Strict glycemic control is the most reliable treatment for diabetic peripheral neuropathy, but expanding knowledge of central and peripheral nervous system processes may help identify therapeutic methods that can effectively target the underlying mechanisms of pathogenesis.
Strict glycemic control is the most reliable treatment for diabetic peripheral neuropathy, but expanding knowledge of central and peripheral nervous system processes may help identify therapeutic methods that can effectively target the underlying mechanisms of pathogenesis.
By Sarnarendra Miranpuri, DPM, MD; Kush Patel; Gurwattan Miranpuri, PhD; Abhishek Chopra; and Ravinderjit Singh
The dramatic rise in the incidence of diabetes worldwide can be attributed to human behavior and lifestyle changes over the last century. In 1864, Marchel de Calvi described diabetic peripheral neuropathy (DPN) as a consequence rather than a cause of diabetes.1 Neuropathy is a common complication of type 1 and type 2 diabetes, with a prevalence of nearly 8% in newly diagnosed patients with diabetes and more than 50% in patients with long-standing disease.2 Several risks factors are associated with DPN, including hyperglycemia, older age, tobacco use, hypertension, obesity, alcohol consumption, and taller height.1,2 The landmark Diabetes Control and Complication Trial demonstrated that proper glycemic control can reduce the development and progression of neuropathy significantly, by 64%.3
The presentation of diabetic neuropathy characteristically involves a burning or tingling sensation, hyperesthesia, sensory loss in the feet or hands, or combinations of these factors.1 Patients may also experience cold or numb sensations. Sensorimotor and autonomic neuropathies are the most common diabetic neuropathies. Sensorimotor neuropathy presents with pain, paresthesia, and sensory loss, while autonomic neuropathy can contribute to myocardial infarction, malignant arrhythmia, and sudden death.
Distal sensory diabetic polyneuropathy affects up to 50% of patients.2 The symptoms frequently affect patients’ ability to perform daily activities. Up to half of these patients, however, may be asymptomatic, and an incidental finding of a painless foot ulceration can confirm the diagnosis.2
Pathophysiologically, DPN involves metabolic and vascular dysfunction. Hyperglycemia, paramount to the pathogenesis of DPN, plays a central role in the cascade of nerve damage, which is described below:1
1) Increased intracellular glucose and nerve and vascular tissue increases sorbitol and fructose and decreases myo-inositol. It also reduces nicotinamide adenine dinucleotide phosphate, which leads to impairment of the primary vasodilatory system. The impaired vascular supply to the nerves ultimately plays a role in DPN.
2) Advanced glycation end products (hemoglobin, plasma albumin, lipoproteins, fibrin, and collagen) cause tissue damage.
3) Oxidative stress is the main pathological process that induces nerve damage by a direct toxic effect or by inhibition of nitric oxide production, thereby leading to nerve ischemia.
4) Intracellular glucose activates protein kinase C, decreases nerve growth factor, and decreases gamma-linolenic acid (subsequently reducing prostacyclin, a vasodilator), which is necessary for ensuring blood flow.
Currently, strict glycemic control is the most tangible treatment for DPN. Expanding knowledge of central and peripheral nervous system processes may help identify therapeutic methods that effectively target the underlying mechanisms of pathogenesis. An improved therapeutic approach may include using protective and regenerative factors to enhance the action to insulin-regulated genes in the endothelial cells to promote antioxidant and anti-inflammatory factors.4
A large number of neuroanatomical, neurophysiologic, and neurochemical mechanisms are thought to contribute to the development and maintenance of DPN.1 Pathogenetic-oriented treatment overlooks the role of glycemic control and cardiovascular risk factors. Combination treatments involving pathogenetic and symptomatic drugs, however, are the most effective treatments for DPN.5
Long-standing therapies
Restoration of function and improved pain control are the treatment goals for DPN. Prior to initiation of medicinal therapy, clinicians can help manage risks of DPN with optimal glycemic control, improved lipid levels, blood pressure regulation, smoking cessation, and reduction of alcohol consumption.2 There are several established modalities used in clinical practice for the treatment of DPN and improving patient quality of life.
