
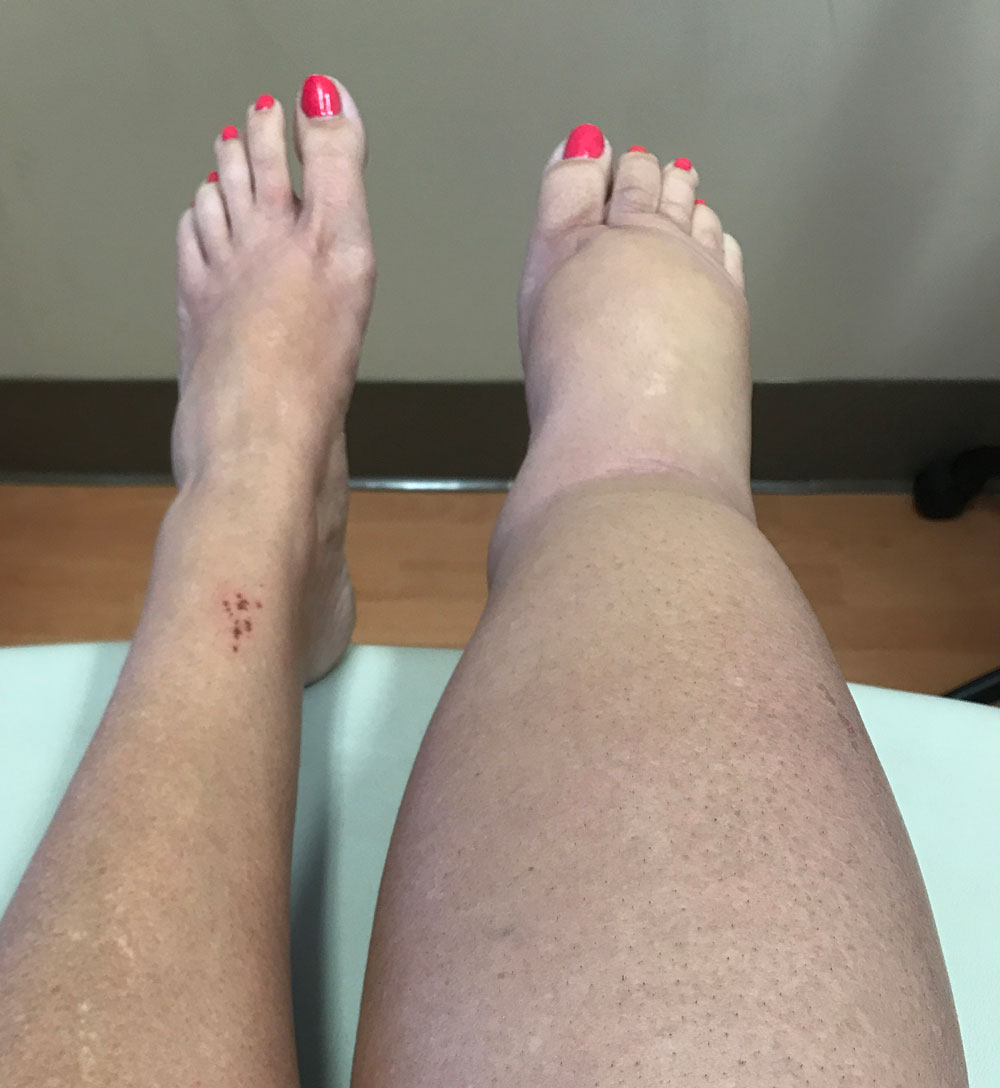
Figure 1. Lymphedema of the right leg. All clinical photos in this article are courtesy of the author.
Often overlooked, lymphedema of the lower extremities is becoming more common as aging, obesity, and cancer therapies all take their toll. Detection is key to getting appropriate treatment.
