 Stem cell therapy is rapidly gaining momentum as a clinical option for indications including meniscal injury, wound healing, and osteoarthritis. But high-level research on the technology’s efficacy and long-term safety is hard to come by.
Stem cell therapy is rapidly gaining momentum as a clinical option for indications including meniscal injury, wound healing, and osteoarthritis. But high-level research on the technology’s efficacy and long-term safety is hard to come by.
By Emily Delzell
The regenerative properties of adult stem cell therapies hold excellent potential for improving care in patients with difficult-to-treat musculoskeletal, orthopedic, and vascular conditions of the lower extremity, and research to date suggests the therapies have a good safety profile. Yet much of the data needed to bring these therapies into the world of evidence-based clinical medicine are lacking or in the early stages of investigation.
Despite the general scarcity of information from large randomized controlled trials, physicians in the US and around the world are administering adult stem cell therapies in research and clinical settings for a wide range of lower extremity indications including peripheral arterial disease (PAD), meniscus tears, and osteoarthritis (OA) pain.1-4
“I think stem cells have fantastic potential, and long term we’ll be able to realize that as we learn about safety and are able to culture these cells and manipulate them,” said Scott Rodeo, MD, an orthopedic surgeon at the Hospital for Special Surgery and professor of orthopaedic surgery at Weill Cornell Medical College in New York City, who uses bone marrow-derived stem cell injections to augment meniscus and bone defect repairs. “I think there’s great potential in any number of applications, including in trauma in tissue that has intrinsically poor healing capacity, like cartilage tendons, ligament, and even bone to some degree.”
Treatments can involve stem cells from a number of allogeneic and autologous sources. Allogeneic mesenchymal stem cells (MSCs), for example, can be derived from the placenta, umbilical cord, or umbilical cord blood. Autologous stem cells include endothelial progenitor cells sourced from bone marrow or peripheral blood, hematopoietic stem cells, adipose-derived stem and regenerative cells (ADRCs), and the most commonly used type, MSCs derived from bone marrow.4 Both the ideal type and number of stem cells needed in specific indications and individual patients remain unknown, and answering these questions is a high priority for researchers.

Chronic nonhealing lower limb ulcers with treated with autologous ADRCs before treatment (above) and 20 days after injection (below). (Images courtesy of Gerardo Marino, MD.)
Bone marrow-derived MSCs are the best characterized adult stem cell type in the literature and more than 100,000 US adults4 receive therapies each year in which clinicians use a point-of-care procedure to draw bone marrow aspirate from their iliac crest or another site, concentrate the aspirate in a centrifuge, and then inject or implant the cells into the treatment area.
The number of stem cells derived from such procedures varies, ranging from about one in 10,000 nucleated cells to one in 100,000,5 and numbers diminish with age and illness.6 Cells can also be expanded in culture to produce millions rather than thousands, a process that is highly regulated in the US (see sidebar, page 20).
Stem cells’ ability to replicate over long periods, as well as their extended life span and relative resistance to cell death, are the features that generate both the enthusiasm for their use and the most serious concern about their safety—the potential to spur malignant tumor growth. Animal studies and early results from human trials show little evidence for malignant transformation, and the vast majority of clinical trials report no major health concerns, but longer-term research is needed to evaluate the potential for tumor formation.7
Once delivered, these multipotent cells can differentiate into bone, cartilage, tendon, ligament, and endothelium.8 They also release a number of bioactive factors, changing the local environment to stimulate tissue repair and wound healing.9-12
Stem cells have been used for decades in procedures such as bone marrow transplant. But new knowledge and applications, along with medical device manufacturers that are creating various systems for harvesting, processing, and concentrating cells, have created a burgeoning field that is in many ways still in its infancy. Meanwhile, investigators are working to establish safety and efficacy, investigate the various technologies, and answer basic questions about stem cell selection, isolation techniques, delivery, and best uses.
Wound healing and PAD
Stem cells encourage wound healing by releasing chemokines and growth factors that promote vascular regeneration and neural survival and regeneration.17-20 Researchers in the US and around the world are studying stem cell products for treatment of diabetic foot ulcers (DFUs) and PAD.