First-line therapy with tricyclic antidepressants (TCAs, including amitriptyline, nortriptyline, imipramine, and desipramine)6-12 in appropriately selected patients has been used widely without approved labeling from the US Food and Drug Administration (FDA). TCAs are thought to relieve pain by blocking neuronal reuptake of norepinephrine and serotonin, thereby inhibiting neurotransmitters in nociceptive pathways.7 In particular, TCAs are used for deep pain (“pins and needles,” “electric,” or numb, achy pain).
A recent Cochrane review assessed five studies on the use of TCAs in the treatment of DPN and revealed overall effectiveness with a number needed to treat (NNT) of 1.3.12 Of the TCAs, amitriptyline has the most published data in support of its use as a treatment for DPN; therefore, it should be recommended as the initial therapy in young patients. Desipramine is recommended for elderly patients and patients who are at risk for anticholinergic adverse effects.7 These adverse effects often lead clinicians or their patients, particularly older adults, to discontinue use despite the overall effectiveness and affordability of this group of medications.
Although used to treat anxiety disorders, serotonin and norepinephrine reuptake inhibitors (SNRIs, including duloxetine, venlafaxine, and milnacipran)2,12 are better tolerated and have fewer drug interactions than the TCAs typically used for DPN. A 2007 Cochrane review revealed an NNT of 3.1 for venlafaxine and 6 for duloxetine.13,14 The selective serotonin reuptake inhibitors paroxetine and citalopram can also be used to treat DPN, but have a limited role.12
Anticonvulsants (eg, gabapentin, pregabalin)14 can be used for DPN if there is inadequate response to TCAs. Gabapentin and pregabalin bind to the alpha-2-delta subunit of the calcium sensitive channels, modulating release of neurotransmitter. The FDA first approved gabapentin in 1994 for use in adult patients with partial epilepsy. In 1998, animal and human studies showed gabapentin has a possible analgesic action.5 However, up-titrating the dosing to the level necessary for achieving a therapeutic effect is commonly needed.5 Pregabalin, conversely, has a narrow therapeutic dosing range, which ensures a predictable response and enables an easy dosing process. Pregabalin is also FDA approved for treatment of DPN pain.
Another anticonvulsant, carbamazepine, has been used in treating neuropathy since the 1960s. It requires laboratory monitoring (renal function, liver function, reticulocyte count, platelet count).15 Topiramate and lamictal are other anticonvulsants that lack evidence for their use in treating DPN.
Opiate (eg, methadone, levorphanol, morphine, oxycontin)1,12 monotherapy, although controversial, is used to treat DPN in patients who fail other therapies. Nine studies showed a consistent benefit with this group of medications, with an approximately 20% to 30% reduction in pain.16 Despite the concern of dependency with chronic opiate therapy, study guidelines do suggest a benefit to patients with DPN.17 Tramadol, a synthetic opiate-like centrally acting narcotic analgesic, acts at mu-opioid receptors and inhibits reuptake of norepinephrine and serotonin. With a lower abuse potential than true opiates, the NNT with tramadol is 3.8.18
Topical therapies (capsaicin cream and lidocaine 5% patches)19,20 have been used to treat neuropathic conditions. Capsaicin, an extract from chili pepper, stimulates depletion of substance P, the most common neurotransmitter in the pain transmission pathway. This cream has been specifically targeted for patients with superficial pain (eg, burning, allodynia, tingling). Most patients experience an initial burning sensation that may persist for several weeks. Lidoderm patches block neuronal sodium channels and have shown limited effectiveness in recent trials. The prime benefit of topical therapy is that it can be used in conjunction with systemic treatment.
