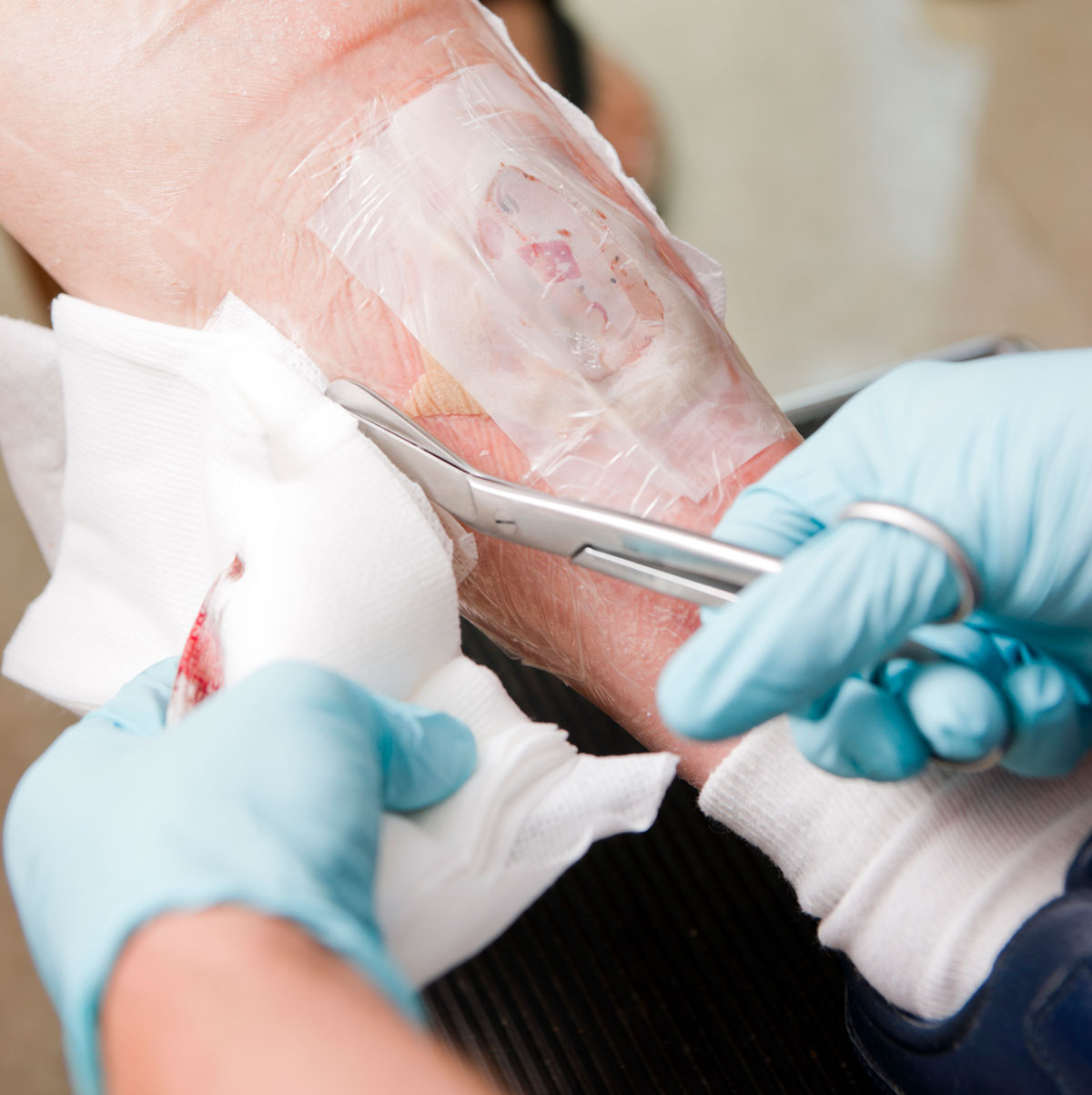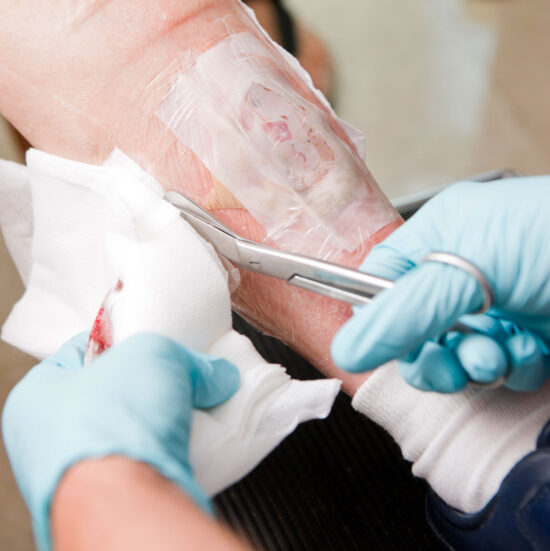
By James McGuire DPM, LPT, LPed, and Avnee J. Patel, DPM
Chronic wounds affect millions of patients worldwide making wound care an integral part of healthcare. Such wounds pose a significant financial burden upon both patients and the healthcare industry and negatively impact the patient’s overall quality of life. For this reason, continued innovation within the field of wound dressings and dynamic wound treatment options is of utmost importance. Nanoparticles are a developing component of wound treatment largely in part due to their biocompatibility, ability to function in many ways and environments, and positive impact on chronic wound healing. Here, we review various nanomaterials, their function in wound healing and action as delivery vehicles for actives and look closer at electrospinning – a method of delivering nanoparticles to a wound bed.