In the US, stem cell therapy for DFUs and PAD is still experimental, said Sheila N. Blumberg, MD, a surgeon at NYU Langone Medical Center in New York City and lead author of a recent review in Diabetes Research and Clinical Practice on the potential role of these therapies in treating chronic foot ulcers.21
“Although animal studies and small clinical studies have demonstrated a tremendous potential for wound regeneration [in DFU], barriers to clinical use remain and include a lack of sufficient evidence to support use, high costs, and a lack of standardized techniques to deliver the stem cells in a meaningful fashion,” she said.
Systems for processing stem cells have been approved for clinical use in some nations, including those in Asia-Pacific and the EU, where Italian researchers reported in 2013 on the use of a commercial system for processing autologous cells.
Investigators at the Second University of Naples recruited 10 patients aged 60 to 70 years with long-term (~20 years) type 2 diabetes, chronic lower limb ulcers, and PAD.22 Patients’ ulcers hadn’t healed after five months of traditional treatment (cleansing and debridement), five months of advanced dressing application, and six months of hyperbaric oxygen therapy.
Investigators used minimally invasive liposuction to harvest abdominal fat, then isolated ADRCs with the commercial system, a process that takes about 2.5 hours. (US investigators are studying this system for use in ischemic heart failure and have recently begun enrollment for a 10-person safety and feasibility study of the therapy for hamstring tears. The manufacturer has also opened international registries to track outcomes of ADRC injection for hamstring injury and knee osteoarthritis pain.)
Investigators injected the concentrated cells around the edges of ulcers and followed patients for 90 days. The concentrate included adipose stem cells as well as endothelial cells, endothelial progenitor cells, vascular smooth muscle cells, and pericytes.
ADRC therapy produced complete healing in six patients and reduced ulcer diameter and depth and ulcer-related pain in all 10. According to lead investigator Gerardo Marino, MD, the four patients who didn’t heal completely had comorbidities, including lupus, severe lesions (total occlusion of the femoral and popliteal arteries), and vascular occlusion, that likely prevented total resolution of wounds.
“Given that we speak of chronic ulcers that do not heal with classic therapies, this study shows that they can be cured by the application of stem cells extracted from adipose tissue. I believe this is a good method for [treating] chronic ulcers as it is an autologous graft, without the risk of rejection,” Marino wrote in an email interview.
Bone marrow-derived stem cells have also shown wound-healing efficacy. A 2011 randomized controlled pilot trial conducted in China compared injection of bone marrow-derived MSCs, bone marrow-derived mononuclear cells, and saline into and around the wound site of 37 limbs of patients with type 2 diabetes, foot ulcers, and bilateral critical limb ischemia (CLI).23
Both stem cell types significantly increased blood flow and decreased pain compared with the control injection, but bone marrow-derived MSCs produced better results overall in terms of increased lower limb perfusion and healing compared with mononuclear cells. At six weeks postinjection the number of healing ulcers in the MSC group was significantly higher than in the mononuclear cell group (10 of 11 vs 5 of 11), which took four weeks longer to achieve healing in all limbs. During the 24-week follow-up period six limbs from the saline group underwent amputation, while none of the limbs treated with stem cells required amputation. Bleeding at the aspiration site and mild pain during and after injection were the only adverse events.
Investigators analyzed the stem cell products before injection and found the bone marrow-derived MSCs released significantly more angiogenic factors than the mononuclear cells, a difference they suggested may account for the greater effects seen in the MSC group.
