By Dr. Jennifaye V. Brown
Introduction
 The somatosensory system is responsible for processing information from the skin and the musculoskeletal system to determine body position and movement. Particularly for the lower extremity when impaired by stroke, somatosensory loss is detrimental to walking which is considered as the highest level of function to be achieved post-stroke.1-4 Somatosensory information is associated with vertical perception and the subsequent ability to balance oneself and therefore, impairment to this system may increase the risk of falls and limit the ability of safe walking.5-6 Often, the focus is on motor recovery post-stroke; however, somatosensory information is critical for motor output.7-9 The focus of this article is to review standardized measures used for testing somatosensation post-stroke and more importantly, interventions to optimize processing of sensory neurons in the penumbra and those not impacted by the stroke.
The somatosensory system is responsible for processing information from the skin and the musculoskeletal system to determine body position and movement. Particularly for the lower extremity when impaired by stroke, somatosensory loss is detrimental to walking which is considered as the highest level of function to be achieved post-stroke.1-4 Somatosensory information is associated with vertical perception and the subsequent ability to balance oneself and therefore, impairment to this system may increase the risk of falls and limit the ability of safe walking.5-6 Often, the focus is on motor recovery post-stroke; however, somatosensory information is critical for motor output.7-9 The focus of this article is to review standardized measures used for testing somatosensation post-stroke and more importantly, interventions to optimize processing of sensory neurons in the penumbra and those not impacted by the stroke.
Clarifying and Defining the Somatosensory System
Somatosensation comprises tactile sensation (commonly assessed as pin prick, light touch, and dull), pressure, proprioception, and pain.10-11 Specifically, Merkel cells detect light touch, pressure (indentation), and texture in the soles of the feet.11-12 Proprioception and kinesthesia have been commonly used interchangeably but they are distinctly different.13-14 Proprioception is the ability of sensory receptors to provide the central nervous system (CNS) with information regarding posture, position or kinetics. These proprioceptors are in the muscles (muscle spindle), tendons (Golgi tendon body), and joints, specifically the fibrous capsules (Ruffini corpuscles-detecting joint position and angle; slow responding and Pacinian corpuscles-detecting joint movement and speed; fast acting).11,14 The stroke itself does not directly impact these structures in the periphery but rather structures by which impulses travel from these sensory organs to get processed in the brain. Kinesthesia is being aware of the body movement in space.13-14 Movement quality as represented by duration, direction, amplitude, speed, acceleration, and timing across joints should be detected.15 For purposes of gait-focused interventions, healthcare practitioners (HCPs) should assess somatosensory impairments that focus on tactile sensation, pressure, proprioception, and kinesthesia.
Assessment
For the best guided treatment interventions, assessment should take into consideration stroke lesion location, size, and severity. One would not perform a detailed tactile sensory exam if the lesion was in the lateral hemisphere of the cerebellum. Research indicates that the Semmes Weinstein Monofilament Test (loss of protective sensation in diabetic neuropathy–pressure), the sensory subscale of the Fugl-Meyer Assessment (light touch and proprioception), and the Nottingham Sensory Assessment (light touch, pin prick, pressure, tactile localization, bilateral simultaneous touch, and proprioception–the latter is not done in the foot) are used with individuals with stroke but have nuanced psychometric problems that render them problematic.16-17 However, the revised version of the Nottingham Sensory Assessment (EmNSA) which includes the assessment of toes has been cited for being an optimal assessment tool.18-19 Individuals with stroke must be able to understand and respond to the directions of the HCP and assume the position or be placed in the position for the test. Furthermore, the HCP must perform the test consistently each and every time. Which may mean, for example, supporting or positioning the lower extremity and how the toe or foot is held for an assessment of joint proprioception or kinesthetic awareness. HCPs perform what they have been taught and document as absent, impaired (decreased [hyposensitive] or increased [hypersensitive] for tactile sensation) or intact. Rarely is “impaired” documented with descriptive markers that could be a part of goal setting for treatment, such as time to respond to a stimulus. One has to preface during the exam, what may be the cause of the delay, such as cognition or mode of response (verbal or nonverbal). If tactile sensation, pressure, and proprioception are impaired, the latter requiring the activation of isometric, eccentric, and concentric muscle activity in weightbearing (WB) and nonweightbearing (NWB) positions during walking to maintain balance, then interventions should target these sensory systems.
