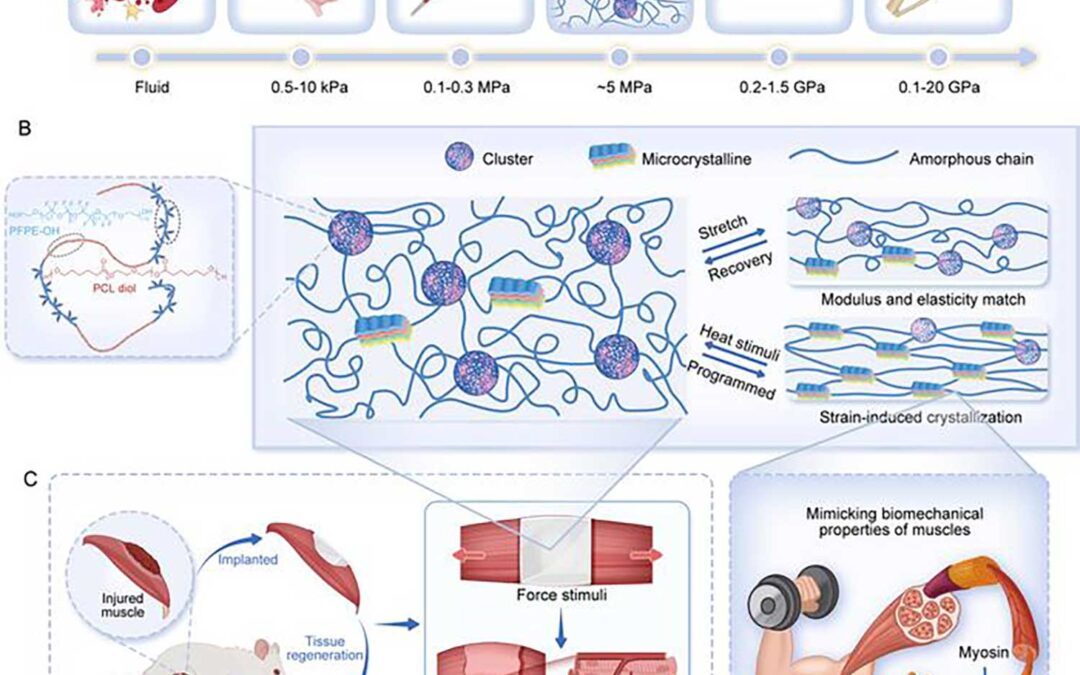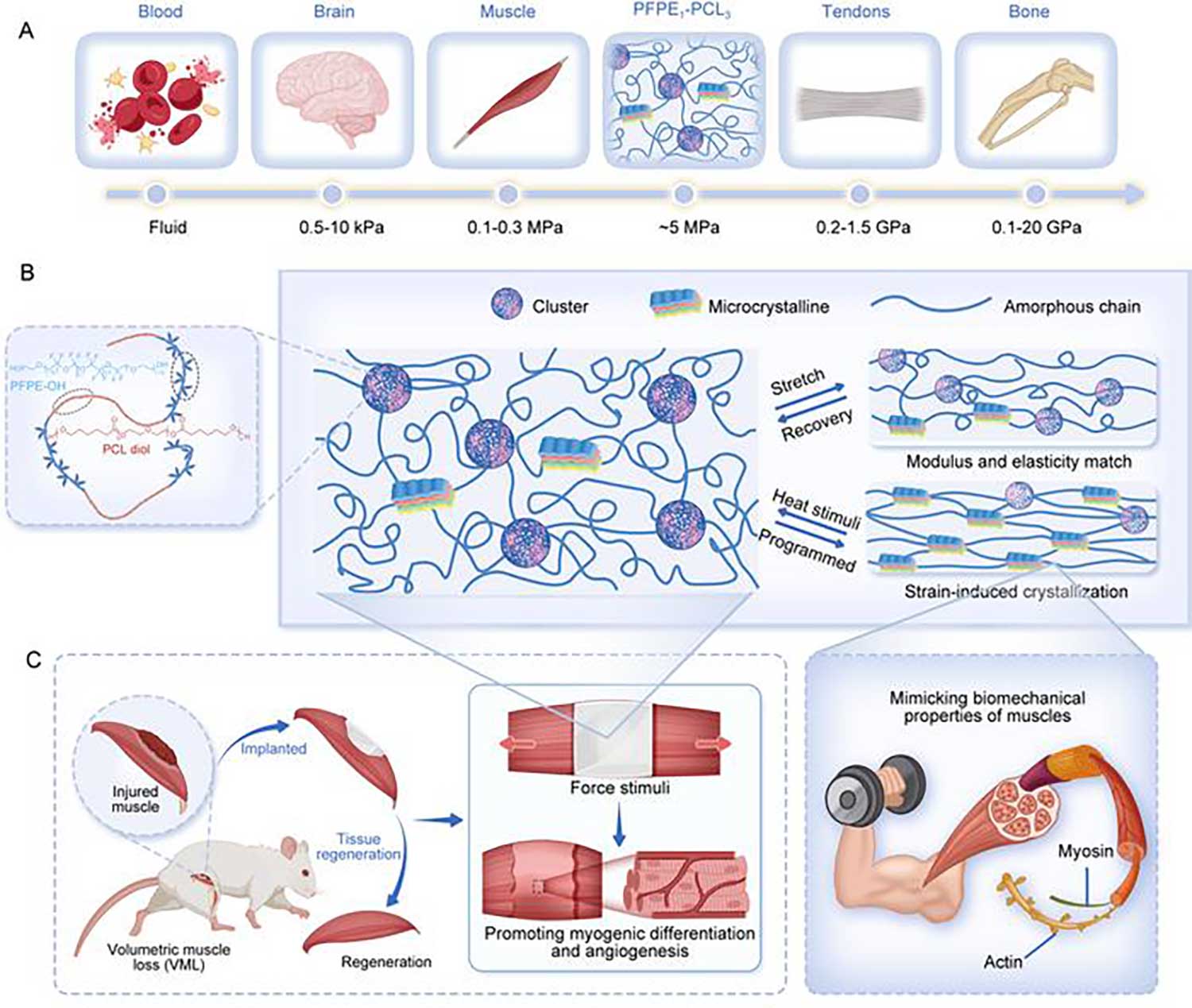
(A) A summary plot illustrating the elastic modulus range of the artificial muscle compared to representative biological tissues, highlighting the biomimetic mechanical properties of the artificial muscle. (B) A schematic representation of the designed multifunctional artificial muscle, showcasing its structural features tailored for enhanced functionality. (C) Demonstrations of the artificial muscle’s versatility: on the left, its ability to treat volumetric muscle loss by promoting myogenic differentiation and angiogenesis; on the right, its capacity to mimic the functional dynamics of natural muscle tissue.
Researchers from China have successfully developed an artificial muscle with mechanical properties similar to natural muscle tissue. They utilized molecular design to synthesize the artificial muscle through block copolymerization of biocompatible perfluoropolyether (PFPE) and polycaprolactone diol (PCL). By precisely tuning the intra- and intermolecular interactions of PFPE and PCL (eg, dipole-dipole interactions) under controlled conditions, they achieved self-assembly and microphase separation, suppressing the crystallization of PCL moiety. Their work has implications for use in prosthetic devices, tissue engineering, and regenerative medicine.
The biocompatible material exhibits an amorphous state at room temperature and without external force, maintaining a low elastic modulus and high elasticity. Under tensile stress, the amorphous polymer chains unfold, align, and reorient along the loading direction, significantly enhancing the material’s tensile strength and toughness. The material also demonstrates remarkable tear and puncture resistance as well as exceptional shape memory and actuation performance. Under thermal stimulation, the material can lift objects weighing more than 5,000 times its own weight and reliably perform reversible contraction and extension motions over multiple heating and cooling cycles.
Histological evaluations after implantation in rats revealed that the material facilitated muscle tissue growth along the scaffold. After 4 weeks, regenerated muscle exhibited well-organized structure and morphology, with muscle contractile force comparable to that of normal rats. Furthermore, vascular regeneration indicated enhanced angiogenesis, crucial for muscle regeneration.