The patients in the Chinese study are among the 25% to 40% of patients with CLI who, because of factors including diffuse vascular disease, unfavorable anatomical location, extensive comorbidities, or previously failed revascularization, are not candidates for bypass or endovascular revascularization.24
In a 2013 meta-analysis, Teraa et al identified 12 randomized controlled trials (510 patients) of bone marrow-derived stem cells for these “no-option” patients, and reported the therapies significantly reduced amputation rates and improved ulcer healing, ankle-brachial index (ABI) and transcutaneous oxygen measurements, pain, and pain-free walking scores compared with standard care.25
The 12 trials involved four different stem cell types and different isolation procedures; six used bone marrow-derived mononuclear cells, two used bone marrow-derived MSCs, two used peripheral blood mononuclear cells, and one used a commercial preexpanded cell product obtained from bone marrow. Because of the limited data, Teraa et al could draw no conclusions about the superiority of one stem cell type or isolation method over another, an area they recommended for further study.
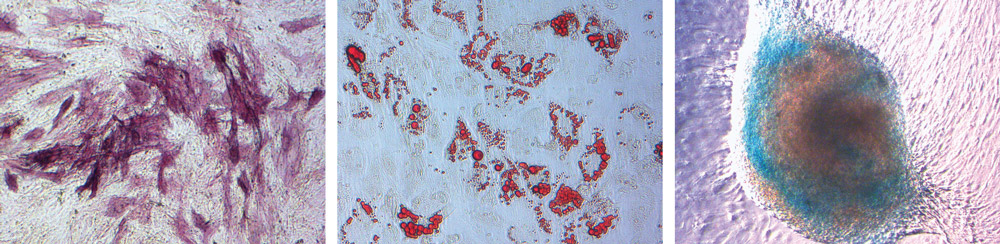
Human MSCs cultured and seeded into adipogenic, chondrogenic, or osteogenic differentiation media for 14 days, revealing osteoblasts (left), adipocytes (center), and chon- drocytes (right). (Images courtesy of Thermo Fisher Scientific’s Life Technologies.)
Their analysis supports previous trial data demonstrating safety,26 but they noted most follow-up has been relatively short, and longer-term data are needed to confirm safety and efficacy.
Vascular surgeon Mark D. Iafrati, MD, is currently following up favorable results from a 2011 pilot trial27 of bone marrow aspirate concentrate (BMAC) injection for patients with no-option CLI as principal investigator for a national randomized controlled double-blinded trial with a planned enrollment of 210 patients at up to 40 centers.
The BMAC product, which is produced with a commercial system also under study in the US for bone healing and cardiovascular disease, is a multicellular mix of MSCs, hematopoietic stem cells, endothelial progenitor cells, platelets, lymphocytes, and granulocytes.
In the pilot trial, Iafrati and colleagues at Tufts University in Boston saw a trend toward improvement in amputation, pain, and quality of life at 12 weeks in 34 patients with rest pain and tissue loss (Rutherford class 4 and 5, respectively) treated with BMAC compared with a control group (n = 14) that received sham injections. Patients in the treatment group had an amputation rate of 16% compared with 31% for controls.
The BMAC group experienced greater improvements than controls with regard to pain (44% vs 25%), wound healing (32% decrease in wound size vs 27%), and hemodynamics (ABI improvement in 32.4% vs 7.1%).
“The BMAC group also had better quality of life, so pretty much everything we were looking at was moving in the right direction,” said Iafrati, who is chief of vascular surgery, director of the Vascular Center, and associate professor of surgery at Tufts.
Iafrati said unpublished interim data from the pilot study, which was not powered for statistical significance, show continued benefit related to amputation risk in the patients who began the trial with tissue loss (Rutherford class 5); at two-year follow-up 45% of these patients had undergone amputation compared with 71% in the control group.
“There’s also a lot of suggestion that quality of life, pain, paresthesias, and neuropathy all seem to be improving in the pilot study, which will reach its five-year endpoint later this year, when we’ll close it out and look at all the data,” he said.
Iarfati is confident about the safety of this method, and has noted only one potential injection site reaction in more than 150 patients.
“I really have no doubt this will meet the bar for a very safe approach, and I suspect when we clear this hurdle, this therapy will have tremendous potential to be much more widely applied,” he said. “Obviously we need to test it in various circumstances, but I see it potentially as a routine adjunct in many severe lower extremity ischemia patients that can be combined with angioplasty and stenting or bypass. There is every reason to think this could result in improved outflow and patency in primary repairs in addition to helping with tissue growth.”
