By Lars I. E. Oddsson, Teresa Bisson, Helen S. Cohen, Ikechukwu Iloputaife, Laura Jacobs, Doris Kung, Lewis A. Lipsitz, Brad Manor, Patricia McCracken, Yvonne Rumsey, Diane M. Wrisley and Sara R. Koehler-McNicholas
Persistent problems with gait and balance function may lead to falls, fractures, and other serious injuries in older adults.
 Persistent problems with gait and balance function may lead to falls, fractures, and other serious injuries in older adults. In addition to other age-related risk factors, sensory peripheral neuropathy (PN), leading to impaired plantar sensation, is an independent risk factor for falls. The incidence of injuries due to falls is 15 times higher in patients with diabetic PN than in healthy individuals.
Persistent problems with gait and balance function may lead to falls, fractures, and other serious injuries in older adults. In addition to other age-related risk factors, sensory peripheral neuropathy (PN), leading to impaired plantar sensation, is an independent risk factor for falls. The incidence of injuries due to falls is 15 times higher in patients with diabetic PN than in healthy individuals.
Although strength and balance training in patients with PN may help reduce fall risk falls, strength training in patients with PN appears to have less impact on balance. Its effects are mainly compensatory and do not address impaired somatosensation, the root cause of balance problems related to PN. Further, balance training activities must be specific and conducted with sufficient intensity and frequency to be helpful. Clear guidelines regarding frequency of balance exercises are currently lacking although 3 sessions a week may be a minimum necessary to see an improvement. Although falls can be prevented, they continue to be a large problem in older adults, indicating a continued need for novel solutions. One such novel solution is Walkasins, a wearable non-invasive sensory prosthesis intended to substitute nerve function related to impaired plantar sensation (Figure 1).
This article reports on the extended long-term use data after 26 weeks of Walkasins use in the walk2Wellness Clinical Trial—data from the 10-week home-based use of Walkins has already shown that individuals with PN and a high risk of falls improved their Functional Gait Assessment (FGA) scores, Gait Speed, and Timed Up and Go (TUG) times. The data also showed a decrease in the number of fall risk factors as well as fall rate from baseline to 10 weeks for individuals who reported falls in the 6 months preceding study participation.
Methods
Forty-four people at 4 study sites participated in the walk2Wellness trial. Inclusion criteria included the following: male or female; ages 21–90; a formal diagnosis of sensory PN prior to participating in the study; self-reported problems with balance; ability for transfers or ambulation on level surfaces at fixed cadence as assessed by trained study personnel; FGA score < 23, the cut-off score for high fall risk; foot size to allow the Walkasins device to function properly; and ability to complete all functional outcome measures without the use of an assistive device. Use of an assistive device during daily activities was permissible.
Participants agreed not to initiate any balance training or balance-related therapy during the first 10 weeks of the trial. They provided information on number of falls experienced and any injuries sustained in the previous 6 and 12 months, as well as a list of their medications, indication, dose, and frequency, which was updated over the course of the study.
Participants then completed the Activities-Specific Balance Confidence (ABC) Questionnaire and the Vestibular Activities of Daily Living Scale (VADL). At baseline, week 10, and week 26, participants were assessed using the Weinstein Enhanced Sensory Test (WEST) monofilament foot test (0.5, 2, 10, 50, and 200g). Site staff also performed a vibration sensation test using a Rydel-Seiffer Tuning Fork to document loss of sensation. Scoring values ≤4 at the first metatarsal joint is categorized as abnormal.
During the baseline visit, participants donned the Walkasins devices and performed a standardized set of standing and walking balance activities focused on orientation and familiarization with the device. Additional outcomes assessments included the 10-Meter Walk Test, TUG, and 4-Stage Balance Test. Participants also completed several self-reported outcome measures: Patient Health Questionnaire (PHQ-9), PROMIS Pain Interference Short Form 6b, PROMIS Pain Intensity Form 1a, Ability to Participate Short Form 8a, and Satisfaction with Participation in Social Roles Short Form 8a. Participants left the baseline visit with the device and a calendar on which to note any fall events they experienced as well as their use of Walkasins.
Participants returned for in-person visits at weeks 2, 6, 10, and 26, except when prohibited due to COVID-19 restrictions. These visits followed most of the same procedures as the baseline visit. ABC to FGA ratios were calculated at each assessment to measure the degree of internal self-perception of balance capability (ABC score) in relation to the externally observed walking balance performance (FGA score).