Other therapies include N-methyl-D-aspartate (NMDA) antagonists, aldose reductase inhibitors, neurotropic factors, vascular endothelial growth factor, gamma linolenic acid, protein kinase C beta inhibitors, immune therapy, and hyperbaric oxygen therapy.1,12,21,22 L-carnitine and alpha-lipoic acid are alternative therapeutic agents that are available over the counter, but more long-term data are required to demonstrate their efficacy. Despite limited clinical data, these alternative options are still being used in patients with DPN.
Emerging therapies
Gene therapy and growth factor treatment have also been studied as viable treatments for DPN.26 Creation of excess matrix metalloproteinase (MMP)-9 and MMP-2 causes deterioration in the extracellular matrix of blood vessels, which in turn can cause sensory or neuropathic difficulties for patients with diabetes.4 Using minocycline and aspirin as nonselective cyclooxygenase inhibitors can be a novel method of decreasing diabetic neuropathy and restoring motor and sensory nerve conduction velocity.25
Resveratrol, a natural phenol, may play a significant role in treating the diabetic foot.26 It may reinstitute insulin sensitivity, cytokine formation, tissue reconstruction, microcirculation, and peripheral nerve function.
Ghrelin, a small peptide, has been shown to have a relevant effect in multiple tissue systems when used to treat DPN due to its anti-inflammatory and regenerative abilities within neural tissues.27
ABT-594, a neuronal nicotinic acetylcholine receptor ligand, is extremely potent in animal models with neuropathic and nociceptive pain. In a placebo-controlled, double-blinded, seven-week study, individuals with painful diabetic neuropathy were randomized into four groups.28 They received twice-daily treatment of either placebo or ABT-594. The experimental treatment was associated with improved pain scores in 50% of patients, but there were adverse effects, including headache, vomiting, dizziness, nausea, and abnormal dreams. Users of nicotine were less sensitive to these adverse effects.
Oxidative and nitrosative stress are key factors to consider when creating potential therapies for DPN.29,30 Coenzyme Q10 acts as a free radical scavenger and an antioxidant to decrease oxidative stress within the peripheral and central nervous system.
Puerarin, one of several known isoflavones, has been found to alleviate the mechanical and thermal nociceptive response triggered by diabetes without interfering with the normal nociceptive process. Furthermore, it reduces levels of nuclear factor-κB and other proinflammatory cytokines, such as interleukin (IL)-6, IL-1β, and tumor necrosis factor (TNF)-α.31
Lipoic acid has therapeutic value as a nutritional supplement for DPN. Because of lipoic acid’s lack of bioavailability due to its accelerated metabolism and instability, pharmacophores in conjunction with lipoic acid hybrids are being tested.32
Animal models
The streptozotocin diabetic rat has, to date, been the most commonly used model of DPN.33 A systematic review of possible natural drugs to combat chronic neuropathic pain include: carotenoids (10%), phenols (10%), alkaloids (14%), terpenes (17%), flavonoids (28%), and others (21%).34 One quarter of these studies looked at streptozotocin-induced diabetic neuropathy.
Baclofen, a gamma aminobutyric acid B (GABAB) receptor agonist, is a novel treatment for DPN that has been tested in animal models.35 The attenuation of DPN can be accomplished by down-regulating the expression of N-methyl-D-aspartate receptor subunit 2B (NR2B) and phosphorylated (p)-cyclic AMP response element-binding protein (CREB) through the activation of the GABAB receptor.
Combination therapies—which include neutral endopeptidase inhibitors, aldose reductase inhibitors, lipoic acid supplementation, and insulin therapy with antioxidants—have been shown to prevent the loss of nerve conduction velocity in a diabetic rat model.36 These therapies decrease levels of neuropeptide Y, tyrosine hydroxylase, somatostatin, IL-1β, and MMP-2 in streptozotocin-induced diabetic bone marrow supernatant.
Treating diabetic rats with a combination of enalapril, alpha lipoic acid, and menhaden oil ameliorated diabetes-induced steatosis, and elevated serum lipid levels and improved motor and sensory nerve conduction and corneal sensitivity of epineurial arterioles of the sciatic nerves.37 These findings suggest the combination therapy’s potential for treatment of vascular and neural complications caused by type 2 diabetes.