Knee
Stem cell therapies for common knee conditions, including knee osteoarthritis, cartilage damage, and meniscus tears, are in clinical use in the US and other nations, often in advance of robust evidence of efficacy.
Along with their regenerative properties, animal studies of osteoarthritis and other conditions have shown that stem cells have powerful anti-inflammatory effects, 28-30 which provide a rationale for their use in many conditions. An Internet search reveals dozens of clinics in the US and internationally offering various stem cell injections for osteoarthritis, tendinitis, ligament tears, and other musculoskeletal indications.
In US clinics these therapies typically involve unexpanded autologous stem cells derived from bone marrow or fat, but potential patients can also access overseas clinics that offer treatment with stem cell therapies not available in the US outside of research settings. Insurance rarely covers these procedures.
Because minimally manipulated autologous stem cell products are not considered drugs, they aren’t subject to FDA oversight, and providers offering these injections can make as-yet unsupported claims of efficacy in various indications and set their own prices.
Marc Darrow, MD, JD, who offers bone marrow-derived MSC injections for cartilage and meniscus repair at the Darrow Sports & Wellness Institute in Los Angeles, told LER that he has noted prices ranging from $5000 to $10,000 per injection in some clinics. In his practice, he said, he has seen stem cell injection improve pain and help patients avoid surgery and noted that he sets his fees lower—about $1800 per injection depending on the individual—to reach the greatest number of patients.
Prices for stem cell therapy vary widely. In Atlanta, for example, the Emory Orthopaedics & Spine Center notes on its website that stem cell injections for joint pathology generally cost between $3000 and $3500 for one joint and $4000 to $5000 for two joints.
Scott Rodeo at the Hospital for Special Surgery said, in his opinion, stem cell therapies are being used too widely in the community.
“These therapies seem to be generally safe with patient’s own cell tissue, but I think we need a lot more information about appropriate indications, appropriate doses, and the best way to deliver cells,” Rodeo said.
Research is starting to answer those questions.
A South Korean proof-of-concept trial published in May in Stem Cells reported that direct injection of autologous adipose tissue-derived culture-expanded MSCs into osteoarthritic knees improved pain and knee function with no treatment-related adverse events.31 Investigators placed 18 patients with knee OA (Kellgren–Lawrence grade 3 or 4 knee OA and knee pain of four or more on a visual analog scale) into three groups receiving low, mid, and high doses of stem cells.
After six months, total WOMAC (Western Ontario and McMaster Universities Osteoarthritis) scores for patients in the high-dose group improved 39%, pain decreased 45%, and knee scores improved 50% from baseline. Imaging studies also showed regenerated articular cartilage in this group as measured by cartilage volume on serial magnetic resonance imaging (MRI) in the medial femoral and tibial condyles. Patients in the mid-dose group showed improvement in some clinical outcomes, while those in the low-dose group had little or no improvement in most outcome measures.
A second Korean trial evaluated knee pain and function and second-look arthroscopic findings of intra-articular injection of adipose-derived stem cells with arthroscopic lavage for 30 patients older than 65 years with knee osteoarthritis.32 At two-year follow-up patients had significantly improved pain and function compared with follow-up at one year, and only five demonstrated worsening symptoms. Arthroscopy at two years showed 87.5% of patients improved or maintained cartilage status, and none underwent total knee replacement during the study period.
Preliminary research also suggests intra-articular injection of stem cells into the knee is effective in promoting healing after meniscal surgery. Investigators for a multicenter US study of an expanded allogeneic MSC product recently reported favorable results from a randomized controlled trial of 57 patients who had undergone partial medial meniscectomy.33
Patients received single injections seven to 10 days after surgery of higher or lower doses of stem cells suspended in hyaluronic acid or of hyaluronic acid alone. Investigators followed them for two years, evaluating safety, meniscal regeneration, and clinical outcomes.
MRI at one year indicated meniscal regeneration in some participants, showing significantly increased meniscal volume in 24% of patients in the lower-dose stem cell group and in 6% of those in the higher-dose group compared with controls. At two years only three patients (all in the lower-dose group) showed meniscal volume increases.