Study sites contacted participants via telephone at weeks 14, 18, and 22 to remind them of study requirements and to collect follow-up information regarding health changes, falls, adverse events, pain scores, device usage, and device functioning, as well as whether they had begun any physical therapy for their balance problems. If participants reported adverse events and/or falls during these contacts, site personnel recorded the details.
A post hoc analysis was conducted to compare participants at baseline who reported falls in the previous 6 months (Pre-Fallers [Pre-F], n = 25) to those who did not (Pre-Non-Fallers [PRE-NF], n = 19).

Figure 1. (Left) The two components of the Walkasins prosthetic device, the pressure sensitive flexible foot pad that is placed in the shoe and connects to the leg unit that contains a rechargeable battery, a microprocessor, supporting electronics, and four mechanical tactile stimulators. The embedded software algorithm evaluates pressure data and activates the mechanical tactile stimulators at relevant times during standing and walking to signal balance-related information to the afferent nervous system. (Right) A Walkasins user wearing the device in the process of turning it on. The Walkasins system is worn bilaterally (unilateral components depicted).
Results
Due to circumstances related to COVID-19, 14 of the 44 participants were unable to participate in the 26-week in-person outcomes testing (FGA, 10MWT, TUG, and 4-Stage Balance Test). However, all were assessed on self-reported outcomes over the phone, and all provided reports of falls.
Average reported weekly device use was 5.1 ± 0.4 days. Participants reporting that they used the device weekly either “Every Day” or “At least 5 Days” was 71.8 ± 10.5%. An average of 94.8 ± 3.3% of reporting participants stated they used the device “1–2 Days” or more per week.
Baseline self-selected gait speed and ABC scores were significantly different between the Pre-F and Pre-NF groups, 0.83 vs. 0.97 m/s and 57.8 vs. 69.6%, respectively. A higher observed mean value in PHQ-9 score for the Pre-F group was nearly statistically significant (5.7 vs. 3.3, respectively, P = 0.052). Mean values for all PROMIS measures were near 50, and any observed differences were minor.
After 26 weeks of device use, FGA scores increased across all individuals from 15.0 to 19.2 (P < 0.00001) (Figure 2); self-selected gait speed increased from 0.89 m/s to 0.97 m/s (P = 0.02); and the 4-Stage Balance Test improved from 25.6 s to 28.4 s (P < 0.01). An increase seen in fast gait speed (1.30 to 1.37 m/s) did not reach statistical significance (P = 0.07). Changes in Rydel-Seiffer tuning fork testing scores suggested a small decrease in sensitivity at the site of the lateral malleolus (mean 3.8 to 3.2, P = 0.032, while a noted decrease at the metatarsophalangeal (MTP) joint did not reach statistical significance (P = 0.07).
The Pre-F cohort improved their mean FGA score from 13.7 at baseline to 18.1 at 26 weeks (P < 0.0001). Their observed increases in self-selected gait speed from 0.82 to 0.90 m/s and from 1.13 to 1.23 m/s for fast gait speed were not statistically significant (P = 0.12 and P = 0.07, respectively). The Pre-F group showed a decrease in vibration sensation at the first MTP joint (mean 2.9 to 1.8, P < 0.005) and at the lateral malleolus that did not reach statistical significance (mean 3.75 to 3.25, P = 0.07).