Gene and stem cell therapy
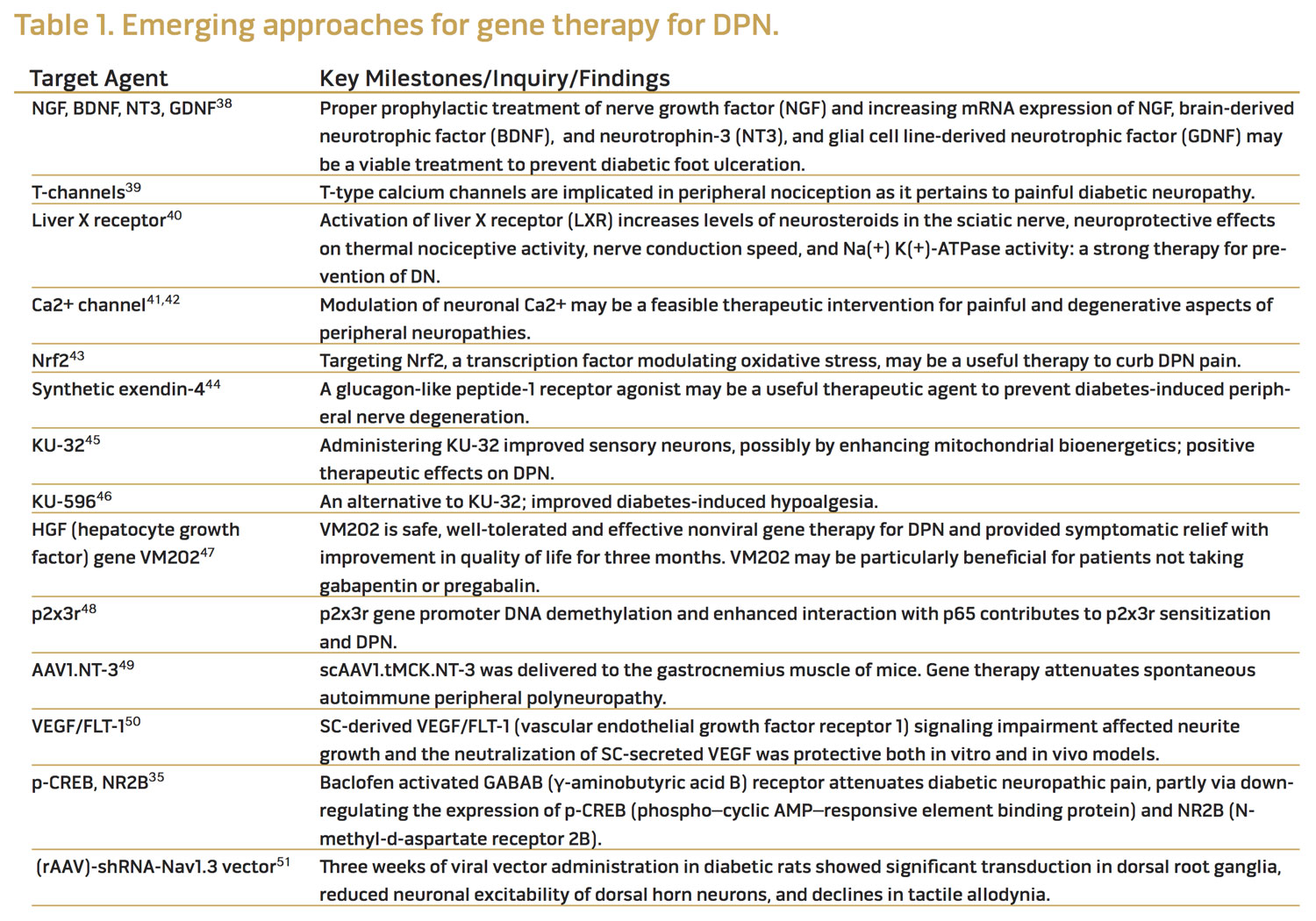
Gene therapy has been a high-yield research focus in the past few years with identification of various well-tolerated and effective gene therapy options for painful diabetic neuropathy. Various emerging gene therapy approaches for DPN35,38-51 are shown in Table 1.