Baseline joint evaluation identified arthritic changes in 37%, 64%, and 66% of the control, lower-dose, and higher-dose groups, respectively, and at one year overall OA progression was unchanged in 76% of all patients. Both groups receiving stem cells had statistically significant improvements in pain at two years compared with those receiving hyaluronic acid alone.
“The meniscus cartilage is hard to heal because it’s avascular, and I didn’t think the meniscus would grow at all, especially not with a single injection,” said C. Thomas Vangsness Jr, MD, lead investigator for the trial and professor of orthopaedic surgery in the Keck School of Medicine at the University of Southern California in Los Angeles. “Safety was our first goal, and we’ve established that—we used huge numbers of cells and found no serious safety issues associated with treatment. In the follow-up studies we’ll probably lower those numbers and do multiple injections.”
Vangsness also plans a trial of the product for patients with moderate OA.
“It makes sense that these cells, with their anti-inflammatory and immuno-regulatory properties could help in patients without advanced disease,” he said.
US researchers are also studying stem cells for repair of knee cartilage defects. A recent review by Anderson et al of preclinical and human studies noted that, while animal studies have shown structural and biomechanical benefits and some small human trials have shown improvements in clinical outcomes and on MRI, there is no evidence from human trials that the stem cells were directly responsible for observed benefits.2 The authors noted that small study sizes, varied delivery systems, and often ill-defined outcome measures hampered their interpretation of the literature.
Brian Cole, MD, professor of orthopedics, anatomy, and cell biology at Rush University Medical Center in Chicago, is lead investigator for a two-year phase 1/11a study using an allogeneic MSCs product derived from umbilical cord blood as an adjunct to microfracture in 12 patients with localized knee cartilage loss.
“This is a single cohort study for safety, and patients are all doing well,” he said. “There appears to be an added benefit [of stem cells] in the early postop phases of less pain, less morbidity, less swelling, less discomfort, and the joint looks ‘quieter’ than it does without them, but that’s an observational viewpoint.”
Cole and colleagues are following up with patients at 12 and 24 months.
“[Stem cell therapy] is one of the most exciting things we have on the horizon, in my opinion, but my enthusiasm is tempered by the regulatory burden and misunderstandings about how stem cells can benefit patients,” he said. “I look at them as biologic adjuncts to existing treatments, and I believe, because of their very potent anti-inflammatory and immuno-modulatory effects, there is real benefit to be had. But we need to be reasonable in how we move forward and be ethically and fiscally responsible in how we deliver these therapies to our patients.”
Foot and ankle
In his clinic in Westerville, OH, Christopher Hyer, DPM, MS, has been using bone marrow-derived stem cells to assist with healing after foot and ankle surgeries for about eight years.
“At first we used [autologous bone marrow-derived MSC injections] in patients we saw as being at high risk for not healing well, such as smokers and people with diabetes, as a way to augment healing. We saw that it was quick and easy, with low morbidity, and have started using it in elective cases as well,” said Hyer, who recently reported on the use of an MSC allograft product for high-risk foot and ankle reconstructions.34
In a case series of 20 consecutive patients identified as high risk for nonunion and undergoing hindfoot or ankle arthrodesis, Hyer and colleagues tightly packed allograft cells into denuded and fenestrated joints prior to internal fixation.
The mean average interval to radiologic union was 11.6 ± 2.0 (range 8-15) weeks. The interval to partial weight bearing was 5.5 ± 1.8 (range 3-12) weeks, to full weight bearing, 8.4 ± 1.9 (range 5-14) weeks, and to wearing shoes, 13.6 ± 3.0 (range 10-20) weeks.
“[The allograft] did improve both the rate and speed and healing versus the traditional reports [in high-risk patients] on that same type of fusion, particularly because our cases were high risk for not healing,” Hyer said.