By Stanley G. Rockson, MD
Lymphedema of the lower limbs is a common, complex, and highly treatable disease that begs recognition by healthcare providers. This debilitating disorder has profound psychological, social, functional, and health implications for the affected individual and it has been far too frequently neglected or overlooked within the healthcare environment. If we acknowledge that the predisposing factors to lymphedema include, most commonly, cancer, chronic vein disease, obesity, and infection, it is readily comprehensible that lymphedema is a disease that is very common and that its prevalence is likely to increase significantly over the next decade.
Before considering the clinical attributes of lower limb lymphedema, it is appropriate to define terms. Lymphedema is a pathology that presents whenever the lymphatic vascular system is intrinsically abnormal in its development or function. This form of the disease, called primary lymphedema, can appear throughout life, although 25% of the cases appear at birth or shortly thereafter. Primary lymphedema, ascribable to inborn errors of structure or function, represents the smallest segment (<1%) of the lymphedema community. While relatively uncommon, primary lymphedema is a very important problem, both because of the genetic implications, which warrant direct evaluation, and because of the profound impact of lifelong disease that often arises in infancy or young adulthood.
Much more prevalent than the primary lymphedemas are those that arise when a previously healthy lymphatic circulation is secondarily damaged by disease, infections, trauma, or medical interventions. In most of these circumstances, the damage is structural, but chronic venous disease and other systemic medical conditions can lead to relative lymphatic failure when, with time and the persistent requirement for greatly enhanced lymphatic flow, the capacity of the system is overwhelmed, and the pathology of lymphedema supervenes.
Consequence of Cancer
Lymphedema as a consequence of cancer and cancer treatment represents the most common form of lower limb lymphedema that is seen today in the United States and the developed world. For many cancers of the skin and of the genitourinary system, among others, staging and treatment require the surgical excision of large numbers of lymph nodes. This iatrogenic lymphatic injury, often accompanied by radiation damage to lymph node–bearing regions, can lead to the relative failure of the system to maintain fluid balance in the tissues, and lymphedema ensues. The incidence of cancer-associated lower limb lymphedema varies by the specific tumor type but, in general, 20-50% of survivors of the relevant cancers will subsequently manifest the problem of lymphedema.
Given the predominant role of cancer-associated lower extremity lymphedema, it is instructive to examine this subtype to best understand the problem in general.
Secondary lymphedema of the legs is most often ascribed to surgical excision of inguino-femoral lymph nodes. Radiotherapy to the respective node-bearing areas can be causal by itself, but most often plays an adjunctive role to the surgical trauma. The disease will present as progressive swelling of one leg or both, most typically within the first 12 months after the treatment for cancer. Once it develops, it is often progressive and rarely, if ever, reverts to normal.
Published incidence estimates vary substantially, depending on the type of cancer, the stage of the cancer, the specific treatment protocol, and the sensitivity of the measurement tool used to detect edema. The highest relative incidence has been observed for vulvar cancer and the lowest after treatment for prostate cancer. Lymphedema incidence increases most predictably when there is adjunctive radiotherapy, where the highest rates are seen.
In most cases of secondary limb lymphedema, the swelling of the limb does not appear immediately at the time of lymphatic trauma. In the context of cancer, the reported timing of lymphedema onset varies substantially. In one published case series, 75% of the lymphedema cases appeared within the first post-treatment year, 19% in the following year, and 6% between 2 and 5 years after diagnosis. Most commonly, in practice, it can be anticipated that the timing of the onset will be within one year of the causal treatment intervention. The patient’s initial complaint is usually painless swelling of one or both of the lower limbs. The patient may also describe the sensation of heaviness in the limb, especially at the end of the day and in hot weather. For affected female patients, symptoms can vary throughout the menstrual cycle.
Detection/Evaluation
Unfortunately, lymphedema is often not detected or evaluated at its earliest and most treatable stage. The first stage of lymphedema may be quite transient, but it is characterized, as is any edematous condition, by the presence of pitting, which describes the ability of the examiner to temporarily displace the excess tissue fluid through pressure applied by the examining finger, thereby creating a fleeting, visible indentation in the surface of the skin. However, the natural history of lymphedema, and one of its unique attributes within the category of edematous disorders, is the tendency to inexorably progress to a non-pitting condition. Ultimately, it is this relatively non-pitting edema that is considered to be a distinguishing hallmark of lymphedema (Figure 1).