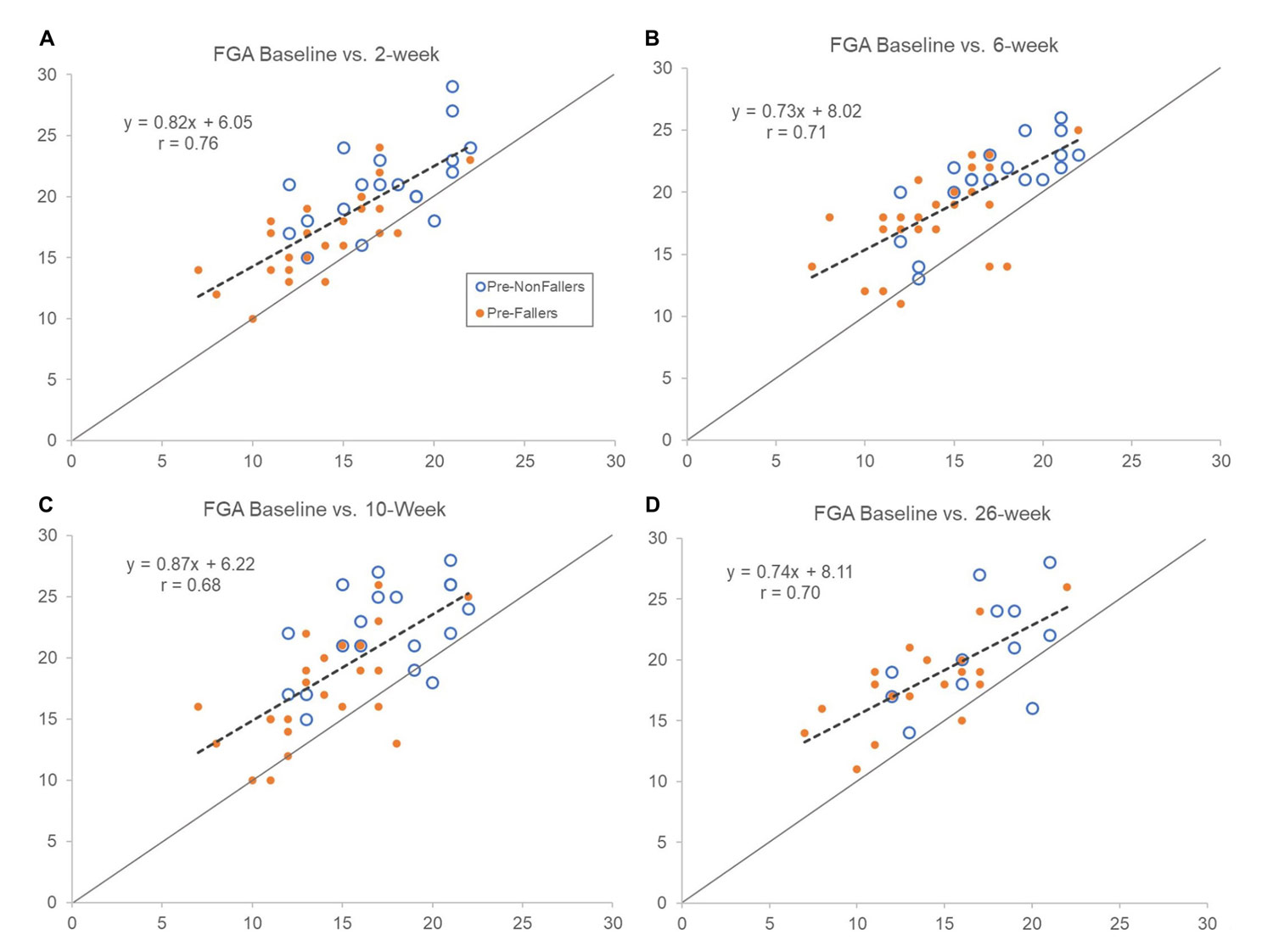
Figure 2. Comparing FGA scores at baseline (horizontal axes) with assessments after 2 (A), 6 (B), 10 (C), and 26 (D) weeks (vertical axes) of device use. Open symbols represent pre-study non-fallers and filled ones are pre-study fallers. Dashed lines show regression line for the whole group. Forty-four participants completed in clinic outcomes testing up to 10 weeks (primary endpoint), and 30 participants completed the 26-week assessment in person. Scores above line of equality indicate improvements and below a decrement in FGA score compared to baseline scores. Notice that regression line slopes are less than 1 indicating slightly larger improvements in FGA score for those with lower baseline scores. Overall, improvements observed after 2 weeks of use appeared sustained throughout the 26 weeks of use.
The Pre-NF group increased their mean FGA score from 17.0 at baseline to 20.8 at 26 weeks (P < 0.004). A small increase in self-selected gait speed from 1.00 m/s to 1.08 m/s was not statistically significant (P = 0.06). Other clinical outcomes, including Rydel-Seiffer tuning fork sensitivity testing scores, remained unchanged at 26 weeks compared to baseline.