A recent Korean study also found using MSCs improved results in ankle procedures in patients at high risk for poor outcomes.35 The investigators placed 65 patients older than 50 years with osteochondral lesions of the talus into two groups; one received microfracture plus injection of autologous MSCs harvested from gluteal fat while the other underwent microfracture alone.
Both groups had decreased pain and improved AOFAS (American Orthopaedic Foot and Ankle Society) Hindfoot scores at final follow-up (mean, 21.8 months; range, 12-44 months), but patients receiving stem cells in addition to microfracture had significantly greater improvement in AOFAS scores and activity levels and shorter return-to-activity times. Patients with large lesions (>109 mm2) or a subchondral cyst treated with MSCs saw the greatest additional benefits relative to microfracture alone.
Hyer noted that, despite his favorable results and those of other researchers, he’s leaning toward using crushed cancellous bone allografts seeded with patients’ autologous bone marrow in the place of the expensive commercial product used in his study.
“It’s an inexpensive off-the-shelf product that’s easy to use,” he said. “You rehydrate it, wash it, and then seed it with bone marrow you pull from the patient, and it’s my belief that it is as good if not better than the stem cell allografts products that are commercially available because it’s the patient’s cells, not foreign stem cells.”
Like the other investigators and clinicians interviewed for this story, Hyer believes that stem cell therapies hold significant promise in a wide variety of lower extremity applications, and that patients will benefit when research answers questions about the ideal stem cell dose and type for specific conditions and individuals.
Stem cell regulation: Beyond minimal manipulation
Practitioners and patients alike are strongly drawn to the concept of regenerative medicine, and the allure has fueled a rapid increase in the number of clinics offering stem cell therapies and of companies developing and supplying related products and equipment. In the US, federal regulators are trying to keep pace with the emerging field, and recent legal decisions affect how researchers and clinics can study and administer these products.
Many experts interviewed by , for example, think the most beneficial way to use adult stem cells may to expand them in culture.
“You take the bone marrow aspirate and then use flow cytometry to isolate the small proportion of true stem cells, and then culture those cells. So instead of thousands, you end up with millions,” said Scott Rodeo, MD, an orthopedic surgeon at the Hospital for Special Surgery and professor of orthopaedic surgery at Weill Cornell Medical College in New York City, who is using bone marrow-derived mesenchymal stem cells for several indications, including for augmentation of meniscus repair and cartilage repair of the knee.
Although such work is going on around the globe, notably in Korea, the EU, and Australia, significant regulatory hurdles hamper its use in the US. Current Food and Drug Administration (FDA) regulations prohibit the use of expanded stem cells unless the product is administered under an investigational new drug (IND) application.13
A key point in the FDA’s restrictions is that autologous cells be “minimally manipulated”; once processing goes beyond this stage, the material is considered a drug with all the regulation that comes with that designation. In recent years, several clinics offering expanded stem cell therapies have been enjoined by the FDA from administering their products in the US and have moved those services to countries with different regulations.14,15
Involved clinicians and companies have questioned the definition of minimal manipulation and other aspects of the FDA regulations with little success. The District of Columbia Circuit U.S. Court of Appeals on February 4 rejected an appeal from a Colorado-based company, which had been blocked by the FDA in 2010 from offering an expanded stem cell product for orthopedic conditions.15 The company continues to provide treatment with unexpanded stem cells in the US and offers cultured stem cell therapy in its Cayman Islands location.16
“The FDA’s concern is safety, and rightly so,” Rodeo said. “Transporting cells to and from [the point of care] to labs for expansion brings into consideration chain of custody issues and good manufacturing practices, and there are also concerns about whether cells may change their behavior in culture, which could theoretically occur.”
Mark D. Iafrati, MD, a vascular surgeon and principal investigator for a trial of autologous bone marrow-derived stem cells in patients with critical limb ischemia, isn’t convinced that, for the conditions he treats, expanding cells produces better results than simply concentrating bone marrow aspirate.
