheumatoid arthritis, and also suggests that ulceration influences health-related quality of life across physical, social, and psychological domains.
By Jill Firth, PhD, BSc (Hons), and Heidi J Siddle, PhD, MSc, BSc (Hons)
Historically, researchers have studied lower extremity ulceration occurring in patients with rheumatoid arthritis (RA) as a single entity. This overlooks the fact that wounds on the leg and foot may have differences in prevalence, underlying pathophysiology, and impact on quality of life and risk factors. Although there are clear clinical pathways for patients with diabetic foot ulceration that include strategies to prevent recurrence, healthcare provision for patients with RA experiencing foot ulceration is less established.
Over the past decade, researchers have focused attention on addressing gaps in our knowledge base specifically relating to foot ulceration occurring in RA: the size of the problem; the impact on quality of life; experiences of healthcare provision; and the identification of risk factors for recurrence.
Rheumatoid arthritis
RA is a systemic inflammatory disease that predominantly affects the peripheral synovial joints. Research reports that foot involvement occurs in up to 94% of patients with RA during the course of their disease.1-3 The importance of foot pathology in RA has been recognized in rheumatology practice, and the current evidence highlights the burden of painful foot disease on physical function and quality of life.4-6 Despite the introduction of more aggressive therapies such as tumor necrosis factor (TNF) inhibitors, the prevalence of foot pain and foot joint involvement does not appear to be diminishing.1,7 Established risk factors for diabetic foot ulceration, such as peripheral vascular disease, neuropathy, raised plantar pressures, foot deformity, and ill-fitting footwear,8 also contribute to ulceration to a greater or lesser extent in patients with RA as systemic and local manifestations of disease.
The overall prevalence of foot ulceration in RA as a discrete entity is estimated to be between 10% and 13% in the UK and the Netherlands.9,10 In the UK, these findings are based on a postal survey of 1130 patients in a secondary care setting that achieved a 78% response rate. Following clinical examination, the validated point prevalence of foot ulceration was 3.39% and the overall prevalence was 9.73%.9 The most common sites of ulceration in the foot were the dorsal aspect of hammer toes (48%), the metatarsal heads (32%), and the medial aspect of the first metatarsophalangeal joint in patients with hallux abducto valgus (20%). The survey highlighted the recurrent nature of the problem; 47% of patients reported a history of multiple episodes of ulceration ranging from two to 30 episodes (median 2; interquartile range 1-4).
In the cross-sectional multicenter study conducted by Matricali et al in the Netherlands,10 285 consecutive patients with RA were assessed for the presence of forefoot problems. The point prevalence of foot ulceration was 5% and overall prevalence 13%, with 4% of patients experiencing multiple wound problems at the time of data collection.10
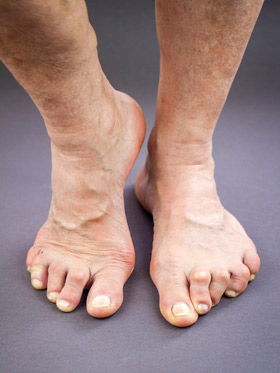 A recent retrospective US population-based study of lower extremity ulceration in RA included 813 patients with 9771 total person-years of follow-up.11 One hundred twenty five patients developed lower extremity ulceration at a rate of 1.8 episodes per 100 person-years (95% CI 1.5, 2 per 100 person-years). The cumulative incidence increased by 1% a year and the rate of ulcer recurrence was substantially increased to 7.9 per 100 person-years (95% CI 5.5, 10.9). This equates to an overall prevalence of 15%, with an estimated 66% of ulcers occurring on or below the malleolus and occurring on the toes in 27% and plantar metatarsals in 11%. Risk of ulceration was associated with advancing age, rheumatoid factor positivity, presence of rheumatoid nodules, and venous thromboembolism. The etiology of ulceration was attributed to pressure in 62 cases (36%), ischemia in 22 (18%), venous disease in 20 (12%), neuropathy in 18 (11%), and vasculitis in two (1%).