Treatment Interventions–Practical Applications to Improve Gait
There is a 3-step process known as priming, augmentation, and practice which can be utilized to drive motor output through somatosensory treatment interventions.20 Priming is the step of preparing the sensorimotor system prior to tasks-specific training (practice).20 Augmentation is combining somatosensory intervention to task-specific training in order to enhance the feedback of tactile and pressure stimulation.20-22 In this case, priming will be through 2 specific sensory techniques known as mobilization and tactile stimulation (MTS).23 However, I do incorporate task-specific training of functional activities prior to gait (practice).24
PRIMING
Mobilization: The premise is to stabilize the hindfoot with the foot and ankle placed in the available range of close chain dorsiflexion and have the person:
- Complete the horizontal phase of sit to stand holding various positions 3 to 5 seconds through the range of trunk movement forward and backward
- Unweight the buttocks during scooting forward and backward and hold positions in the increment of ranges for both activities 3 to 5 seconds
- Come to partial stand clearing the buttocks from the surface 1 to 5 inches and holding the position at various heights 3 to 5 seconds; I place different diameter objects under the buttocks to assure there is a change in height difference for lift off
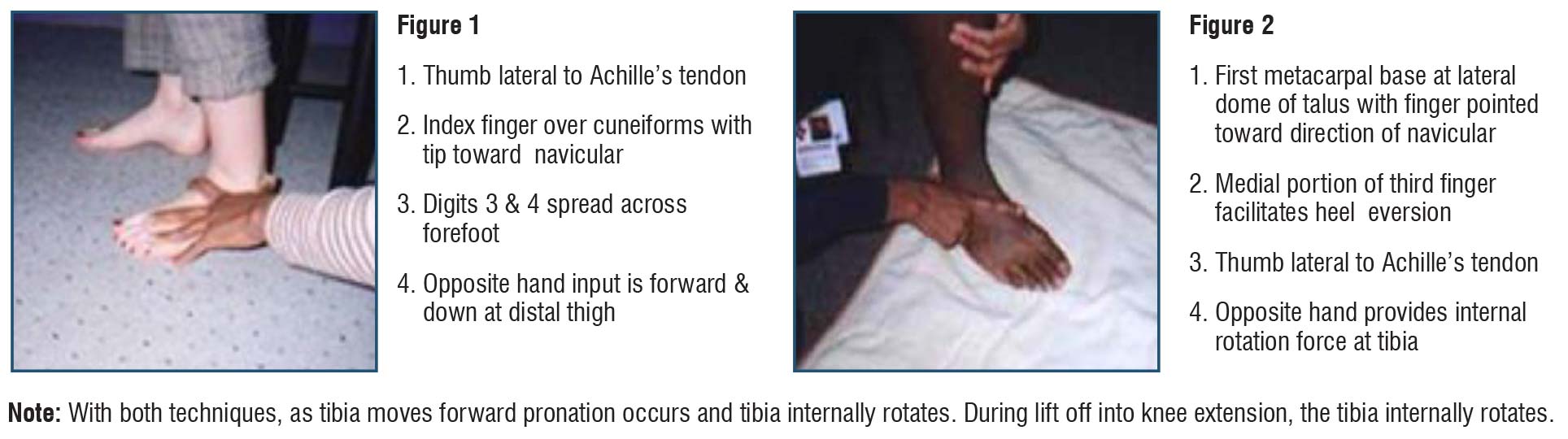
These activities increase load on the ankle joint to stimulate proprioceptors and movement through a variety of muscle contractions. Load and movement enhance balance reactions and ankle strategies. Muscles of the leg and foot are activated isometrically, eccentrically, and concentrically during various movements. As more range of motion is gained at the talocrural joint to increase close change dorsiflexion in sitting, this simulates what is required for sit to stand and terminal stance during gait (10˚–20˚).25-26 The key difference is that proprioceptive assessment is done in open chain whereas this treatment intervention focuses on close chain to prepare for the stance phase of gait in which the body weight has to come from behind to in front of the ankle. See Figures 1 and 2.