Wound Healing Review
Due to the ever-increasing number of patients with wounds, wound healing has grown into a multidimensional subdivision of medicine. While for centuries the majority of wounds were acute in nature, chronic wounds have become a major part of modern healthcare, making the continued investigation into optimizing treatment of chronic wounds paramount.
Poor healing environment, concomitant microbial infection and prolonged inflammation complicate wound healing potential.1 While acute wounds heal without incident in the expected period of 8-12 weeks, chronic wounds heal slowly, are often associated with underlying disease, and are complicated by other factors such as microbial infection.2 The implication of poor wound healing is made abundantly clear in podiatric wound care where poor vascular health, uncontrolled diabetes, neuropathy, and repetitive injury to the lower extremity often result in non-healing or delayed-healing wounds.
Traditional dressings primarily function as a passive part of the wound healing process – preventing continued abrasion or trauma to the wound site or maintaining some degree of moisture as seen with petroleum-impregnated gauze. Alone, though, they neither maintain nor encourage the wound healing cascade, functioning instead to protect an open wound from worsening, but minimally contributing to its improvement. In this paper, we discuss novel materials available for wound treatment, applications to wound healing, and explore electrospinning as a method of wound dressing.
Physiology of Wound Healing and Podiatric Applications
Wound healing has been well-described as having 4 main stages: hemostasis, inflammation, proliferation, and remodeling.1,2,4-6 While acute wounds progress through these stages at an expected rate, recruiting the necessary cells, chemokines, and growth factors in a highly regulated fashion, chronic wounds have been described as becoming stalled in the inflammatory stage. Acute wounds heal to produce an ordered extracellular matrix (ECM) and the matrix develops at a rate that exceeds the rate of tissue breakdown at the wound site leading to complete remodeling of the wound.1 In the setting of a persistent inflammatory response, phenotypically abnormal inflammatory infiltrate disrupts the coordinated cellular response at the wound site resulting in a chronic, non-healing wound.4
Integral to podiatric wound healing is avoiding amputation, of which 85% are preceded by non-healing wounds such as diabetic ulcers, venous ulcers, and pressure ulcers.2,5,7 Biofilms, which cause persistent inflammation and worsen infection risk, are a common complicating factor of chronic wounds which adds to their delayed healing, especially in those who are immunocompromised.7,8 Nanoparticle technology possesses the capability to function as an active, biocompatible, non-irritating dressing that is stage-specific and prevents biofilm formation thereby supporting a normal wound healing cascade.2,3,9
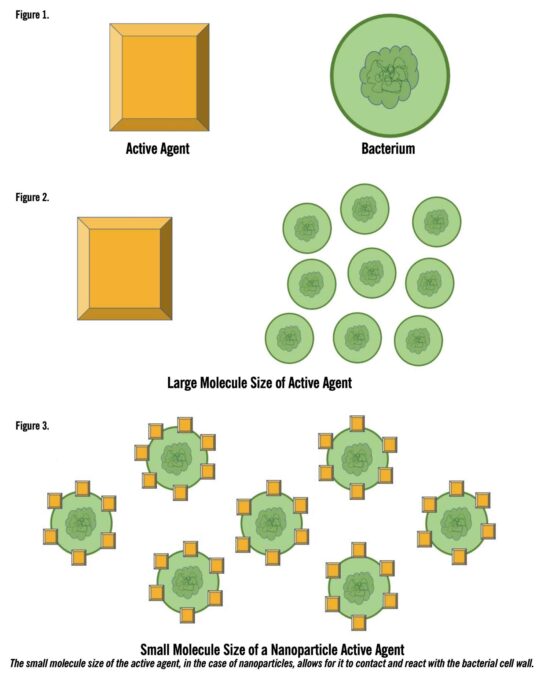 Characteristics of the Ideal Wound Dressing
Characteristics of the Ideal Wound Dressing
In the podiatric setting, wounds often possess irregular borders and varying depths making active, moldable dressings integral to facilitating healing. Current wound treatment protocols often involve traumatic, painful debridement of the wound bed to granular tissue and application of dressings which often do not conform to the wound bed and border. This may lead to patient discomfort with the dressing, slippage of the dressing, and the requirement for frequent changes. This introduces a significant burden on not only the patient and family, but also the healthcare system with the increased cost of home nursing and additional office visits for dressing changes and follow-up. To promote atraumatic, efficient wound healing, the ideal wound dressing should conform to the wound bed and require infrequent changes.10 Additionally, the dressing’s ability to promote tissue and ECM proliferation is imperative.11