“If you culture-expand cells you get more activity, but you only get more activity from whatever cell line you’re selecting, and don’t think anybody knows yet what one type of cell does versus another. Also, I very much suspect that the platelets and other products that we’re bringing along in very high numbers [from bone marrow aspirate] are part of the package, which I think is a signaling feedback process providing the whole milieu that you require to generate angiogenesis and vasculogenesis,” Iafrati said. “Not only do I think it’s easier to do, faster, and less expensive when [done at] the point of service, but I think there is plenty of reason to think it would be ultimately more effective.”
Iafrati has traveled to Mexico, Chile, Argentina, Brazil, and Germany to train clinicians in the use of the therapy he’s investigating.
Although the US has been slower to move this therapy into clinical use, he noted, “I’m happy the FDA is holding out for some real data. If we didn’t put it through this rigor, we’d never be able to refine the therapy.”
1. Bashir J, Sherman A, Lee H, et al. Mesenchymal stem cell therapies in the treatment of musculoskeletal diseases. PM_R 2014:6(1):61-69.
2. Anderson JA, Little D, Toth AP, et al. Stem cell therapies for knee cartilage repair: the current status of preclinical and clinical studies.Am J Sports Med 2013 Nov 12. [Epub ahead of print]
3. Blumberg SN, Berger A, Hwang L, et al. The role of stem cells in the treatment of diabetic foot ulcers. Diabetes Res Clin Pract 2012;96(1):1-9
4. Young, RR. Adult stem cell fact sheet. Stem Cell Summit website. http://www.stemcellsummit.com/pdf/StemCell_Fact_Sheet2012.pdf. Accessed March 19, 2014.
5. Chamberlain G, Fox J, Ashton B, Middleton J. Concise review: Mesenchymal stem cells: Their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells 2007;25(11):2739-2749.
6. Stolzing A, Jones E, McGonagle D, Scutt A. Age-related changes in human bone marrow-derived mesenchymal stem cells: Consequences for cell therapies. Mech Ageing Dev 2008;129(3):163-173.
7. Wang Y, Han ZB, Song YP, Han ZC. Safety of mesenchymal stem cells for clinical application. Stem Cells Int 2012;2012:652034.
8. Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science 1999;284(5411):143-147.
9. Shabbir A, Zisa D, Suzuki G, Lee T. Heart failure therapy mediated by the trophic activities of bone marrow mesenchymal stem cells: A noninvasive therapeutic regimen. Am J Physiol Heart Circ Physiol 2009;296(6):H1888-H1897.
10. Nixon AJ, Watts AE, Schnabel LV. Cell- and gene-based approaches to tendon regeneration. J Shoulder Elbow Surg 2012;21(2):278-294.
11. Doorn J, Moll G, Le Blanc K, et al. Therapeutic applications of mesenchymal stromal cells: Paracrine effects and potential improvements. Tissue Eng Part B Rev 2012;18(2):101-115.
12. Jiang XX, Zhang Y, Liu B, et al. Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells. Blood 2005;105(10):4120-4126.
13. Lysaght T, Campbell AV. Regulating autologous adult stem cells: the FDA steps up. Cell Stem Cell 2011;9(5):393-396.
14. Cyranoski D. Controversial stem-cell company moves treatment out of the United States: Celltex to send patients to Mexico. Nature website. http://www.nature.com/news/controversial-stem-cell-company-moves-treatment-out-of-the-united-states-1.12332. Published January 30, 2013. Accessed March 19, 2014.
15. Moylan T. “D.C. Circuit Says FDA Can Regulate Stem Cell Drugs; Company Properly Enjoined.” http://www.lexisnexis.com/legalnewsroom/litigation/b/litigation-blog/archive/2014/02/04/d-c-circuit-says-fda-can-regulate-stem-cell-drugs-company-properly-enjoined.aspx. LexisNexis website. Published February 4, 2014. Accessed March 22, 2014.
16. FDA Clarification. Regenexx website. http://www.regenexx.com/common-questions/regenexx-fda-clarification/. Accessed March 22, 2014.
17. Wu Y, Wang J, Scott PG, Tredget EE. Bone marrow-derived stem cells in wound healing: a review. Wound Repair Regen 2007;15(Suppl 1):S18-S26.
