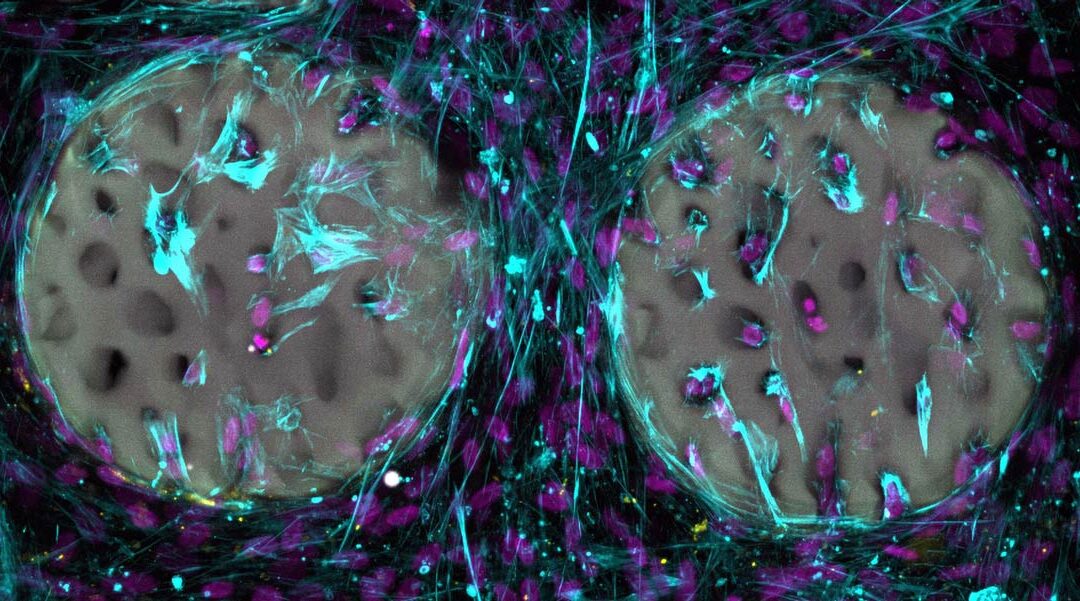
A promising sign: bone-forming cells (light blue) with nuclei (purple) have already colonized a hydrogel-based bone structure (gray). Image courtesy of Margherita Bernero/ETH Zurich.
If a broken bone is too severe to heal on its own or a bone tumor needs to be removed, surgeons insert an implant that enables the bone to grow back together. These implants often consist of pieces of the patient’s own bone, known as autografts, or metal or ceramic parts. A key drawback of many implants is that they require a second surgery to harvest the tissue for the autografts. Additionally, metal implants tend to be too rigid and may loosen over time, compromising stability. A successful repair of this nature depends on various cell types that must colonize the implant before forming new bone tissue. “For proper healing, it is vital that biology is incorporated into the repair process,” said Xiao-Hua Qin, professor of biomaterials engineering at ETH Zurich.
Toward this end, Qin, along with his team and ETH Professor Ralph Müller, has created a novel hydrogel that is as soft as jelly, dissolves gradually in the body, and could potentially be used for personalized bone implants. The hydrogel, which is made up of 97% water and 3% biocompatible polymer, is modeled on the natural bone healing process. To make it solidify, the researchers introduced 2 special molecules: 1 that links the polymer chains together and another that, when exposed to light, triggers the reaction.
Wanwan Qiu, Qin and Müller’s former doctoral student, developed the connecting molecule specifically for this application. The polymer chains are linked as soon as laser pulses of a certain wavelength hit the hydrogel. The irradiated areas immediately solidify, while the non-irradiated parts can be washed out later. In this way, the researchers can use the laser beam to print any shapes and structures into the hydrogel with very fine resolution and extreme precision. The structures can be as small as 500 nanometers.
In their study, the researchers created complex, structured hydrogels that resemble real bone and feature a fine network of bone trabeculae. They used medical imaging as a template. While the material has only been tested in a test tube, results showed that bone-forming cells rapidly colonize the structured hydrogel and begin forming collagen, a vital component of bone. The tests also confirmed that the material is biocompatible and does not damage the bone-forming cells. The researchers have patented the base material and plan to make it available to the medical industry with the goal for the hydrogel-based implant to be used in clinics to repair broken bones.