Tactile Stimulation: Individuals with stroke have impaired tactile stimulation which can be hyposensitive or hypersensitive. I heighten or calm these impaired sensations of touch with the use of cloths on the sole of the foot. I start in NWB rubbing the sole of the foot as if the person is drying their foot with a towel in a tailor sit position or the heel supported on a stool. Then I cut the different materials in the shape of the insole of the shoe worn the most. I have them put on both shoes without socks and add the material to the hemi foot.
AUGMENTATION
Then the person completes weight shifting activities as described above. This added pressure and proprioceptive input using a tactile stimulus may and often improves the perception of maladaptive tactile stimulation depending on lesion location, size, and severity. Then I repeat the sequence with the material in the less impaired or unaffected foot. I ask at the end of the session does the material feel the same under both feet and if different, how so. I document the intensity of noxiousness on a scale of 0–10 with the goal of it to decrease as somatosensation improves. See Figure 3.
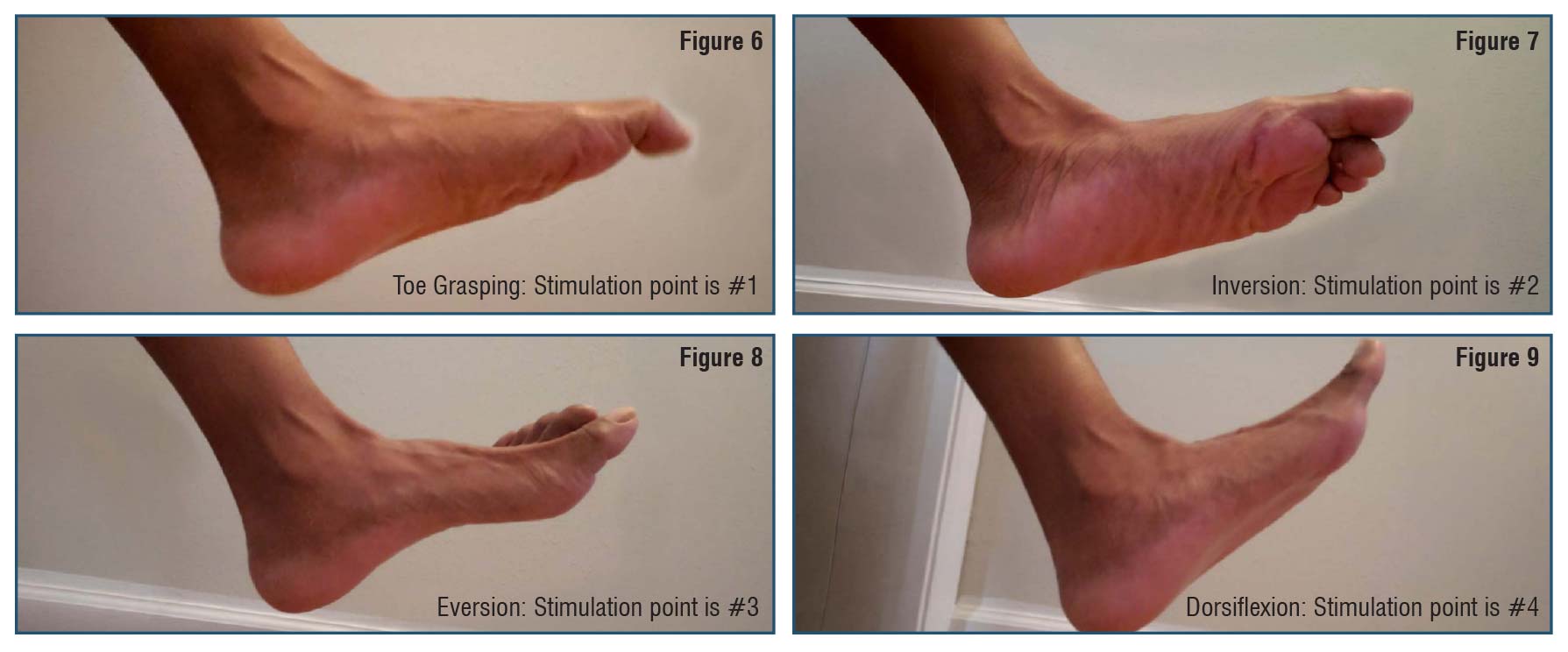
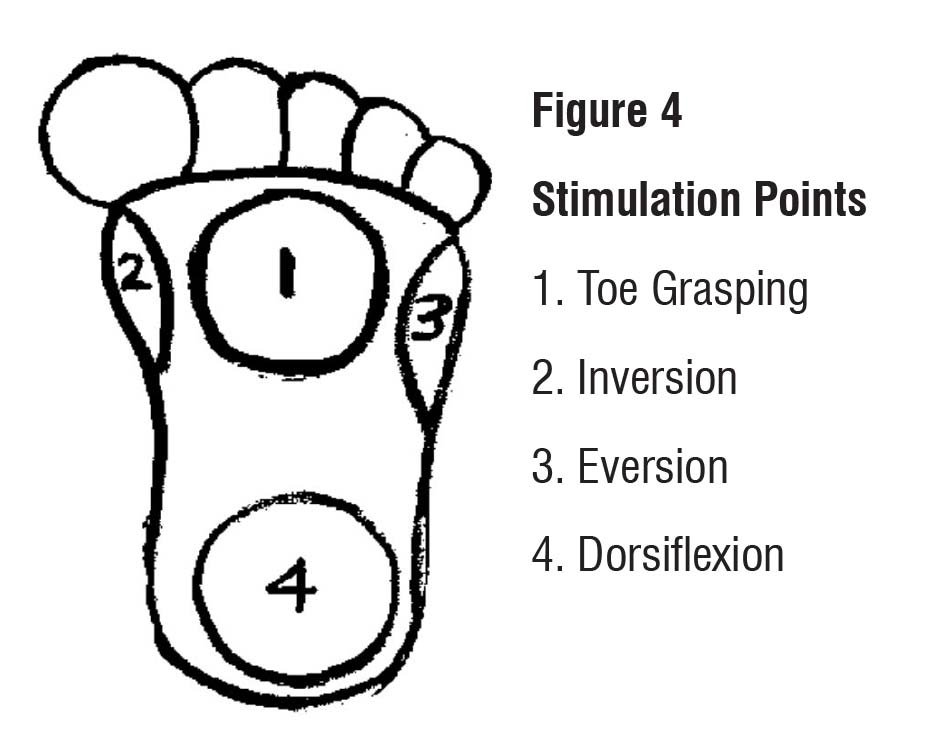
I often test for pathological reflexes of the foot as described by Duncan.27 See Figure 4. As seen on the insole in Figure 5, I denoted stimulation points for toe grasping, inversion, eversion, and dorsiflexion among other things such as the location of the navicular bone and the lateral arch in proximation to the cuboid. For example, if the foot tends to invert when stimulated in WB, I add a stimulus (glue a bead the size of a split pea) under the base of the 5th metatarsal to stimulate eversion bringing the foot to neutral which will allow for pronation as the tibia moves forward. See Figures 6–9.
PRACTICE
 Now on to task-specific training of walking, I simply follow the principles of gait training that simulate the literature in this article to drive motor recovery.
Now on to task-specific training of walking, I simply follow the principles of gait training that simulate the literature in this article to drive motor recovery.
- The hemi lower extremity is in WB and I stabilize the ankle as previously described as the opposite extremity steps backward, forward and perhaps to increase intensity, the foot is placed on steps of different heights in front of the hemi foot. To further build on intensity, the steps are placed further away from the hemi foot requiring more dorsiflexion of the ankle and eccentric activity of the calf muscles.