Clinical and basic science studies are beginning to highlight that stem cell therapies may be a suitable option in the near future for regenerative medical therapy. Indeed, stem cell therapy is continually being investigated worldwide in hopes of developing safer and more efficacious treatment options for diabetic neuropathy. Emerging stem cell therapies for DPN are shown in Table 2.52-61
Future perspectives
As therapeutic approaches to DPN have evolved, researchers’ understanding of the mechanisms underlying DPN pathogenesis has expanded (Table 3).38-44,62-68 In addition, recent clinical trials are evaluating the efficacy of multiple proteins involved in the treatment of DPN, such as erythropoietin analogs, angiotensin II receptor type 2 antagonists, and sodium channel blockers.69
This knowledge will be instrumental for the development of gene and cell therapies that will provide a foundation for the treatment of future generations.
Sarnarendra Miranpuri, DPM, MD, is a podiatric surgeon with Aurora Health Care in Oshkosh, WI. Kush Patel is an undergraduate research scholar; Gurwattan Miranpuri, PhD, is a senior scientist; Abhishek Chopra is an undergraduate research scholar; and Ravinderjit Singh is an undergraduate research scholar, all in the Department of Neurological Surgery at the University of Wisconsin School of Medicine and Public Health in Madison.
- Kaur S, Pandhi P, Dutta P. Painful diabetic neuropathy: an update. Ann Neurosci 2011;18(4):168-175.
- Deli G, Bosnyak E, Pusch G, et al. Diabetic neuropathies: diagnosis and management. Neuroendocrinology 2013;98(4):267-280.
- Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329(14):977-986.
- Rask-Madsen C, King G. Vascular complications of diabetes: mechanisms of injury and protective factors. Cell Metab 2013;17(1):20-33.
- Várkonyi T, Putz Z, Keresztes K, et al. Current options and perspectives in the treatment of diabetic neuropathy. Curr Pharm Des 2013;19(27):4981-5007.
- Dick I, Brochu R, Purohit Y, et al. Sodium channel blockade may contribute to the analgesic efficacy of antidepressants. J Pain 2007;8(4):315-324.
- Joss J. Tricyclic antidepressant use in diabetic neuropathy. Ann Pharmacother 1999;33(9):996-1000.
- Crutchfield C, Breaud A, Clarke W. Quantification of tricyclic antidepressants in serum using liquid chromatography electrospray tandem mass spectrometry (HPLC-ESI-MS/MS). Methods Mol Biol 2016;1383:265-270.
- Hayasaka Y, Purgato M, Magni L, et al. Dose equivalents of antidepressants: Evidence-based recommendations from randomized controlled trials. J Affect Disord 2015;180:179-184.
- Berm E, Paardekooper J, Brummel-Mulder E, et al. A simple dried blood spot method for therapeutic drug monitoring of the tricyclic antidepressants amitriptyline, nortriptyline, imipramine, clomipramine, and their active metabolites using LC-MS/MS. Talanta 2015;134:165-172.
- Saarto T, Wiffen PJ. (2005). Antidepressants for neuropathic pain. Cochrane Database Syst Rev 2007;(4):CD005454.
- Lindsay T, Rodgers B, Savath V, et al. Treating diabetic peripheral neuropathic pain. Am Fam Physician 2010;82(2):151-158.
- Wernicke JF, Pritchett YL, D’Souza DN, et al. A randomized controlled trial of duloxetine in diabetic peripheral neuropathic pain. Neurology 2006;67(8):1411-1420.