A recent retrospective US population-based study of lower extremity ulceration in RA included 813 patients with 9771 total person-years of follow-up.11 One hundred twenty five patients developed lower extremity ulceration at a rate of 1.8 episodes per 100 person-years (95% CI 1.5, 2 per 100 person-years). The cumulative incidence increased by 1% a year and the rate of ulcer recurrence was substantially increased to 7.9 per 100 person-years (95% CI 5.5, 10.9). This equates to an overall prevalence of 15%, with an estimated 66% of ulcers occurring on or below the malleolus and occurring on the toes in 27% and plantar metatarsals in 11%. Risk of ulceration was associated with advancing age, rheumatoid factor positivity, presence of rheumatoid nodules, and venous thromboembolism. The etiology of ulceration was attributed to pressure in 62 cases (36%), ischemia in 22 (18%), venous disease in 20 (12%), neuropathy in 18 (11%), and vasculitis in two (1%).
The etiology of lower limb ulceration is poorly understood, and one cannot assume the risk factors for leg and foot ulceration are the same. Historically, lower extremity wounds in RA have been found to be multifactorial,12,13 and the prevalence of vasculitis has perhaps been overemphasized. To determine the predictors of foot ulceration in patients with RA unaffected by diabetes, we recently undertook a multicenter case control study14 of 83 cases with 112 current ulcers and 190 ulcer-naive controls.
Univariate analysis demonstrated that risk of ulceration increases with loss of sensation, abnormality of ankle-brachial pressure index (ABPI), and foot deformity. Plantar pressures and RA disease activity (as measured by swollen joint counts) were not significant predictors of ulceration. In logistic regression modeling, ABPI (odds ratio [OR] = .04; 95% CI .01-.28), forefoot deformity (OR = 1.14; 95% CI 1.08-1.21), and loss of sensation (OR 1.22; 95% CI 1.10-1.36) predicted risk but increased plantar pressures did not. Perhaps surprisingly, these findings were unchanged in a subgroup analysis of the 33 patients affected by plantar ulceration.
Vasculitis has often been cited as a factor in lower extremity wounds in patients with RA, but the evidence for this is scarce. In our study, only three cases and two controls presented with suspected cutaneous vasculitis.14 Similarly, a review of biopsy specimens from the margins of lower extremity wounds in 12 patients revealed evidence of vasculitis in only three.15 In fact, vasculitis is estimated to affect only around 3.6% of RA patients at any given time, so it is not common.16 Vasculitic ulcers have characteristics that are distinct from those of other wounds and tend to appear and enlarge rapidly. Vasculitis should be suspected when an ulcer has inflamed wound margins or a punched-out appearance, or when there are other signs of cutaneous vasculitis present, such as nail fold infarcts or purpuric rash.
Qualitative data also point to the role of friction from footwear at the interface with foot deformity in the onset of ulceration, which was described in interviews by around half the participants in a study of 23 nondiabetic adults with RA and open foot ulceration.17
It is possible that there may be different causal pathways within this patient population. Recent cluster analysis of causal pathways in diabetic foot ulceration identified four dominant clusters from previous research: neuropathy, deformity, callus, and raised plantar pressures; peripheral vascular disease; penetrating trauma; and ill-fitting shoes.18 Within the rheumatoid population there is also likely to be diversity, but there is as of yet no research to elucidate this further. Unfortunately a lack of clear care pathways has precluded a multicenter prospective cohort study of lower extremity ulceration in a population of RA patients.
The impact on quality of life
While the impact of lower extremity wounds on quality of life has been extensively studied in the diabetic population and in patients with leg ulceration, until recently researchers did not have any insight into the added impact of foot ulceration in patients already coping with RA. Qualitative interviews with 23 nondiabetic adults with RA and open foot ulceration revealed that ulceration influenced health-related quality of life across physical, social, and psychological domains.17 Participants felt ulcer pain was distinct from foot pain associated with RA. The pain created new walking disability, which added to existing limitations in undertaking household tasks and personal care independently. Keeping the ulcer dry caused problems with personal hygiene and new restrictions in leisure activities, leading to reduced social participation. Increased footwear and clothing restrictions affected body image, and female participants described loss of self-esteem and issues with confidence. Low mood, anxiety, and frustration were consequences of these physical and social impacts of ulceration. The duration of ulceration, progress toward healing, and potential for recurrence all had an impact on psychological status.