18. Kataoka K, Medina RJ, Kageyama T, et al. Participation of adult mouse bone marrow cells in reconstitution of skin. Am J Pathol 2003;163(4):1227-1231.
19. Wu Y, Chen L, Scott PG, Tredget EE. Mesenchymal stem cells enhance wound healing through differentiation and angiogenesis. Stem Cells 2007;25(10):2648-2659.
20. Li H, Fu X, Ouyang Y, et al. Adult bone- marrow-derived mesenchymal stem cells contribute to wound healing of skin appendages. Cell Tissue Res 2006;326(3):725-736.
21. Blumberg SN, Berger A, Hwang L, et al. The role of stem cells in the treatment of diabetic foot ulcers. Diabetes Res Clin Pract 2012;96(1):1-9.
22. Marino G, Moraci M, Armenia E, et al. Therapy with autologous adipose-derived regenerative cells for the care of chronic ulcer of lower limbs in patients with peripheral arterial disease. J Surg Res 2013;185(1):36-44.
23. Lu D, Chen B, Liang Z, et al. Comparison of bone marrow mesenchymal stem cells with bone marrow-derived mononuclear cells for treatment of diabetic critical limb ischemia and foot ulcer: a double-blind, randomized, controlled trial. Diabetes Res Clin Pract 2011;92(1):26-36.
24. Norgren L, Hiatt WR, Dormandy JA, et al. Inter-society consensus for the management of peripheral arterial disease (TASC II). J Vasc Surg 2007;45(suppl S):S5-S67.
25. Teraa M, Sprengers RW, van der Graaf Y, et al. Autologous bone marrow-derived cell therapy in patients with critical limb ischemia: a meta-analysis of randomized controlled clinical trials. Ann Surg 2013;258(6):922-999.
26. Fadini GP, Agostini C, Avogaro A. Autologous stem cell therapy for peripheral arterial disease. Meta-analysis and systematic review of the literature. Atherosclerosis 2010;209(1):10-17.
27. Iafrati MD, Hallett JW, Geils G, et al. Early results and lessons learned from a multicenter, randomized, double-blind trial of bone marrow aspirate concentrate in critical limb ischemia. J Vasc Surg 2011;54(6):1650-1658.
28. Prockop DJ, Oh JY. Mesenchymal stem/stromal cells (MSCs): role as guardians of inflammation. Mol Ther 2012;20(1):14-20.
29. Diekman BO, Wu CL, Louer CR, et al. Intra-articular delivery of purified mesenchymal stem cells from C57BL/6 or MRL/MpJ superhealer mice prevents post-traumatic arthritis. Cell Transplant 2013;22(8):1395-1408.
30. van Buul GM, Villafuertes E, Bos PK, et al. Mesenchymal stem cell secrete factors that inhibit inflammatory processes in short-term osteoarthritic synovium and cartilage explant culture. Osteoarthritis Cartilage 2012;20(10):1186-1196.
31. Jo CH, Lee YG, Shin WH, et al. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: A proof-of-concept clinical trial. Stem Cells 2014;32(5):1254-1266.
32. Koh YG, Choi YJ, Kwon SK, et al. Clinical results and second-look arthroscopic findings after treatment with adipose-derived stem cells for knee osteoarthritis. Knee Surg Sports Traumatol Arthrosc 2013 Dec 11. [Epub ahead of print]
33. Vangsness CT Jr, Farr J 2nd, Boyd J, et al. Adult human mesenchymal stem cells delivered via intra-articular injection to the knee following partial medial meniscectomy: a randomized, double-blind, controlled study. J Bone Joint Surg Am 2014;96(2):90-98.
34. Scott RT, Hyer CF. Role of cellular allograft containing mesenchymal stem cells in high-risk foot and ankle reconstructions. J Foot Ankle Surg 2013;52(1):32-35.
35. Kim YS, Park EH, Kim YC, Koh YG. Clinical outcomes of mesenchymal stem cell injection with arthroscopic treatment in older patients with osteochondral lesions of the talus. Am J Sports Med 2013;41(5):1090-1099.