- The stepping sequence is repeated with different types of paired cloths on the insole of the shoes. During this phase of activities, I add:stepping out to the side with the less affected lower extremity
- facilitated hemi heel rise isolating terminal stance (30˚ metatarsal head [MTH]) and preswing (60˚ MTH hyperextension with 40˚ of passive knee flexion) of the hemi lower extremity
- stepping backward with hemi lower extremity first focusing on loading the toes then heel
- walking short distances on different flooring; I have rolled reams of carpet that may be similar to that which is in the home
- incline walking up and down, sideways, and backward
- stair training step over step up and down
- I conduct intermittent assessment of somatosensory system impairments to demonstrate any improvement, which in turn will guide treatment intervention modifications. At this time, I also assess gait speed (10-foot walk test, 5- or 10MWT) and gait quality (Rancho Los Amigos Medical Center, Physical Therapy Department Gait Analysis: Full Body).28
If you are asking what should be done if a therapist is not present to do the hands-on mobilization? I have an answer. Get ready to read part 2: Sensory Input is Needed for Motor Output–Self Work for the Best Foot Work Matters.
If you have any questions regarding this article, leave them in the comment box.
Jennifaye V. Brown, PT, MSPT, PhD, NCS, CAPS is an American Physical Therapy Association 4-time 10-year board certified neurologic physical therapist in Charleston, South Carolina, specializing in stroke rehabilitation, specifically gait analysis and treatment, AFO design, and the redesign of lived spaces allowing individuals with disabilities to age in place. She is the author of the book, Brace Yourself: Everything You Need to Know About AFOs After Stroke.
- Gorst T, Rogers A, Morrison SC, et al. The prevalence, distribution, and functional importance of lower limb somatosensory impairments in chronic stroke survivors: a cross sectional observational study. Disabil Rehabil. 2018;41:2433–2450. doi: 10.1080/09638288.2018.1468932
- Tyson SF, Crow JL, Connell L, Winward C, Hillier S. Sensory impairments of the lower limb after stroke: a pooled analysis of individual patient data. Topics Stroke Rehabil. 2013;20:441–449. doi: 10.1310/tsr2005–441
- Sánchez-Blanco I, Ochoa-Sangrador C, López-Munain L, Izquierdo-Sánchez M, Fermoso-Garcia J. Predictive model of functional independence in stroke patients admitted to a rehabilitation programme. Clin Rehabil. 1999; 13:464–475. doi: 10.1191/026921599672994947
- Jette DU, Latham NK, Smout RJ, Gassaway J, Slavin MD, Horn SD. Physical therapy interventions for patients with stroke in inpatient rehabilitation facilities. Phys Ther. 2005;85:238–248.
- Saeys W, Vereeck L, Truijen S, et al. Influence of sensory loss on the perception of verticality in stroke patients. Disabil Rehabil. 2012; 34(23): 1965–1970.
- Duysens J, Massaad F. Stroke gait rehabilitation: Is load perception a first step towards load control? Clin Neurophysiol. 2015; 126 (2): 225–226. doi.org/10.1016/j.clinph.2014.07.001
- Borich MR, Brodie SM, Gray WA, Ionta S, Boyd LA. Understanding the role of the primary somatosensory cortex: Opportunities for rehabilitation. Neuropsychologia. 2015;79(Pt B):246-255. doi:10.1016/j.neuropsychologia.2015.07.007
- Zhao M, Marino M, Samogin J, Swinnen SP, Mantini D. Hand, foot and lip representations in primary sensorimotor cortex: a high-density electroencephalography study. Sci Rep. 2019;9(1):19464. Published 2019 Dec 19. doi:10.1038/s41598-019-55369-3
- Cardellicchio P, Hilt PM, Dolfini E, Fadiga L, D’Ausilio A. Beta Rebound as an Index of Temporal Integration of Somatosensory and Motor Signals. Front Syst Neurosci. 2020;14:63. Published 2020 Sep 2. doi:10.3389/fnsys.2020.00063
- Carey LM. Stroke Rehabilitation Insights From Neuroscience and Imaging. Oxford University Press; 2012.