- Argoff CE, Backonja MM, Belgrade MJ, et al. Consensus guidelines: treatment planning and options. Diabetic peripheral neuropathic pain. Mayo Clin Proc 2006;81(4 Suppl):S12-S25.
- Standaert D, Young A. Treatment of central nervous system degenerative disorders. In: Hardman J, Limbird L, eds. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill; 2001:533.
- Eisenberg E, McNicol E, Carr DB. Opioids for neuropathic pain. Cochrane Database Syst Rev 2006;(3):CD006146.
- Chou R, Fanciullo GJ, Fine PG, et al. Clinical guidelines for the use of chronic opioid therapy in chronic noncancer pain. J Pain 2009;10(2):113-130.
- Hollingshead J, Dühmke RM, Cornblath DR. Tramadol for neuropathic pain. Cochrane Database Syst Rev 2006;(3):CD003726.
- Mason L, Moore RA, Derry S, et al. Systematic review of topical capsaicin for the treatment of chronic pain. BMJ 2004;328(7446):991.
- Meier T, Wasner G, Faust M, et al. Efficacy of lidocaine patch 5% in the treatment of focal peripheral neuropathic pain syndromes: a randomized, double-blind, placebo-controlled study. Pain 2003;106(1):151-158.
- Zhang W, Murakawa Y, Wozniak K, et al. The preventive and therapeutic effects of GCPII (NAALADase) inhibition on painful and sensory diabetic neuropathy. J Neurol Sci 2006;247(2):217-223.
- Salinthone S, Yadav V, Bourdette D, et al. Lipoic acid: a novel therapeutic approach for multiple sclerosis and other chronic inflammatory diseases of the CNS. Endocr Metab Immune Disord Drug Targets 2008;8(2):132-142.
- Tahrani A, Askwith T, Stevens M. Emerging drugs for diabetic neuropathy. Expert Opin Emerg Drugs 2010;15(4):661-683.
- Kuhad A, Singh P, Chopra K. Matrix metalloproteinases: potential therapeutic target for diabetic neuropathic pain. Expert Opin Ther Targets 2015;19(2):177-185.
- Bhatt L, Veeranjaneyulu A. Minocycline with aspirin: a therapeutic approach in the treatment of diabetic neuropathy. Neurol Sci 2010;31(6):705-716.
- Bashmakov Y, Assaad-Khalil S, Petyaev I. Resveratrol may be beneficial in treatment of diabetic foot syndrome. Med Hypotheses 2011;77(3):364-367.
- Fields D, Miranpuri S, Miranpuri G, et al. The multifunctional and multi-system influence of Ghrelin in the treatment of diabetic and spinal cord injury induced Neuropathy. Ann Neurosci 2011;18(3):118-122.
- Dutta S, Hosmane BS, Awni WM. Population analyses of efficacy and safety of ABT-594 in subjects with diabetic peripheral neuropathic pain. AAPS J 2012;14(2):168-175.
- Fakhrabadi MA, Ghotrom AZ, Mozaffari-Khosravi H, et al. Effect of coenzyme Q10 on oxidative stress, glycemic control and inflammation in diabetic neuropathy: a double blind randomized clinical trial. Int J Vit Nutr Res 2014;84(5-6):252-260.
- Zhang Y, Song C, Yuan Y, et al. Diabetic neuropathic pain development in type 2 diabetic mouse model and the prophylactic and therapeutic effects of coenzyme Q10. Neurobiol Dis 2013;58:169-178.
- Wu J, Zhang X, Zhang B. Efficacy and safety of puerarin injection in treatment of diabetic peripheral neuropathy: a systematic review and meta-analysis of randomized controlled trials. J Traditional Chinese Med 2014;34(4):401-410.
- Ziegler D, Reljanovic M, Mehnert H, et al. Alpha-lipoic acid in the treatment of diabetic polyneuropathy in Germany: current evidence from clinical trials. Exp Clin Endocrinol Diabetes 1999;107(7):421-430.