As previously mentioned, the bioburden carried by chronic wounds in the form of biofilms covering the wound bed introduces an additional variable necessitating consideration. In chronic wounds, there is often an increase in exudate comprised of increased levels of inflammatory cells and proteins indicative of infection.8 Dressings that maintain a moist wound bed are sufficient for sterile wounds, however necrotic wounds and those which carry a bioload that is inconsistent with normal dermal flora are best served with active, functional dressings.11 Nanoparticles and other emerging wound healing technologies support the delivery of actives, such as antibiotics, anti-inflammatories, and growth factors, to the wound bed, thus increasing the efficiency and efficacy of wound dressings.
Wound Treatment Materials, Podiatric Applications, and Mechanisms of Antimicrobial Activity
There is no defined gold standard protocol for the treatment of acute and chronic wounds. There are, however, many effective conventional and adjunctive treatment options including hyperbaric oxygen therapy, negative pressure wound therapy, advanced wound dressings, and new revascularization surgeries which aim to optimize the wound environment in an effort to promote healing.5 Key complicating factors for these methods warrant consideration, including but not limited to unaddressed systemic diseases, nutrition, access to available dressings or therapies, insurance coverage, and patient adherence issues – all of which ultimately delay wound healing.2,5 Especially in podiatric wound care, where persistent chronic wounds often plague patients with key comorbidities, such as diabetes and vascular disease, development of a multispecialty, dynamic, and targeted wound treatment plan could prove invaluable.
External pathogenic contamination of chronic wounds, commonly polymicrobial in nature, necessitates the use of an aggressive wound hygiene program and antimicrobial dressings. Wound dressings including gold, zinc, and silver have been well-studied and shown to have antimicrobial properties.1,2,5,6,11 Conventional antimicrobial agents, such as oral and topical antibiotics, pose contraindications and complications in the setting of chronic wounds. Specifically, bacterial resistance due to antibiotic-overuse, dermatitis, and other adverse cutaneous reactions to topical agents could pose an issue. Antimicrobial nanoparticles could circumvent these potential complications by delivering bioactives on biologically inert molecules.11
Nanomaterials in Wound Healing
Nanomaterials can target specific stages in the wound healing cascade, have antimicrobial and anti-inflammatory properties, and promote angiogenesis – making them a novel technology to be considered in the world of wound healing.1-3 Nanoparticles are up to two-times more potent than pure ionic forms, largely in part due to their high surface-area-to-volume ratio allowing for greater elution of bioactive compounds. Additionally, their small size allows them to better attach to and infiltrate the target cell walls of bacteria. In the setting of biofilms, this may allow for more efficient targeting of the involved bacteria. Importantly, molecular differences exist, underscoring the importance in molecule selection in the treatment of individual wounds.