- Wang L, Ma L, Yang J, Wu J. Human Somatosensory Processing and Artificial Somatosensation. Cyborg Bionic Syst. 2021;2021:9843259. Published 2021 Jul 2. doi:10.34133/2021/9843259
- Abraham J, Mathew S. Merkel Cells: A collective review of current concepts. Int J Appl Basic Med Res. 2019;9(1):9–13. doi:10.4103/ijabmr.IJABMR_34_18
- Rosker J, Sarabon N. Kinaesthesia and methods for its assessment Sport Sci Rev. 2021;19:165-208. Doi: 10.2478/v-0237-011-0037-4
- Mukhopadhyay K. Proprioception and kinesthesia: The sixth sense organ. Adv Health Exerc. 2021;1(1), 12–17.
- Shakoor N, Agrawal A, Block JA. Reduced lower extremity vibratory perception in osteoarthritis of the knee. Arthritis Rheum. 2008;59(1), 117–121.
- Chia FS, Kuys S, Low Choy N. Sensory retraining of the leg after stroke: systematic review and meta-analysis. Clin Rehabil. 2019;33(6):964–979. doi:10.1177/0269215519836461
- Lin JH, Hsueh IP, Sheu CF, Hsieh CL. Psychometric properties of the sensory scale of the Fugl-Meyer assessment in stroke patients. Clin Rehabil. 2004;18(4):391–397. doi:10.1191/0269215504cr737oa
- Stolk-Hornsveld F, Crow JL, Hendriks EP, van der Baan R, Harmeling-van der Wel BC. The erasmus MC modifications to the (revised) Nottingham sensory assessment: a reliable somatosensory assessment measure for patients with intracranial disorders. Clin Rehabil. 2006;20(2):160–172. doi:10.1191/0269215506cr932oa
- Wu CY, Chuang IC, Ma HI, Lin KC, Chen CL. Validity and responsiveness of the revised Nottingham sensation assessment for outcome evaluation in stroke rehabilitation. Am J Occup Ther. 2016;70(2):7002290040p1–7002290040p8. doi:10.5014/ajot.2016.018390
- Pomeroy V, Aglioti SM, Mark VW, et al. Neurological principles and rehabilitation of action disorders: rehabilitation interventions. Neurorehabil Neural Repair. 2011; 25 (5 Suppl):33S43S. doi:10.1177/1545968311410942
- Cruz-Almeida Y, Black ML, Christou EA, Clark DJ. Site-specific differences in the association between plantar tactile perception and mobility function in older adults. Front Aging Neurosci. 2014;6:68. Published 2014 Apr 11. doi:10.3389/fnagi.2014.00068
- Kavounoudias A, Roll R, Roll JP. The plantar sole is a ‘dynamometric map’ for human balance control. Neuroreport. 1998;9(14):3247–3252. doi:10.1097/00001756-199810050-00021
- Hunter SM, Crome P, Sim J, Donaldson C, Pomeroy VM. Development of treatment schedules for research: a structured review to identify methodologies used and a worked example of ‘mobilisation and tactile stimulation’ for stroke patients. Physiotherapy. (2006) 92:195–207. doi: 10.1016/j.physio.2006.01.001
- Gu J, Kim T-H. Effects of sit-to-stand exercise combined with joint mobilization with movement on lower extremity functions, balance, and gait performance in patients with chronic stroke. J Musculoskelet Sci Technol. 2025; 9(1):87–94. doi.org/10.29273/jmst.2025.9.1.87
- Adams JM, Cerny K. Observational Gait Analysis. A Visual Guide. SLACK Incorporated; 2018.
- Carr J, Shepherd R. stroke rehabilitation: guidelines for exercise and training to optimize motor skill. (2nd ed). Butterworth-Heinemann (Elsevier);2003.
- Duncan W. Tonic reflexes of the foot. J Bone Joint Surg Am. 1960; 42: 859–868.
- The Pathokinesiology Service & The Physical Therapy Department. Observational Gait Analysis Handbook. Los Amigos Research and Education Institute, Inc.; 2001