The findings of these interviews also highlighted the need for clearer pathways for RA patients affected by foot ulceration.19 Marked variations in access to care in the UK were influenced by past experience of ulceration and knowledge of the healthcare system gained from experience of living with RA. A number of participants expressed uncertainty about which practitioner to see, and how and when to seek help. Once a patient had experience in the healthcare system, knowledge acquisition appeared to lead to increased trust in health professionals, self-efficacy, and changes in health behavior. In contrast, anxiety and frustration arose when education needs were not met and uncertainty about progress towards healing existed. Specialist care delivered in the context of a therapeutic patient-professional relationship was highly valued by participants; when multiple healthcare providers were involved, care pathways were less clear and continuity of care was compromised.
Although these broad themes may be relevant internationally, the inclusion of patient perspectives in the design and evaluation of foot health and wound care services has the potential to improve the patient experience.
Broader clinical implications
Patients with RA and persistent foot and ankle symptoms, including ulceration, frequently seek help from multiple health professionals on multiple occasions, often with limited success. Despite the growing body of research to support individual treatment modalities such as methods for offloading the foot and debriding callus overlying pressure-vulnerable sites,20,21 together with the introduction of local and national guidelines for people with RA,22 there remains no clear evidence-based clinical care pathway for identifying patients at risk of ulceration and subsequently managing the wound site. This is exacerbated by a lack of podiatry support in most rheumatology units in the UK.23
Deformity and prominent joints in patients with RA are subject to excessive shear and compressive stresses during gait, frequently resulting in the build up of painful callosities, particularly over the plantar aspect of the metatarsal heads,24 which is a potential site of ulceration. The rationale and therapeutic benefit of callus reduction to reduce peak pressures, prevent ulceration, and facilitate wound healing is firmly established in patients with diabetes.25,26 However, sharp scalpel debridement of painful forefoot plantar callosities in people with RA has repeatedly produced no improvement in symptoms compared with not debriding calluses.21,27 On this basis, the authors recommend that sharp scalpel debridement in patients with RA should be reserved for the reduction of high-risk states or short-term alleviation of severe pain.21 Further work is required to determine whether this procedure is beneficial to people with RA who are specifically at risk for or experiencing ulceration, with particular focus required on the impact of scalpel debridement on wound healing and alleviation of symptoms.
Peripheral vascular disease and neuropathy have been shown to increase risk of ulceration by virtue of impaired tissue viability and loss of protective sensation, but screening is not routine in all foot health assessments for patients with RA, unlike patients with diabetes. Evidence of increased prevalence of peripheral neuropathy and peripheral vascular disease in RA is based on an emerging body of research28-30 that will need to be integrated into future training and education opportunities for podiatrists and nurses working in foot health and wound care.
Routine assessment of foot health should include vascular and neurological evaluation to identify problems early and reduce the threat to tissue viability. Patient education needs to include teaching the importance of reporting changes in sensation and new symptoms affecting the lower limbs.
Medication to manage the underlying inflammatory disease, such as steroids or biologic therapies, are known to be associated with increased risk of skin and soft tissue infection in patients with RA.31 Open wounds, in turn, present a relative contraindication to biologic treatment and may require suspending a patient’s medication, which, in turn, may increase the time required for the wound to heal15 or predispose the patient to subsequent ulceration and increased pain from the inflammation—or, indeed, a deterioration of the ulceration.
Increased foot pain is associated with ulceration,21 and qualitative research indicates wound pain is a major feature of ulceration that can lead to a cascade of consequences.17 The nature of pain arising from ulceration is distinct from arthritic pain and warrants individual assessment and tailored intervention.