Gold
Gold nanoparticles (AuNPs) are well-studied and have many applications to wound healing, largely in part due to their bacteriostatic and bactericidal properties.3,6,11 Additionally, they have been shown to have antioxidant properties.11,12 AuNPs impart their bacteriostatic activity by binding bacterial DNA and inhibiting DNA uncoiling to ultimately decrease bacterial DNA replication; they convey their bactericidal activity by directly targeting bacterial cell walls, making it especially helpful in locally targeting multi-drug resistant bacteria.11 In the setting of chronic wounds, which are commonly seen in patients with underlying pathology, directed local antimicrobial therapy to the affected sites may be the preference, considering the physiological toll systemic antimicrobial treatments often have. In addition, AuNPs have been shown to have a stimulatory effect on angiogenesis and epithelialization leading to faster wound closure.1,6,11-13
Among the keys to more efficient wound healing are targeted therapies such as AuNPs. In a rat model, full thickness excisional wounds showed decreased inflammatory cells and increased fibroblasts in the granular tissue of the wound bed after AuNP and silver nanoparticle (AgNP) topical application.13 In this example, AuNPs and AgNPs directly acted upon the inflammatory and proliferative phases of the wound healing cascade to optimize and accelerate the healing process. Complete re-epithelialization and development of a collagen scaffold was seen in 14 days with topical application of AuNPs.13
AuNPs are chemically stable, biodegradable, and biocompatible, making them ideal additions to other treatments used for the wound bed.3,11,14 The intrinsic anti-inflammatory and antioxidant capabilities of AuNPs may be enhanced by combination with anti-inflammatory or antioxidant medications to specifically target the angiogenic and inflammatory phases of the wound healing cascade.3 In doing so, chemical and pharmacotherapeutic modulators are directly targeted to the wound bed to elicit greater affect. For example, when conjugated with AuNPs, vancomycin had greater activity against vancomycin-resistant enterococcus and gram-negative bacteria.14 Reactive oxygen species which potentiate inflammation and lead to delayed wound healing have also been treated with AuNPs.12 In combination with photo biomodulation therapy (PBMT), a light therapy that improves cell migration thereby promoting actions such as epithelialization and angiogenesis, AuNPs were shown to efficiently target inflammation at the wound bed to promote augmented wound healing.12
Zinc
Zinc is an essential mineral metal oxide element. Zinc nanoparticles (ZnNPs) are commonly used in skin creams and antiseptic topicals primarily due to anti-inflammatory properties, biocompatibility, hemostatic ability, and the element’s mechanical strength and tensile properties.1,6,15 Zinc induces bactericidal activity by disrupting cell membrane activity and causing bacterial cell death after adsorption into the bacterial cytoplasm.1,11 In addition to its bactericidal activity, when incorporated into hydrogel-based dressings, it may be used as a resorbable template to induce keratinocyte migration to encourage epithelialization at the wound bed.6,11,15 In the setting of chronic wounds, where the wound bed is sequestered in a constant inflammatory cycle, a topical dressing that promotes epithelialization is paramount. For this reason, it is important to note that there is toxicity associated with ZnNPs – namely that it produces reactive oxygen species to damage bacterial cell membranes. However, this is also known to be associated with keratinocyte dysfunction which may potentiate the inflammatory cycle at the wound bed.1,11 ZnNPs have been found to have size-dependent bactericidal activity at the wound bed surface, with 8nm showing greater than 95% of S. aureus growth inhibition as compared to 50nm showing 40% bactericidal activity.1 This should be taken into consideration when embedding ZnNPs in wound dressings.
Embedded ZnNPs have properties which promote wound healing. As a part of hydrogels, collagen dressings, and cellulose sheets, ZnNPs have been shown to exhibit antibacterial activity and promote tissue regeneration.1 Additionally, ZnNPs have been shown to be compatible with other materials commonly seen in wound dressings. Balaure et al explored combining collagen, one of the most abundant extracellular matrix proteins, with Zinc to exploit its antibacterial activity in combination with the ability of collagen to act as a wound bed scaffold.15 They found that 1% orange essential oil functionalized zinc oxide NPs (ZnO@PorT) which were not only biocompatible, but also accelerated wound healing and prevented bacterial bioload at the wound bed of third degree burns.11,15 Similarly, Koga et al explored crosslinking an alginate-aloe vera gel with ZnNPs and found that although this gel did not show accelerated healing, there was greater degree of exudate absorption.16 This underscores the versatility of ZnNPs in wound dressing technology and its ability to have stage-specific applications to the wound healing cascade.