In lymphedema, the distribution of the edema is asymmetrical. A positive Stemmer sign (the examiner’s inability to tent the skin at the base of the second toe) is a useful, but not invariate, clinical finding that, when present, is considered to be pathognomic for lymphedema. The edema can spread either proximally or distally along the lower limbs in the early stages, but after the first year, while the magnitude of the edema can continue to increase, the extent of involvement along the axis of the limb uncommonly changes after the first year.
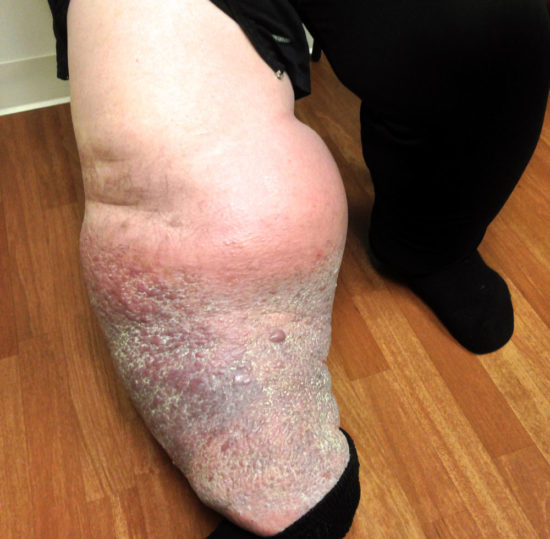
The failure to address lymphedema with appropriate treatment increases the likelihood that the disease will progress in severity. The end organ damage in uncontrolled lymphedema is sustained by the skin and its supporting structures. In such cases, with the passage of time, the lymphedematous leg accumulates increasing pathological changes in the skin. These lymphedematous changes can be quite dramatic, with accentuated skin creases, progressive loss of skin elasticity, hyperkeratosis, lichenification, and papillomatosis (Figure 2). Symptomatically, nearly all patients describe the presence of limb heaviness, fullness, or chronic dull pain. Additional subjective features of lower limb lymphedema include tightness of the shoes or frank inability to wear footwear; loss of hair on the affected limb; burning or itching of the legs, feet, or toes; and sleep disturbances. It is interesting to note that the published medical literature supports the observation that patients with lower extremity lymphedema are more likely to present with swelling, heaviness, tightness, and skin alterations than those who experience problems with upper limb lymphedema.
In severe cases, skin breakdown does occur, as does lymphorrhea (exudation of tissue fluid through the skin surface). These latter conditions predispose the patient to an increased risk of soft-tissue infection. Indeed, even in the absence of lymphorrhea or skin breakdown, recurrent infection, cellulitis, and lymphangitis are exceedingly common in lower limb lymphedema; more than one-half of these patients experience at least one episode of cellulitis, and perhaps one-third of the patients experience repeated episodes of infection. Repeated infection can further damage the lymphatic structures and thereby reduce lymphatic function, creating a vicious cycle of infection and sequential worsening of limb health.
Clinical Diagnosis/Differential Diagnosis
The clinical diagnosis of lower limb lymphedema rests upon observations that can be made in the examining room. The documentation and quantitation of edema is based upon measures of limb circumference and/or volume. When only one leg is swollen, concurrent measurement of the uninvolved leg can be used to assess the extent of swelling in the affected limb. However, the disease often affects both lower limbs. In addition, in some patients, pre-existing asymmetry in the two lower limbs may hamper the ability to accurately compare the limbs for the assessment of edema presence and magnitude. Circumferential measurements of the limbs can be unreliable, but if performed methodically along the axis of the leg, the data can be used to accurately calculate limb volume using established geometric formulas. Water displacement volumetry, although not commonly used, directly measures limb volume. This method is cumbersome but accurate. Surveillance of at-risk patient groups is increasingly performed with bioimpedance spectroscopy, a non-invasive technique that can detect subtle degrees of asymmetry in extracellular fluid volume before edema becomes clinically manifest.
