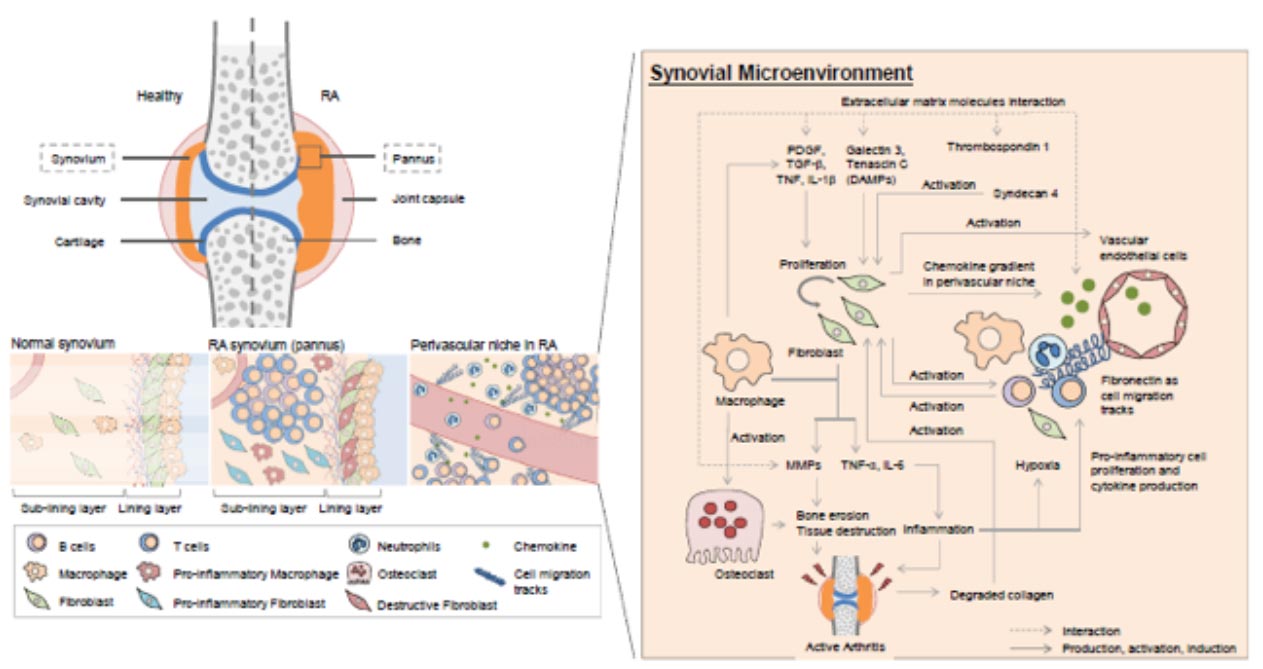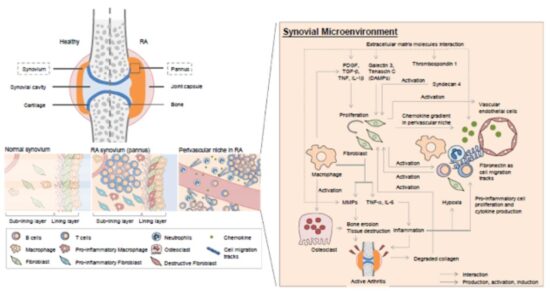
Figure. Synovial microenvironment in the healthy and RA joint. The synovium is composed of a lining layer and a sub-lining layer. Synovial fibroblasts and synovial macrophages constitute the lining layer. The sub-lining layer comprises a loose, highly innervated, and vascularized ECM with the residing cells. In addition to immune cells, synovial fibroblasts, synovial macrophages, and ECM molecules participate in the precipitation of chronic inflammation in the RA synovium. The ECM molecules also maintain the chronic inflammation in RA synovium. Thrombospondin 1, an ECM molecule with increased expression in RA, interacts with other ECM molecules and modulates their activity and function. The increasing galectin 3, tenascin C, and syndecan 4 activate synovial fibroblasts, which produce MMPs, TNF-a, and IL-6. TNF-a and IL-6 induce inflammation and further tissue hypoxia. On the other hand, the MMPs degrade the ECM and result in tissue destruction. The degraded collagen released from damaged tissue activates synovial fibroblast more efficiently than intact collagen. DAMP, damage associated molecular pattern; ECM, extracellular matrix; MMP, matrix metalloproteinases; RA, rheumatoid arthritis.
The recent advance in treatments for rheumatoid arthritis (RA) has significantly improved the prognosis of RA patients. However, these novel therapies do not work well for all RA patients. The unmet need suggests that the current understanding about how inflammatory response arises and progresses in RA is limited. Recent accumulating evidence reveals an important role for the tissue microenvironment in the pathogenesis of RA.
The synovium, the main tissue where the RA activity occurs, is composed by a unique extracellular matrix (ECM) and residing cells. The ECM molecules provide environmental signals that determine programmed site-specific cell behavior. Improved understanding of the tissue microenvironment, especially how the synovial architecture, ECM molecules, and site-specific cell behavior promote chronic inflammation and tissue destruction, will enhance deciphering the pathogenesis of RA. Moreover, in-depth analysis of tissue microenvironment will allow us to identify potential therapeutic targets.
Research is underway to explore potential candidates, both cellular and ECM molecules, to develop novel therapies. This article reviews recent advances in knowledge about how changes in cellular and ECM factors within the tissue microenvironment result in propagation of chronic inflammation in RA.
Source: Cheng CF, Liao HJ, Wu CS. Tissue microenvironment dictates inflammation and disease activity in rheumatoid arthritis. J Formos Med Assoc. 2022;121(6):1027-1033. doi: 10.1016/j.jfma.2022.01.026.