Analgesic needs may be altered by the onset of ulceration and during the course of healing, but the strategies may include debridement; the choice of dressing; offloading strategies; and, in some cases, surgery. Clinical experience indicates that polyurethane foam dressings are the most suitable choice, providing a soft silicone layer that doesn’t stick to the wound surface or traumatize the surrounding skin and will cushion prominent bony areas without adding pressure from the interface with the footwear.
Although increased plantar pressure has not been identified as a risk factor for ulceration in RA,14 the benefits of offloading an active ulceration site on healing and pain have not been evaluated. Various methods of offloading painful sites are available, including orthoses, rocker bottom footwear, pressure relief walkers, and pneumatic walkers. However, patients with RA may find the walkers cumbersome and heavy due to muscle weakness and problematic proximal joints and difficult to put on due to reduced manual dexterity. Surgery is very rarely needed to manage the wound itself, however, if the wound occurs over a prominent bony site or abnormal weight-bearing area, it may be advisable to consider surgical intervention to aid healing or prevent recurrence.
Further work should address the development of practical criteria for identifying patients at risk of ulceration so that strategies to reduce that risk may be implemented. For those affected, attention needs to be paid to addressing information needs and access to care pathways that optimize opportunities to deliver best practice in the field of lower extremity ulceration management.
Jill Firth, PhD, BSc (Hons), is a consultant nurse in rheumatology at the Pennine Musculoskeletal Partnership and an honorary senior research fellow at the University of Leeds in the UK. Heidi Siddle, PhD, MSc, BSc (Hons), is the specialist rheumatology podiatrist at the Leeds Teaching Hospitals NHS Trust and senior research fellow in the Leeds Institute of Rheumatic and Musculoskeletal Medicine at the University of Leeds.
1. Grondal L, Tengstrand B, Nordmark B, et al. The foot: still the most important reason for walking incapacity in rheumatoid arthritis: distribution of symptomatic joints in 1,000 RA patients. Acta Orthop 2008;79(2):257-261.
2. Otter SJ, Lucas K, Springett K, et al. Foot pain in rheumatoid arthritis prevalence, risk factors and management: an epidemiological study. Clin Rheumatol 2010;29(3):255-271.
3. van der Leeden M, Steultjens MP, Ursum J, et al. Prevalence and course of forefoot impairments and walking disability in the first eight years of rheumatoid arthritis. Arthritis Rheum 2008;59(11):1596-1602.
4. Helliwell P, Reay N, Gilworth G, et al. Development of a foot impact scale for rheumatoid arthritis. Arthritis Rheum 2005;53(3):418-422.
5. Turner DE, Helliwell PS, Emery P, Woodburn J. The impact of rheumatoid arthritis on foot function in the early stages of disease: a clinical case series. BMC Musculoskelet Disord 2006;7:102.
6. Williams AE, Nester CJ, Ravey MI. Rheumatoid arthritis patients’ experiences of wearing therapeutic footwear – a qualitative investigation. BMC Musculoskelet Disord 2007;8:104.
7. Otter SJ, Lucas K, Springett K, et al. Comparison of foot pain and foot care among rheumatoid arthritis patients taking and not taking anti-TNF alpha therapy: an epidemiological study. Rheumatol Int 2011;31(11):1515-1519.
8. Jeffcoate WJ, Harding KG. Diabetic foot ulcers. Lancet 2003;361(9368):1545-1551.
9. Firth J, Hale C, Helliwell PS, et al. The prevalence of foot ulceration in patients with rheumatoid arthritis. Arthritis Care Res 2008;59(2):200-205.
10. Matricali GA, Boonen A, Verduyckt J, et al. The presence of forefoot problems and the role of surgery in patients with rheumatoid arthritis. Ann Rheum Dis 2006;65(9):1254-1255.
11. Jebakumar AJ, Udayakumar PD, Crowson CS, et al. Occurrence and effect of lower extremity ulcer in rheumatoid arthritis –a population-based study. J Rheumatol 2014;41(3):437-443.