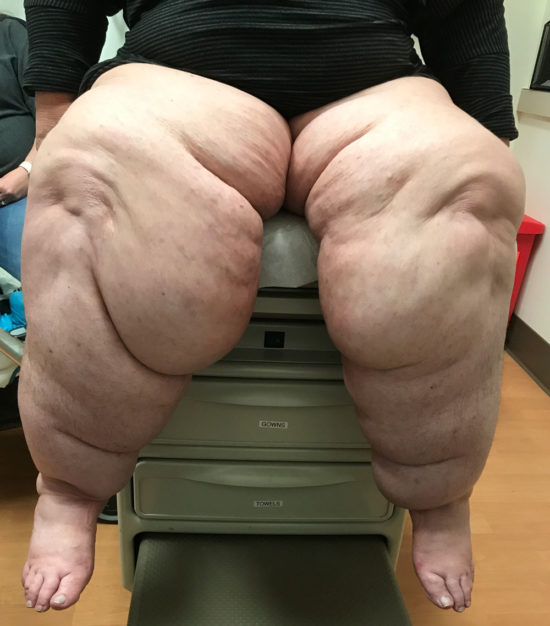
As in all medical presentations, there is a differential diagnosis for lower limb lymphedema. Chief among these is the recognition of edema due to venous pathologies. As already mentioned, the presence of chronic venous edema can lead to functional, secondary lymphatic failure. Therefore, combined lymphaticovenous edema can commonly be encountered. A rigorous search for evidence of venous hypertension (Figure 3), including such findings as superficial varicosities, dependent rubor, hemosiderin deposits, and lipodermatosclerosis can assist the clinician in identifying components of the presentation that are amenable to correction or amelioration. While reversal of venous hypertension will not eradicate concomitant manifestations of established secondary lymphedema, such strategies can certainly mitigate the extent of disease, limit disease progression, and render the lymphedema more responsive to the appropriate physical interventions.
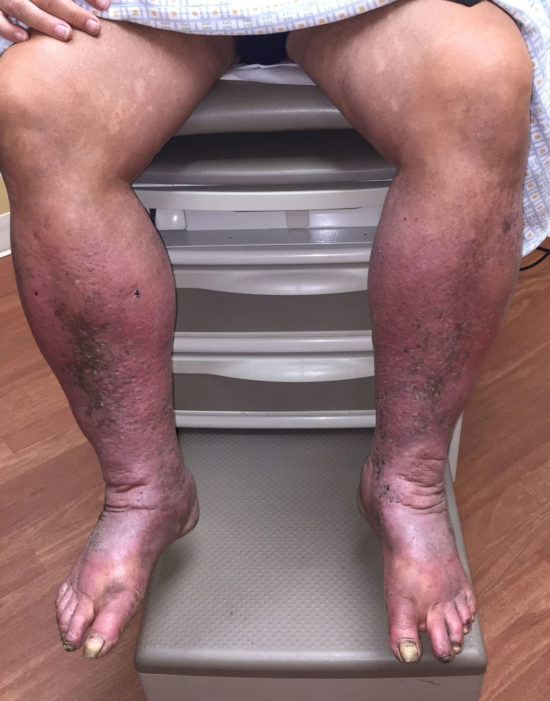
Lipedema is another pathology that can be mistaken for lower limb lymphedema. Lipedema is relatively common but still poorly understood. A problem almost exclusively of women, it typically arises either at puberty, in association with pregnancy, or during menopausal change. Lipedema is a disease of the subcutaneous fat in the lower extremities. Its pathogenesis is likely initiated by lymphatic microvascular pathology (perhaps promoted by hormonal and genetic factors), yet, in contrast to lymphedema, pitting edema will not be present unless lymphedema supervenes in the late stages of the disease. Also in contrast to lymphedema, the involvement of the limbs is quite symmetrical. Foot involvement in lymphedema is variable, but in lipedema, the feet are typically uninvolved (Figure 4). Lipedema is characterized by a significant pain component and often occasions easy bruising, which is not a feature of lymphedema.