- Biessels G, Bril V, Calcutt N, et al. Phenotyping animal models of diabetic neuropathy: a consensus statement of the diabetic neuropathy study group of the EASD (Neurodiab). J Peripher Nerv Syst 2014;19(2):77-87.
- Quintans JS, Antoniolli A, Almeida J, et al. Natural products evaluated in neuropathic pain models – a systematic review. Basic Clin Pharmacol Toxicol 2014;114(6):442-450.
- Liu P, Guo W, Zhao X, et al. Intrathecal baclofen, a GABAB receptor agonist, inhibits the expression of p-CREB and NR2B in the spinal dorsal horn in rats with diabetic neuropathic pain. Can J Physiol Pharmacol 2014;92(8):655-660.
- Dominguez J, Yorek M ,Grant B. Combination therapies prevent the neuropathic, proinflammatory characteristics of bone marrow in streptozotocin-induced diabetic rats. Diabetes 2015;64(2):643-653.
- Davidson E, Holmes A, Coppey L, et al. Effect of combination therapy consisting of enalapril, α-lipoic acid, and menhaden oil on diabetic neuropathy in a high fat/low dose streptozotocin treated rat. Eur J Pharmacol 2015;765:258-267.
- Anand P. Neurotrophic factors and their receptors in human sensory neuropathies. Prog Brain Res 2004;146:477-492.
- Latham J, Pathirathna S, Jagodic M, et al. Selective T-type calcium channel blockade alleviates hyperalgesia in ob/ob mice. Diabetes 2009;58(11):2656-2665.
- Cermenati G, Giatti S, Cavaletti G, et al. Activation of the liver X receptor increases neuroactive steroid levels and protects from diabetes-induced peripheral neuropathy. J Neurosci 2010;30(36):11896-11901.
- Fernyhough P, Calcutt N. Abnormal calcium homeostasis in peripheral neuropathies. Cell Calcium 2010;47(2):130-139.
- Nazıroğlu M, Dikici D, Dursun S. Role of oxidative stress and Ca²⁺ signaling on molecular pathways of neuropathic pain in diabetes: focus on TRP channels. Neurochem Res 2012;37(10):2065-2075.
- Negi G, Kumar A, Joshi R, et al. Oxidative stress and Nrf2 in the pathophysiology of diabetic neuropathy: old perspective with a new angle. Biochem Biophys Res Commun 2011;408(1):1-5.
- Liu W, Jin H, Lee K, et al. Neuroprotective effect of the glucagon-like peptide-1 receptor agonist, synthetic exendin-4, in streptozotocin-induced diabetic rats. Br J Pharmacol 2011;164(5):1410-1420.
- Urban M, Pan P, Farmer K, et al. Modulating molecular chaperones improves sensory fiber recovery and mitochondrial function in diabetic peripheral neuropathy. Exp Neurol 2012;235(1):388-396.
- Ma J, Pan P, Anyika M, et al. Modulating molecular chaperones improves mitochondrial bioenergetics and decreases the inflammatory transcriptome in diabetic sensory neurons. ACS Chem Neurosci 2015;6(9):1637-1648.
- Kessler J, Smith A, Cha B, et al. Double-blind, placebo-controlled study of HGF gene therapy in diabetic neuropathy. Ann Clin Transl Neurol 2015;2(5):465-478.
- Zhang H, Hu J, Zhou Y, et al. Promoted interaction of nuclear factor-κB with demethylated purinergic P2X3 receptor gene contributes to neuropathic pain in rats with diabetes. Diabetes 2015;64(12):4272-4284.
- Yalvac M, Arnold W, Braganza C, et al. AAV1.NT-3 gene therapy attenuates spontaneous autoimmune peripheral polyneuropathy. 2016;23(1):95-102.
- Taiana M, Lombardi R, Porretta-Serapiglia C, et al. Neutralization of schwann cell-secreted VEGF is protective to in vitro and in vivo experimental diabetic neuropathy. PLoS One 2014;9(9):e108403.