Regarding self-reported outcomes, across all participants, a slight but statistically significant increase was seen in PROMIS Ability to Participate scores after 26 weeks of device use (49.9 to 52.6, P < 0.05). All baseline values were maintained at 26 weeks for the Pre-F group while an increase in the PROMIS Satisfaction with Social Roles scores (49.9 to 55.5, P < 0.006) for the Pre-NF group was noted.
The ABC/FGA ratio for the overly confident individuals gradually decreased from high values at baseline to week 6 when the ratio essentially aligned with the low self-confidence participants, who maintained a consistent ABC/FGA ratio of about 3.5 throughout the 26-week trial. However, both groups increased their FGA scores in a similar fashion although the overly confident participants showed higher levels of improvement.
Falls were reported throughout the 26-week period for all 44 participants and separately for the Pre-F (n = 25) and Pre-NF (n = 19) groups. The 44 participants reported a total of 53 falls over 6 months prior to participating in the trial while 39 falls were documented during the 26 weeks of the trial, corresponding to a pre-study mean fall rate of 6.7 falls/1000 patient days (median = 5.6 falls/1000 patient days) and a post-study mean fall rate of 4.8 falls/1000 patient days (median = 0 falls/1000 patient days). Across all participants, the median of the post-study fall rate was lower than the pre-study fall rate (P = 0.044), reflecting a 28% decrease in fall rate. Of the 44 participants, 25 had fallen in the past 6 months (Pre-F); and after 26 weeks, 20 of the 44 participants had reported falling. Overall, 31 of the 39 falls required no treatment while 8 falls (~20%) required treatment with 4 of those (~10%) causing severe injury (2 fractures).
The Pre-F cohort reported 31 falls after 26 weeks compared to 53 falls pre-study corresponding to a pre-study fall rate of 11.8 falls/1000 patient days (median = 11.1), which decreased to 6.7 falls/1000 patient days at 26 weeks (median = 5.0), a 43% decrease in fall rate (P = 0.0043). During the study, 12 did not fall, a 48% statistically significant decrease (P < 0.0001). Of the 31 falls experienced by the Pre-F cohort, 6 (~20%) required treatment. Three of the 4 falls that led to severe injury occurred in the Pre-F group.
Seven of the 19 Pre-NF participants reported falling during the trial (P < 0.0001) indicating an increase in fall rate from 0 to 2.3 falls/1000 patient days at 26 weeks that was statistically significant (P = 0.023). Eight falls were reported by the 7 Pre-NF participants who fell during the study; 2 required treatment and 1 sustained a severe injury. (See Figure 3.)
Discussion
Walkasins is a device that provides mechanical tactile stimuli related to foot pressure for individuals with PN and gait and balance problems. Overall, the study authors’ findings from analyzing outcomes after 26 weeks of wearing Walkasins show that improvements in outcomes seen after 10 weeks of use are sustained longer term.
FGA scores were improved across all participants. Interestingly, these improvements were seen for individuals across the full range of baseline FGA scores (Figure 2) with a tendency to be higher for individuals in the lower FGA range. Similar improvements were seen for the Pre-F and Pre-NF cohorts. Moreover, the 46% decrease in fall rate, compared to pre-study falls that were reported after 10 weeks of use, was sustained at 43% after 26 weeks.
All clinical outcomes improved compared to baseline, but changes in TUG and 4-Stage Balance did not reach statistical significance, likely because the study was underpowered for these measures. It is encouraging to report a statistically significant 0.08 m/s increase in gait speed across all participants in this trial, which is beyond a small meaningful change (0.05 m/s) and close to the range for a substantial meaningful change (0.10 m/s).
