By Cristina Acevedo, PhD; Ian Crane; Rohit Kankaliya; Rajib Mondal, PhD; Albert Ortega; Kevin Urman, PhD
Material selection can strongly impact medical device performance and patient experience. Selection of the optimum materials in combination with the design, will influence how the device performs in promoting recovery, increasing lymphatic fluid, regulating moisture and temperature, providing lasting antimicrobial, antifungal and/or antiviral performance, skin soothing, amongst many others. These additional performance benefits cannot come at the cost of having a negative impact on the skin or environment by the use of harsh chemicals. The expectation is a shift toward a cleaner, more pure solution that reduces the number of chemistries applied and only uses a natural ingredient.
Medical compression therapy is commonly used as either first line of defense or to treat various stages of venous lymphatic disease. However, adverse events and contraindications have been reported related to compression therapy. In a recent review article, skin irritation, allergic skin reaction, discomfort and pain, forefoot edema and lymphedema, bacterial and fungal infection were described as non-severe adverse events, while soft tissue damage necrosis, nerve damage, arterial impairment, venous thromboembolism, and cardiac decompensation were described as severe events. Majority of events were found to occur very rarely (<1/10,000 cases), except skin irritation and discomfort & pain which were reported as common incidences (>1/100 to <1/10). Developing non-skin irritation materials that reduce edema while optimizing patient comfort could help avoid these common adverse effects, and ultimately improve patient compliance.
Methods
A novel polymer based on high performance nylon which incorporates and maintains zinc in its ionic form at molecular level was developed. The polymer was converted into different forms with different features without any additional coating, chemical or textile processing to: 1) offer design flexibility of compression devices and modern wound dressings, 2) provide non-cytotoxic antimicrobial material to not compromise healthy skin. Nonwoven and knit materials with this novel polymer were constructed and evaluated for antimicrobial efficacy against significant pathogens using industry stand ISO20743. Cytotoxicity was investigated using “MEM Elution Test”. To determine the irritation potential of a test material, each were evaluated against the skin and wear trial was conducted to verify real use application.
Results
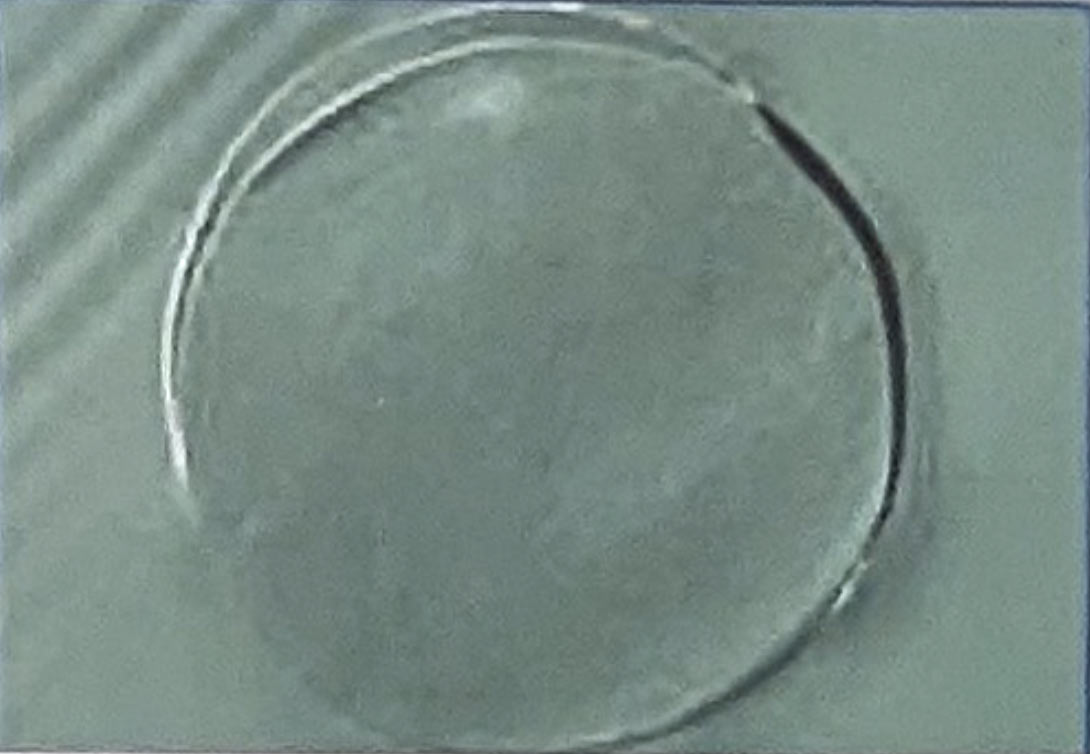
Novel Polymer: Zinc ions were embedded in the polymer matrix during the polymerization process. The active ingredient was distributed throughout the polymer matrix without forming particulates, as confirmed by SEM photos (Fig 1). This process is possible due to the chemical compatibility between zinc ions and monomers used to produce nylon 6,6.