Treatment Goals
The treatment goals in lower limb lymphedema are straightforward: to prevent disease progression, to achieve meaningful reduction of edema volume and to thereafter maintain the reduction of limb size, to ease the symptomatic burden of lymphedema, to improve limb function, and to prevent skin infection. The indicated treatment strategy will depend upon the symptoms and the severity of the condition. While pharmacological and surgical interventions can play a role in lymphedema management, the mainstay of treatment intervention is comprised of conservative physical approaches. These treatment strategies, known collectively as chronic decongestive therapy, include:
- manual lymphatic massage (otherwise known as manual lymphatic drainage, or MLD)
- multilayer bandaging and other complex decongestive physiotherapeutic maneuvers
- appropriate application of compression garments
- limb exercises and limb elevation
- pneumatic biocompression, and
- low-level laser therapy
These therapeutic modalities all have acute and chronic implications for their use. In general, decongestive therapy with multilayer bandaging is initiated by highly trained lymphedema therapists and utilized in an intensive course of 4-6 weeks of these interventions, designed to minimize the existing edema volume. Thereafter, the patient is taught to undertake self-management with skin care, exercise, and the daily (and sometimes nightly) application of properly fitted compression garments. Home use of pneumatic biocompression and other devices can be optionally included in the treatment regimen.
It should be underscored that compression garments do not, by themselves, reduce limb volume; rather, they are designed to prevent edema re-accumulation. Hence, garments do not play a role, and should not be prescribed, in the initial care of an untreated patient (unless the goal is to stabilize a newly diagnosed, mild or subclinical edema).
The surgical management of lower limb lymphedema may optionally include debulking interventions, such as suction-assisted lipectomy, or microsurgical procedures designed to restore lymphatic vascular function. Historically, pharmacological therapy has not played a major role in the management of lymphedema, but this may change in the future as newer insights guide anti-inflammatory and other therapeutic strategies to minimize the pathology of lymphedema. In addition, consideration of chronic antibiotic suppression may be appropriate for selected patients that manifest recurrent, severe episodes of soft tissue infection.
Failure to Address Impacts Quality of Life
In most cases, the diagnosis of lower limb lymphedema should be quite straightforward, and treatment interventions, particularly if undertaken early in the course of the disease, effectively stabilize the affected limb and produce significant improvement in limb size and function. Nevertheless, patients with swollen limbs continue to suffer from the persistent limited motivation of healthcare professionals to recognize and treat the problem. The published medical literature documents that, in cancer-associated lymphedema, patients encounter delays in diagnosis, barriers in the access to treatment, and conflicting advice on the management of their lymphedema. In general, treatment of lower limb lymphedema, when it is recognized and evaluated, receives treatment at a relatively advanced stage. The lay public awareness of lymphedema of the leg lags behind its recognition of breast cancer-associated lymphedema of the upper limb. In cancer treatment centers, healthcare professionals increasingly provide advice and services for breast cancer survivors, yet comparable interventions for the prevention and early detection of lower limb lymphedema are generally much less readily available.
Among cancer patients, the advent of lymphedema is reported to be one of central issues in survivorship. In these patients, as in those with lymphedema of any cause, its presence has a profound impact on clothing and footwear choices, quality of life, financial status, and physical and social engagement. Lymphedema has an objectively documented impact on emotional well-being. The impact of lymphedema in the upper limb is comparable in nature, yet in those with leg involvement, reports of fatigue are much more prevalent and the intensity and level of distress in lower limb lymphedema is exaggerated.
