By Laura Fonda Hochnadel
According to 2004 figures, about 6 million people suffer from chronic wounds due to bedsores and vascular, inflammatory, and rheumatologic sources, with medical costs for this care reaching almost $10 billion.* Opioids and non-opioid treatments are often used to manage the pain associated with these chronic wounds but come with issues such as the potential for addiction or abuse. Patients taking non-opioids might even reach a point whereby greater dosages do not reap improved pain management. Topical solutions, such as lidocaine, offer an alternative to ingested medications. However, lidocaine, which has been on the market for about 70 years, has its own shortcoming in that it must be reapplied every 3 to 8 hours.
The purpose of this study was to the test the efficacy of a 4% lidocaine/TRI-726 (MilanaPharm, Montgomery, AL) gel to alleviate wound pain in a sustained manner. TRI-726 is a biocompatible hydrogel used to deliver drugs topically, with sustained release. The drug to be delivered to the patient is mixed with this viscous solution, which is triggered by body temperature to change to a gel form.
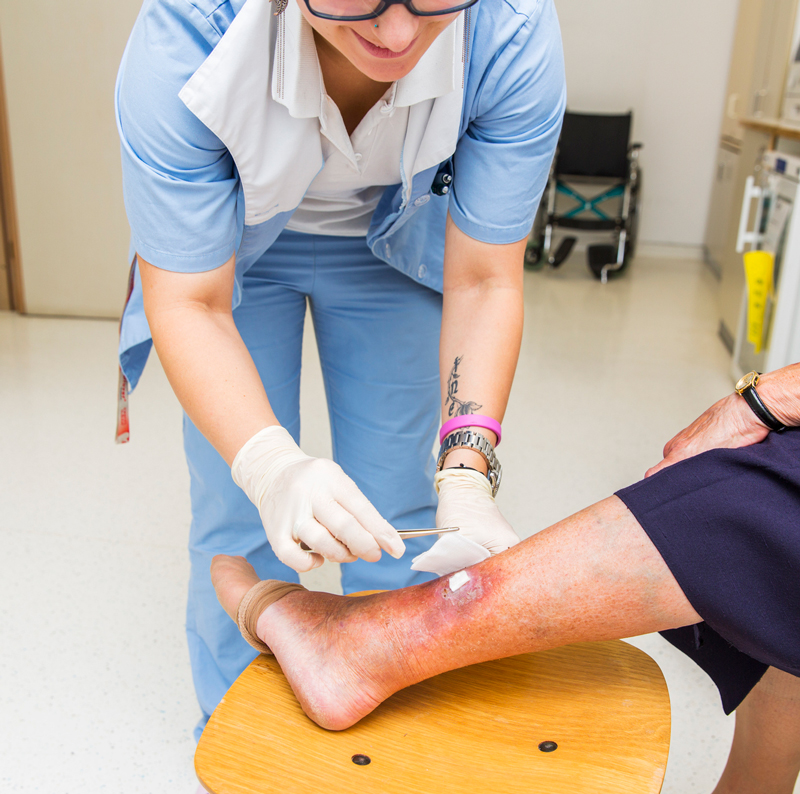
The cohort comprised 14 men and 19 women with painful chronic wounds, 5-50cm2 and judged not to require debridement. Ages ranged from 53 to 89 years. The chronic wounds included venous leg, diabetic foot, pressure, and vascular ulcers, a traumatic wound, an abdominal wound, and second-degree burns. The wounds were cleaned, dried, covered with a thick layer of the 4% lidocaine gel, and remained bandaged for 7 days.
The study participants kept a diary to record the effect of the 4% lidocaine gel. At the onset of the study, 6 patients indicated their pain was at a level 10, 18 patients reported pain at level 6 to 9, and 9 patients reported pain at level 3 to 5. No patients reported pain at the 0 to 2 level.
Over the course of the week, 2 patients did not experience any pain relief. By day 2, 95% continued to feel pain relief, and by day 3, 77% of the cohort continued to feel pain relief. Further, 58% continued to feel pain relief for up to 4 days. According to the study authors, “Patients’ comfort level and acceptance of the new lidocaine gel were very high throughout the study. No adverse actions were reported….” Further studies are warranted to examine long-term use and study the gel’s affect in a larger cohort.
Source: Treadwell T, Walker D, Nicholson BJ, Taylor M, Alur H. Treatment of pain in wounds with a topical long acting lidocaine gel. Chronic Wound Care Manag Res, 2019; 2019(117-121).
Laura Fonda Hochnadel is Associate Editor of LER.
*Editor’s note: Treadwell et al cites data from 2004. More current data from a 2018 retrospective analysis of the Medicare 5% Limited Data Set for 2014 identified approximately 8.2 million Medicare beneficiaries had wounds with or without infections and that Medicare costs ranged from $28.1 billion to $96.8 billion. The retrospective analysis, by SR Nussbaum et al., appears in the January 2018 issue of Value Health.