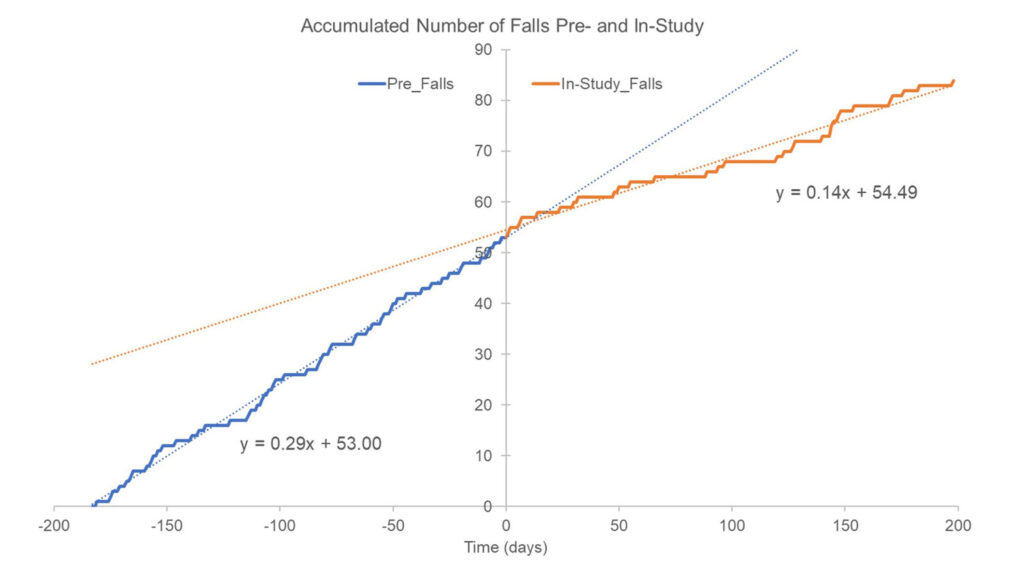
Figure 3. Accumulated number of falls reported 6 months pre-study (blue trace and blue dotted linear regression line) and falls documented in-study (orange trace and orange dotted regression line). The 53 falls reported for the prior 6 months would correspond to 0.29 falls/day (53/180 ~0.29), which represents the slope of the regression line (blue trace). For illustration purposes, the 53 pre-study falls were randomly distributed across the 6 months since their exact time occurrence was unknown. In-study falls are shown as they occurred and were reported by participants throughout the 26 weeks. Notice how the rate of in-study falls appear to begin deviating from pre-study fall rate (slope of dotted blue line) after approximately 20 days of device use.
Participants had a decrease in sensitivity to vibration, an overall small decrease at the lateral malleolus and a larger change at the MTP joint in the Pre-Faller group. Such changes did not occur at the 10-week assessment. This decrease in tuning fork vibration perception may indicate a progression of some participants’ PN and increased sensory loss.
The improvements in clinical outcomes were gained from a single training visit on the use of the device and then simply by wearing it during regular daily activities. In fact, during the first 10 weeks of use, participants were prohibited from doing any physical therapy or balance-related exercise activities to help isolate the effect of the device. Knowing, however, that such interventions may help improve gait and balance function, the participants were permitted to engage in such after the 10 weeks. Therefore, to control for any potential effects of balance exercise interventions, participants were asked during follow-up phone calls at 14, 18, and 22 weeks whether they had begun any physical therapy for their balance problems. Only 1 of the participants answered “yes,” further suggesting that long-term use of the device was the main cause of improvement in clinical outcomes and not exercise or balance interventions.
Interestingly, early observations from a pilot study of 5 participants in the cohort studied here, who completed 26 weeks of device use, show neuroplastic changes in brain network connectivity related to postural control and balance that were associated with improved changes in FGA scores. This finding indicates a direct effect of the sensory prosthesis initiating plastic changes related to sensorimotor interaction and postural control.
One of the most important findings from this trial is the 43% decrease in fall rate for participants with a fall history following 26 weeks of device use. The data on injuries reported from falls are probably clinically significant. In this study, 57% of participants reported falling in the 6 months prior to the study while 45% fell during 6 months of the trial. Eight of the 39 falls during the trial (20.5%) led to injuries where treatment was sought, half of those considered serious. Consequently, both fall rate and injury rate are lower than previous reports of similar populations.
Conclusion
Patients with PN who have gait and balance problems with a high risk of falls including a history of falls can improve their walking balance and decrease their fall rates from long-term use of a wearable non-invasive sensory prosthesis.
This article has been excerpted from “Extended Effects of a Wearable Sensory Prosthesis on Gait, Balance Function and falls after 26 Weeks of Use in Persons with Peripheral Neuropathy and High Fall Risk—The walk2Wellness Trial” Frontiers in Aging Neuroscience. 2022;14:931048. doi: 10.3389/fnagi.2022.931048. Editing has occurred, including the renumbering of tables, and references have been removed for brevity. Use is per CC BY.








