By Windy Cole, DPM, CWSP
A crucial component of wound management is regular debridement. The goal of debridement is the removal of all necrotic, fibrous, and devitalized tissue from the wound bed. Devitalized tissue in wounds produces a physical barrier to formation of new tissue and therefore decreases healing rates. If devitalized tissue remains in the wound bed, bacterial colonization is more likely. Standard of care remains that unhealthy tissue be sharply debrided to bleeding tissue to 1) allow for visualization of the extent of the ulcer, 2) to detect underlying exposed structures, deep bacterial contamination, or abscesses, and 3) to assess the quality of the peri-wound tissue. Frequent and thorough debridement reduces bacterial bioburden. In some cases, although the debridement adequately removes devitalized tissue, the remaining wound bacteria may become problematic.
Bioburden is an all-encompassing term that includes necrotic material, non-viable tissue, wound exudate, as well as bacteria and other microbes (eg, fungi). Bioburden tends to continually accumulate in chronic wounds because of the underlying pathogenic abnormality caused by systemic conditions such as diabetes or venous disease. The inability to fully resolve these fundamental physiologic issues makes chronic wound bed management with aggressive and complete debridement even more crucial.
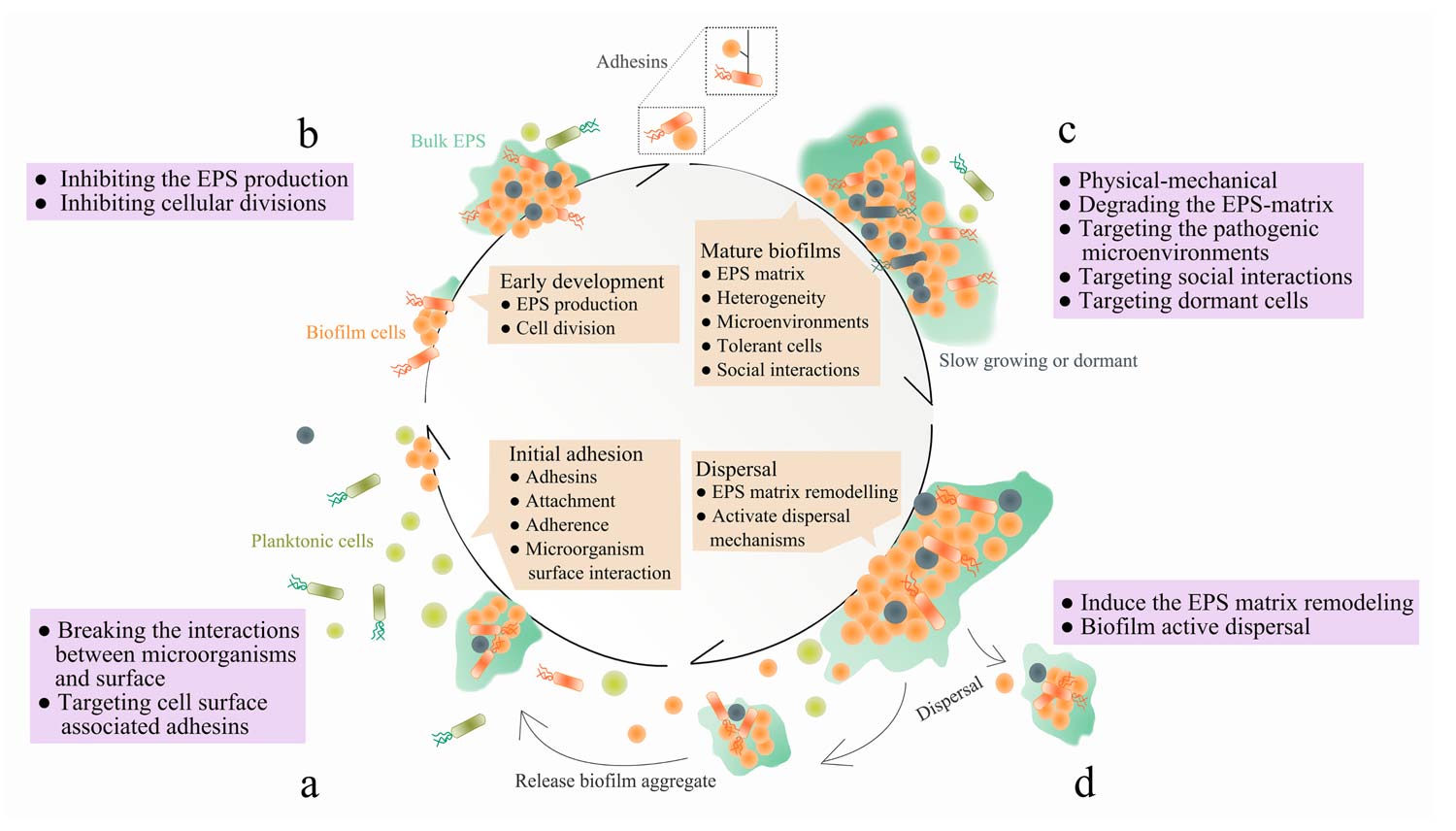
Figure. Four stages of biofilm formation: a) “initial adhesion”—microorganisms bind to host or medical device surfaces through cell surface associated adhesins; b) “early biofilm formation”—cells begin to divide and produce extracellular polymeric substance (EPS) to enhance adhesion, while forming matrix that embeds the cells; c) “biofilm maturation”—EPS matrix develops 3-D structures which is multi-functional and protective, allowing heterogeneous chemical and physical microenvironments to be formed where microorganisms co-exist within polymicrobial and social interactions; d) “dispersal”—cells leave the biofilm, returning to the planktonic phase. Therapeutic interventions can be targeted at each stage of biofilm development. a) For example, the initial phase of biofilm formation can be disrupted by breaking the interactions between microorganisms and surface, through targeting cell-surface associated adhesins. b) The early stages of biofilm development can be disrupted by inhibiting the EPS production and cellular divisions. c) Mature biofilms can be removed by physical-mechanical approaches, degrading the EPS-matrix, targeting the pathogenic microenvironments and social interactions within polymicrobial biofilms, as well as eliminating dormant cells. d) Induce the EPS matrix remodeling or biofilm active dispersal events. From: Jiang Y, Geng M, Bai L. Targeting biofilms therapy: current research strategies and development hurdles. Microorganisms. 2020;8(8):1222. Use is per CC BY.