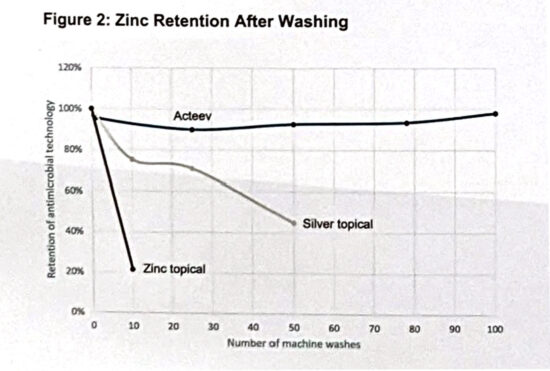
Different form factors were constructed for easy incorporation into compression devices or wound dressings. Unlike topical coated products, zinc retention analyses per inductively coupled Plasma Spectroscopy demonstrated that the embedded zinc ions remain in the polymer after >100 machine wash cycles per AATCC protocol (Fig 2).
The antibacterial efficacy varied depending on the forms used and bacterial species. This technology demonstrated high effectiveness against most of the strains tested in both sensitive and multidrug resistant isolates: Log 1-Log 8 according to ISO20743. Antibacterial efficacy was similar after washing for most strains tested, with exceptions observed.
 Skin Testing: Material A, B, and knitted fabric with 8% spandex (Material C) show non-cytotoxicity during MEM elution test using L929 cells. The same materials were utilized in a Human Repeat Insult Patch Test to evaluate skin irritation: 100% of the subjects showed no reaction according to the International Contact Dermatitis Research Scoring Group scale (Fig 3).
Skin Testing: Material A, B, and knitted fabric with 8% spandex (Material C) show non-cytotoxicity during MEM elution test using L929 cells. The same materials were utilized in a Human Repeat Insult Patch Test to evaluate skin irritation: 100% of the subjects showed no reaction according to the International Contact Dermatitis Research Scoring Group scale (Fig 3).
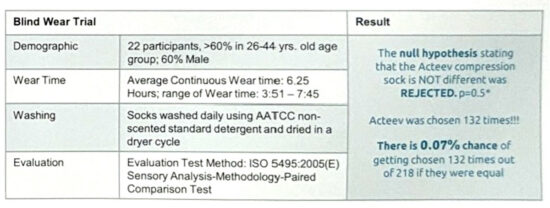
Wear Trial: A compression sock with zinc ion embedded texturized nylon 6.6 yar was constructed and confirmed antibacterial efficacy. In the same facility, they constructed a sock with their current yarn. The null hypothesis stating that the Acteev compression sock is not different was rejected (P=0.5) (Fig 4).
Conclusion
 Substrates using novel zinc ion embedded nylon antimicrobial technology demonstrate that zinc can be incorporated into the polymer, reducing chemicals in the finishing process. These materials have been dermatologically tested. This technology can potentially reduce irritation associated with compression devices, offer odor protection and keep products clean from bacteria. Wear trials indicate a significant preference for this technology over controls. Further study in a clinical setting is recommended.
Substrates using novel zinc ion embedded nylon antimicrobial technology demonstrate that zinc can be incorporated into the polymer, reducing chemicals in the finishing process. These materials have been dermatologically tested. This technology can potentially reduce irritation associated with compression devices, offer odor protection and keep products clean from bacteria. Wear trials indicate a significant preference for this technology over controls. Further study in a clinical setting is recommended.
Source: Acevedo C, Crane I, Kankaliya R, Mondal R, Ortega A, Urman K. Natural Zinc Ion Based Technology for Next Generation Compression and Wound Care Devices. Poster presented at 2023 Union of International Phlebotomy World Congress and 37th Annual Congress of the American Vein & Lymphatic Society. Sept. 17-21, 2023; Miami Beach, FL.