Silica
Silica, an inorganic compound, is known for having highly specific surface area and functional capabilities. It has been described as biocompatible and cytocompatible, and has the ability to enhance cell proliferation.3,17-19 Additionally, silica nanoparticles (SiNPs) can be easily produced, which is particularly important considering the significant financial burden wound care presents to many healthcare systems.19 Silica does not innately harbor wound healing capacity; however, its ability to function as a scaffold allows it to have diverse applications to wound healing.17 In combination with platelet lysate (PL), porous silicon microparticles (PSi) were examined for their ability to promote wound healing and in vitro assays found PL-modified PSi induced a significant proliferative effect when compared to positive and negative controls.18 Similarly, it has been shown that when PSi are coated with biopolymers, such as chitosan or hyaluronic acid, they may be loaded with antibiotics such as vancomycin to provide a local controlled release of the drug.18,20,21 The local delivery of drugs is key to reduction of multi-drug resistance in bacteria and decreasing the systemic burden of oral medications on patients, especially in the setting of chronic wounds and those with active infection in addition to considerable comorbidities.
Silver, a common component of commercial wound dressings, has been shown to be stable in combination with silica, with silica acting as a scaffold for silver, forming Ag-MSNs (disulfide bond-bridged nanosilver-decorated mesoporous silica nanoparticles).17 Ag-MSNs showed both antibacterial and adhesive activities and increased the rate and efficiency of wound closure compared to sutures with little local and systemic toxicity.17 In a nanoparticle solution, silica functions as an adhesive by forming a hydrogel which functions alongside tissues without changing in permeability or rigidity, allowing it to conform to the wound bed.22 This nano-adhesive Ag-MSN system showed antibacterial activity against S. aureus and E. coli, two common flora present on chronic wound beds and were shown to act as an effective and efficient system to close wound edges.17,22
SiNPs have been shown to act as suitable scaffolds for the delivery of other nanoparticles and drugs, however it is important to note that particle size and surface chemistry determined cytotoxicity.18,21 As a drug delivery system, SiNPs have been studied extensively in systemic chemotherapeutic transport. Transcutaneously, SiNPs were shown to penetrate the epidermis without causing skin damage or organ toxicity.21 Despite silica adhesion strength increasing with increase in particle size, wound bed size and surface area should be taken into account because an increase in surface area of SiNPs have been found to be generally more cytotoxic.21,22 Considering the versatility of the compound and its ability to be modified to specifically target flora and promote epithelializaton, and with appropriate consideration to their size, SiNPs could be especially beneficial to those patients suffering from chronic wounds which carry a bioload.18,19 (18,19).
Copper
Copper is an essential element which is known to be widely bactericidal.1,6,23,24 Additionally, it is known to promote angiogenesis, fibroblast proliferation, collagen formation, and ECM cross-linking.24,25 In studies of copper-impregnated wound dressings, six days after application of the sterile dressing, wound size was measured to be significantly smaller compared to the control group. There was an increase in proangiogenic factors seen at one day after dressing application and angiogenic effects, including increased oxygenation of the tissues and decreased inflammation of the wound bed, were observed.24,25 This is consistent with the ability of copper ions to increase gene expression to promote angiogenesis and ultimately, wound closure. CuNPs have been shown to have a high rate of absorption, adsorption, penetration, and availability and have wide activity to microbes commonly seen in the setting of diabetic ulcers, such as S. aureus, E. coli and P. aeruginosa.6,23,26,27 Additionally, CuNPs and topical copper applications are known to be safe to humans. CuNPs exert their bactericidal properties by releasing ionic copper which disrupts the bacterial cell membrane causing cytoplasmic degradation and bacterial cell death.1,26,27
Copper also harbors the ability to act as a scaffold, allowing it to exert dual action with other active molecules. While typically unstable in physiologic proteins, copper metal-organic frameworks (Cu-MOFs) allow it to perform in combination with other molecules.28,29 In combination with nitrous oxide (NO), a Cu-MOF was electrospun to contain a NO core resulting in a delayed CuNP release over an extended period of time.30 This presents an alternate approach to wound treatment – rather than stage-specific nanoparticle action, nanoparticles may sustain ion release over time thereby extending their action. This is especially important in the setting of biofilms, where bacteria in the colony may be present in variable stages of cell division. It was seen that MRSA and E. coli biofilm cells were almost completely dead four days after exposure to CuNPs and S. aureus biofilm was reduced by 50% after 48 hours.23,27 In combination, CuNP’s many traits – biocompatibility and bactericidal action, innate ability to promote wound healing by stimulating anti-inflammatory effects and angiogenesis, and ability to act as a scaffold for other actives with a delayed release of ions – could be beneficial in the treatment of chronic and infected wounds.