12 McRorie ER, Jobanputra P, Ruckley CV, Nuki G. Leg ulceration in rheumatoid arthritis. Br J Rheumatol 1994;33(11):1078-1084.
13. Pun YL, Barraclough DR, Muirden KD. Leg ulcers in rheumatoid arthritis. Med J Aust 1990;153(10):585-587.
14. Firth J, Waxman R, Law G, et al. The predictors of foot ulceration in patients with rheumatoid arthritis. Clin Rheumatol 2014;33(5):615-621.
15. Shanmugam VK, Demaria DM, Attinger CE. Lower extremity ulcers in rheumatoid arthritis: features and response to immunosuppression. Clin Rheumatol 2011;30(6):849-853.
16. Turesson C, O’Fallon WM, Crowson CS, et al. Extra-articular manifestations in rheumatoid arthritis: incidence trends and risk factors over 46 years. Ann Rheum Dis 2003;62(8):722-727.
17. Firth J, Nelson EA, Briggs M, Gorecki C. A qualitative study to explore the impact of foot ulceration on health-related quality of life in patients with rheumatoid arthritis. Int J Nurs Stud 2011;48(11):1401-1408.
18. Lavery LA, Peters EJ, Armstrong DG. What are the most effective interventions in preventing diabetic foot ulcers? Int Wound J 2008;5(3):425-433.
19. Firth J, Nelson EA, Briggs M, Gorecki C. Experiences of healthcare provision for foot ulceration occurring in people with rheumatoid arthritis. Musculoskelet Care 2013;11(3):159-167.
20. Hennessy K, Woodburn J, Steultjens MP. Custom foot orthoses for rheumatoid arthritis: A systematic review. Arthritis Care Res2012;64(3):311-20.
21. Siddle HJ, Redmond AC, Waxman R, et al. Debridement of painful forefoot plantar callosities in rheumatoid arthritis: the CARROT randomised controlled trial. Clin Rheumatol 2013;32(5):567-574.
22. Podiatric Rheumatic Care Association. Standards of Care for People with Musculoskeletal Foot Health Problems. http://www.prcassoc.org.uk/file/Full%20Colour%20Foot%20Health%20Standards.pdf. Published April 22, 2008. Accessed June 30, 2014.
23. Redmond AC, Waxman R, Helliwell PS. Provision of foot health services in rheumatology in the UK. Rheumatology 2006;45(5):571-576.
24. Woodburn J, Helliwell PS. Relation between heel position and the distribution of forefoot plantar pressures and skin callosities in rheumatoid arthritis. Ann Rheum Dis 1996;55(11):806-810.
25. Young MJ, Cavanagh PR, Thomas G, et al. The effect of callus removal on dynamic plantar foot pressures in diabetic patients. Diabet Med 1992;9(1):55-57.
26. Murray HJ, Young MJ, Hollis S, Boulton AJ. The association between callus formation, high pressures and neuropathy in diabetic foot ulceration. Diabet Med 1996;13(11):979-982.
27. Davys HJ, Turner DE, Helliwell PS, et al. Debridement of plantar callosities in rheumatoid arthritis: a randomized controlled trial. Rheumatology 2005;44(2):207-210.
28. Maradit-Kremers H, Nicola PJ, Crowson CS, et al. Cardiovascular death in rheumatoid arthritis. Arthritis Rheum 2005;52(3):722-732.
29. Agarwal VV, Singh RR, Wiclaf, et al. A clinical, electrophysiological, and pathological study of neuropathy in rheumatoid arthritis. Clin Rheumatol 2008;27(7):841-844.
30. Wilson O, Kirwan JR. Measuring sensation in the feet of patients with rheumatoid arthritis. Musculoskelet Care 2006;4(1):12-23.
31. Dixon WG, Watson K, Lunt M, et al. Rates of serious infection, including site-specific and bacterial intracellular infection, in rheumatoid arthritis patients receiving anti-tumor necrosis factor therapy: results from the British Society for Rheumatology Biologics Register. Arthritis Rheum 2006;54(8):2368-2376.