Biofilm management however is a more complicated problem. Biofilm is an assemblage of surface-associated microbes enclosed in a self-produced matrix. Identifying and managing biofilm have recently become two of the most important aspects of wound care. The US Centers for Disease Control and Prevention and the National Institutes of Health have estimated that 65% – 80% of infections are caused from biofilms. Generally, it is believed that biofilms develop in stages (Figure). The initial stage is composed of small communities of bacteria that begin to attach to the wound surface. These polymicrobial colonies are commonly composed of bacteria, fungi, and other microorganisms. Biofilm is most susceptible to debridement and topical therapies at this stage. Unlike planktonic bacteria, biofilm bacteria form attachments with one another. At this juncture, the community has formed more permanent attachments to the wound surface, creating a more cohesive symbiotic community. They then can share information and gene-expression through a cell–cell communication mechanism called quorum sensing. Finally, biofilm colonies will begin to secrete a protective glycocalyx that also adheres to the wound surface. This entire process typically occurs in 2 to 4 days, unless disrupted. This extra polymeric substance is difficult to penetrate with systemically administered antibiotics and topical therapies.
Mature biofilms house mostly senescent bacteria that function at a lower energy state than active planktonic bacteria. As biofilms continue to evolve, they continue to change their phenotype and they share their resistance to antibiotics with the community. These factors make effective biofilm elimination a complicated matter. Even with thorough debridement biofilms may persist. These bacterial colonies have a significant impact on wound healing by causing prolonged inflammatory responses in the patient as well as contributing to acute bacterial infections.
Wound care clinicians must understand the best practices in biofilm management to be successful. Debridement is still thought to be the most effective way to remove biofilm. Even after an aggressive debridement biofilm can reform in as little as 24 hours. It is unlikely that complete removal of biofilm can be achieved with debridement alone. Debridement only temporarily eliminates biofilms. Employing adjunctive therapies in addition to regular debridement is often necessary. Research has shown that the best window of opportunity for biofilm prevention exists directly following debridement up to 24 hours. Biofilm is at its most susceptible and data illustrates that topical therapies are most effective during this period. Optimizing the effects of regular debridement utilizing a multi-tiered treatment approach is the most promising way to win the biofilm struggle.
Windy Cole, DPM. CWSP, is an adjunct professor and Director of Wound Care Research at Kent State University College of Podiatric Medicine. Dr Cole also serves as the National Director of Professional Development and Clinical Education for Woundtech. She is board certified by the American Board of Foot and Ankle Surgery and the American Board of Wound Management. She is a wound care advocate on the forefront of wound research and was the 2020 World Union of Wound Healing Silver Medal Award recipient for her work in Technology-driven Research. And she is a member of the LER Editorial Advisory Board.