Nearly all of the provoking factors for lower limb lymphedema are commonly encountered in the general public, which serves to explain the high population prevalence of chronic lower limb edema. Furthermore, the incidence of cancer and the prevalence of obesity continue to rise, thereby predicting a parallel increase in the incidence and the public health burden of lower limb lymphedema. There is an obvious imperative to provide appropriate guidelines not only for patients but also for healthcare professionals. One of the barriers to the development of evidence-based guidelines on risks, diagnosis, and management is the historical lack of methodically conducted research on these topics. Undoubtedly, there is still much to be learned and studied yet, certainly, the current body of knowledge is sufficient to create concern over a disease whose impact and population burden are likely to be underestimated. For cancer patients in particular, focused educational efforts can help to identify the potential triggers for the onset of overt lymphedema and thereby provide effective strategies for risk reduction and disease modification. Patients can be informed to facilitate appropriate decisions regarding treatments and elective lifestyle behaviors. For those who experience the onset of overt disease, reassurance can be derived from the knowledge that treatment of limb lymphedema has the capacity to ameliorate function and restore quality of life.
Stanley G. Rockson, MD, is the Allan and Tina Neill Professor of Lymphatic Research and Medicine at the Stanford University School of Medicine. He is also co-founder of the Lymphatic Research and Education Network (LE&RN), an internationally recognized non-profit organization founded in 1998 to fight lymphatic diseases and lymphedema through education, research, and advocacy.
Suggested Reading
- Gianesini S, Obi A, Onida S, et al. Global guidelines trends and controversies in lower limb venous and lymphatic disease: Narrative literature revision and experts’ opinions following the vWINter international meeting in Phlebology, Lymphology & Aesthetics, 23-25 January 2019. Phlebol. 2019;34(1 Suppl):4-66. doi: 10.1177/0268355519870690.
- Lerman M, Gaebler JA, Hoy S, et al. Health and economic benefits of advanced pneumatic compression devices in patients with phlebolymphedema. J Vasc Surg. 2019;69(2):571-580.
- Rockson SG, Tian W, Jiang X, et al. Pilot studies demonstrate the potential benefits of antiinflammatory therapy in human lymphedema. JCI Insight. 2018;3(20). pii: 123775. doi: 10.1172/jci.insight.123775.
- Rockson SG, Keeley V, Kilbreath S, Szuba A, Towers A. Cancer-associated secondary lymphoedema. Nat Rev Dis Primers. 2019;5(1):22. doi: 10.1038/s41572-019-0072-5.
 Dr. Stanley G. Rockson is the Founding Chair of the Scientific & Medical Advisory Council for the Lymphatic Education & Research Network (LE&RN). LE&RN is an internationally recognized non-profit organization founded in 1998 to fight lymphatic diseases (LD) and lymphedema (LE) through education, research, and advocacy. Among its many programs and initiatives to make LE and LD a global priority, in 2016 LE&RN wrote the bill that was unanimously passed in the US Senate establishing March 6th as World Lymphedema Day. Please visit LE&RN’s website: LymphaticNetwork.org
Dr. Stanley G. Rockson is the Founding Chair of the Scientific & Medical Advisory Council for the Lymphatic Education & Research Network (LE&RN). LE&RN is an internationally recognized non-profit organization founded in 1998 to fight lymphatic diseases (LD) and lymphedema (LE) through education, research, and advocacy. Among its many programs and initiatives to make LE and LD a global priority, in 2016 LE&RN wrote the bill that was unanimously passed in the US Senate establishing March 6th as World Lymphedema Day. Please visit LE&RN’s website: LymphaticNetwork.org








Hi to those who have reached out to lymphedema sufferers.
I have had this in my legs for many many years and am now 77 years of age.
I have had to wear stockings also for years and certainly knew very little of the disease, the help I could get or anything at all about it. It was so frustrating for a long time and over the years I cried so many times I am surprised I have any tears left. Not to mention the pain etc. It has been so enlightening to read this article. Not even my doctor could tell me much about it over the years.
I live in Victoria Australia. Years ago not many people actually knew of the disease. Thankfully now they do, and so do I.
So happy to have seen and read what has been written recently.
Thank you.