- Tan A, Samad O, Dib-Hajj S, et al. Virus-mediated knockdown of Nav1.3 in dorsal root ganglia of STZ-induced diabetic rats alleviates tactile allodynia. Mol Med 2015;21:544-552.
- Kim B, Lee J, Schuchman E, et al. Synergistic vasculogenic effects of AMD3100 and stromal-cell-derived factor-1α in vasa nervorum of the sciatic nerve of mice with diabetic peripheral neuropathy. Cell Tissue Res 2013;354(2):395-407.
- Kramerov A, Ljubimov A. Stem cell therapies in the treatment of diabetic retinopathy and keratopathy. Exp Biol Med 2016;241(6):559-568.
- Nasli-Esfahani E, Ghodsi M, Amini P, et al. Evaluation of fetal cell transplantation safety in treatment of diabetes: a three-year follow-up. J Diabetes Metab Disord 2015;14:33.
- Xia N, Xu J, Zhao N, et al. Human mesenchymal stem cells improve the neurodegeneration of femoral nerve in a diabetic foot ulceration rats. Neurosci Lett 2015;597:84-89.
- Himeno T, Kamiya H, Naruse K, et al. Mesenchymal stem cell-like cells derived from mouse induced pluripotent stem cells ameliorate diabetic polyneuropathy in mice. Biomed Res Int 2013:259187.
- Kim B, Lee J, Schuchman E, et al. Synergistic vasculogenic effects of AMD3100 and stromal-cell-derived factor-1α in vasa nervorum of the sciatic nerve of mice with diabetic peripheral neuropathy. Cell Tissue Res 2013;354(2):395-407.
- Wang Y, Xue M, Xuan Y, et al. Mesenchymal stem cell therapy improves diabetic cardiac autonomic neuropathy and decreases the inducibility of ventricular arrhythmias. Heart Lung Circ 2013;22(12):1018-1025.
- Kojima H, Kim J, Chan L. Emerging roles of hematopoietic cells in the pathobiology of diabetic complications. Trends Endocrinol Metab 2014;25(4):178-187.
- Han J, Sin M, Yoon Y. Cell therapy for diabetic neuropathy using adult stem or progenitor cells. Diabetes Metab J 2013;37(2):91-105.
- Yang M, Sheng L, Zhang TR, et al. Stem cell therapy for lower extremity diabetic ulcers: where do we stand? Biomed Res Int 2013:462179.
- Miyazaki Y, Koike H, Akane A, et al. Spinal cord stimulation markedly ameliorated refractory neuropathic pain in transthyretin Val30Met familial amyloid polyneuropathy. Amyloid 2011;18(2):87-90.
- Talbot S, Couture R. Emerging role of microglial kinin B1 receptor in diabetic pain neuropathy. Exp Neurol 2012;234(2):373-381.
- Brederson J, Joshi S, Browman K, et al. PARP inhibitors attenuate chemotherapy-induced painful neuropathy. J Peripher Nerv Syst 2012;17(3):324-330.
- Taliyan R, Sharma P. Possible mechanism of protective effect of thalidomide in STZ-induced-neuropathic pain behavior in rats. Inflammopharmacology 2012;20(2):89-97.
- Wang D, Couture R, Hong Y. Activated microglia in the spinal cord underlies diabetic neuropathic pain. Eur J Pharmacol 2014;728:59-66.
- Choi J, Chandrasekaran K, Inoue T, et al. PGC-1α regulation of mitochondrial degeneration in experimental diabetic neuropathy. Neurobiol Dis 2014;64:118-130.
- Guo S. Insulin signaling, resistance, and the metabolic syndrome: insights from mouse models to disease mechanisms. J Endocrinol 2014;220(2):T1-T23.
- Javed S, Alam U, Malik R. Treating diabetic neuropathy: present strategies and emerging solutions. Rev Diabet Stud 2015;12(1-2):63-83.