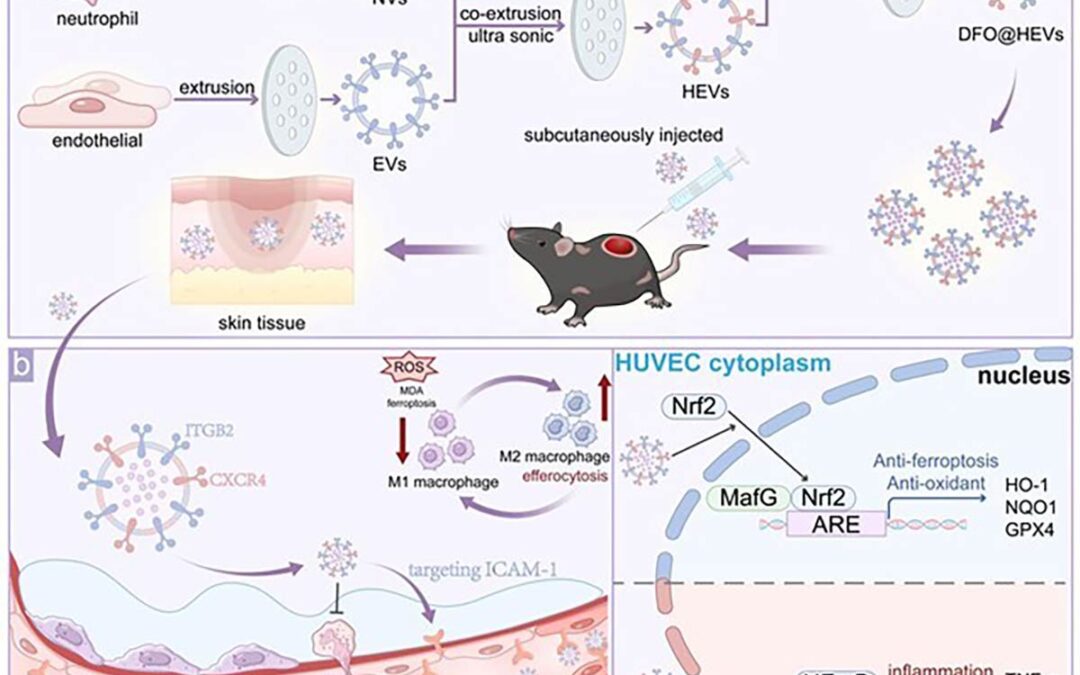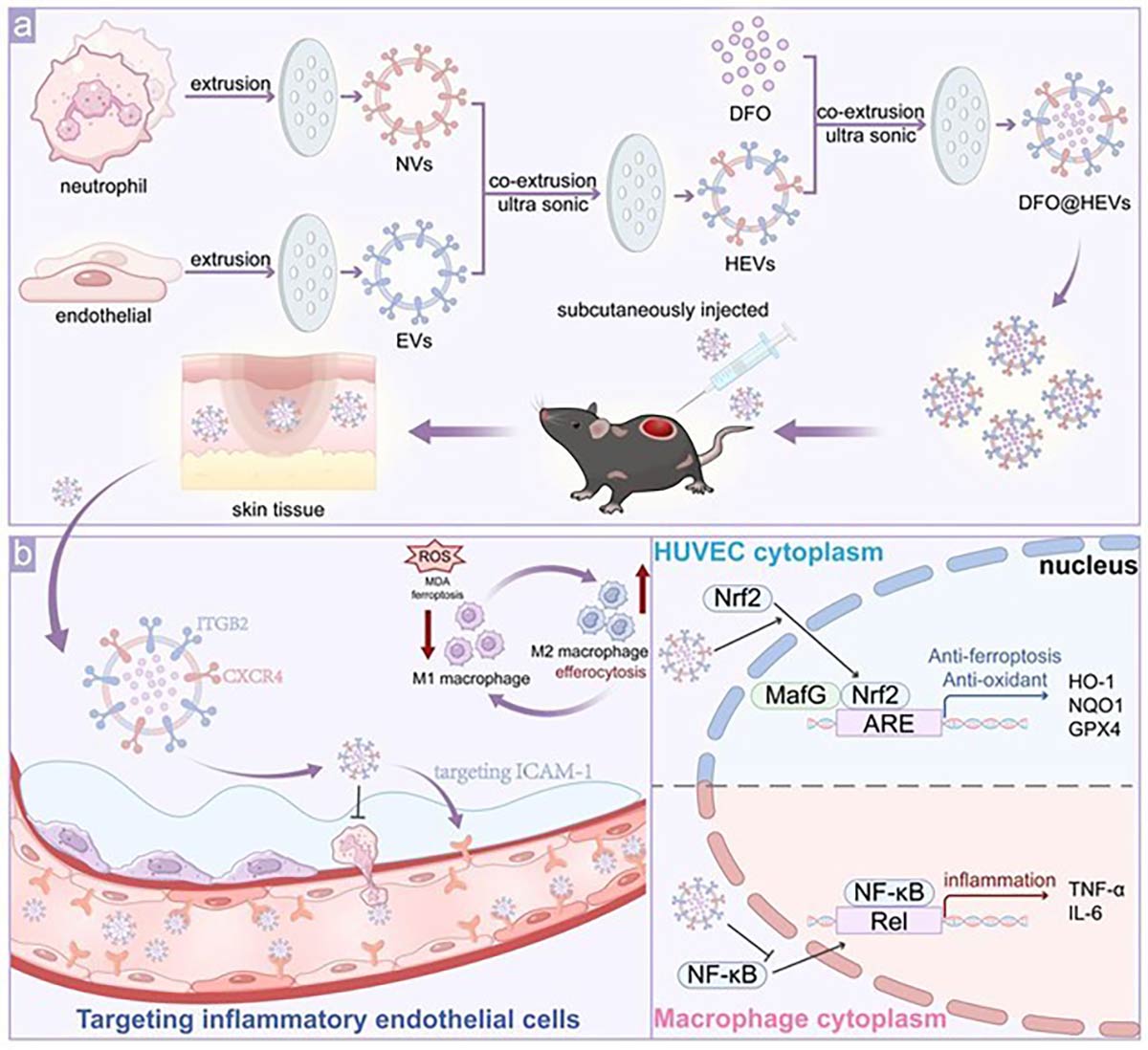
Schematic illustration of the design, targeting behavior, and therapeutic mechanisms of DFO-loaded hybrid extracellular vesicles in diabetic wound repair.
Researchers from Huazhong University of Science and Technology report a novel nanotherapeutic platform that accelerates diabetic wound healing by simultaneously targeting blood vessels and inflamed tissue. The research team engineered hybrid extracellular vesicles by merging vesicles derived from endothelial cells and neutrophils, then loading them with deferoxamine. By fusing endothelial- and neutrophil-derived membranes, the system achieves precise dual targeting while delivering antioxidant and anti-inflammatory signals–the nanovesicles homed to damaged blood vessels through CXCR4 signaling while simultaneously targeting inflamed tissue via β2-integrin interactions. Once delivered, the system addressed multiple pathological drivers of diabetic wounds.
In endothelial cells exposed to diabetic conditions, the nanovesicles restored cell survival, migration, and tube formation by activating the PI3K/AKT/HIF-1α pathway and boosting VEGF-mediated angiogenesis. At the same time, iron chelation suppressed lipid peroxidation and ferroptosis through Nrf2-dependent antioxidant responses. Immune regulation represented another critical advantage. The nanovesicles reduced neutrophil over-adhesion, shifted macrophages from a pro-inflammatory M1 state toward a reparative M2 phenotype, and enhanced efferocytosis, the clearance of dying cells that is essential for inflammation resolution.
In diabetic mouse wound models, treatment led to faster wound closure, thicker re-epithelialization, increased collagen deposition, and dense new blood vessel formation. Inflammatory cytokines, oxidative stress markers, and ferroptosis indicators were all significantly reduced, demonstrating a comprehensive remodeling of the wound microenvironment. This research highlights a new direction for treating chronic wounds by moving beyond single-target interventions. The hybrid nanovesicle strategy could be adapted to deliver other therapeutic agents or tailored for different inflammatory or ischemic diseases. Its strong biocompatibility and precise targeting suggest translational potential for diabetic foot ulcers, pressure sores, and other non-healing wounds.