Silver
There are commercial silver-based wound dressings which have been shown to have a positive impact on wound healing.3,11 In addition to their bactericidal properties, AgNPs allow for a higher surface area-to-volume ratio compared to topical silver compounds.1-3,11,31 This accommodates for innate silver cytotoxicity, a key consideration in its use, by requiring a lower concentration of silver to be used to augment wound healing.1,3,11 Silver creates action by promoting granulation tissue, collagen deposition, and wound contractility by differentiation of myofibroblasts from normal fibroblasts.2,6,11 Silver exercises its antimicrobial activity by deactivating respiratory enzymes after uptake into cells to generate reactive oxygen species to disrupt cellular processes.31 This ultimately controls biofilm growth.32 In combination with pharmaceutical antimicrobial agents, silver is able to exert additional antimicrobial activity as evidenced by accelerated healing seen in two different studies when in combination with tetracyclines and gentamicin.6,11
Inflammation is a key component of the wound healing cycle – chronic wounds are described as being caught in this phase. Silver is a key modulator of anti-inflammatory cytokines in addition to retaining antimicrobial activity.6,11 Silver has been shown to have activity against multi-drug resistant bacteria, making it a valuable wound care treatment in the setting of chronic wounds where patients have often exhausted conventional antibiotic treatments.2,6Additionally, AgNPs have activity as part of dressings and dermal substitutes.33 In a murine study looking at in vivo infection rates, 72 hours after application of the AgNP-loaded dressing, there was a decrease in bioburden to the wound.34 As previously stated, this was similarly seen in a rat model, where there was a decrease in inflammatory cells and increased fibroblast differentiation in granular wound beds after AgNPs topical application.13,34
In addition to the ability of AgNPs to be transferred to dressing materials without requiring chemical processing, silver may be combined with materials such as AgNO3 and reduced during the electrospinning process to AgNPs to have the aforementioned antimicrobial activity.35 AgNP-based dressings have few complications when applied to the wound base for extended periods of time, and showed compatibility with collagen scaffold.1,11,33 Similarly, on a copper-based framework, sustained release of silver ions generated reactive oxygen species leading to enhanced wound healing.29
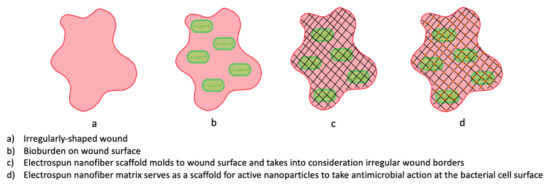 Electrospinning
Electrospinning
Electrospinning is a technique that permits control over the application of nanomaterials during the production of scaffolds, allowing them to retain properties similar to the extracellular matrix.3,9,11 It is a simple, cost-effective, flexible, and modifiable method of developing wound dressings from both synthetic and natural polymer solutions.36-38 Electrospinning is performed by an electrical potential being applied to a polymer liquid. At a critical voltage, the fluid is ejected and solvent evaporates leaving fibers ranging from 2nm to several micrometers in diameter. These fibers are then collected to form the electrospun nanofiber scaffolding used in active therapeutic dressings.36-40 As previously mentioned, in addition to being compatible with the wound bed surface, electrospun dressings are also versatile, allowing bioactive molecules and therapeutic agents to be incorporated into the nanofiber matrix.
Bioactive molecules and other therapeutic agents, such as antibiotics, may be combined with polymers by blending, surface modification, coaxial spinning, and emulsion processing.36,41 Blending requires that the drug is dissolved or dispersed in the polymer solution which encapsulates the bioactive therapeutic agent. The emulsion is then processed to develop the electrospun nanofiber. Depending on the interaction between and polarity of the bioactive agent and the polymer in solution, the active agent may be dispersed through the polymer or localized to the periphery resulting in a slow release or burst release, respectively.36 Surface modification involves the bioactive agent being adhered to the fiber surface, which makes it biochemically and structurally like native tissue and allows for a slow, controlled delivery. This is ideal in the setting of growth factors or other agents which act at stage-specific timepoints necessitating pulsed release.36 Co-axial electrospinning produces fibers with a core-shell appearance; the core is composed of the therapeutic agent and the shell is the polymeric matrix.36,41 This is especially beneficial in cases where sustained, local delivery of a therapeutic agent is desired; however, generating these complex structures poses a challenge. Emulsion electrospinning has the therapeutic agent and polymer emulsified together and electrospun in conjunction as compared to alone in co-axial electrospinning.36,41
An ideal drug delivery system should enable the therapeutic agent to have action and enhance its ability to act at the wound bed. Nanofibers have been successfully used as a drug cargo delivery vehicle for therapeutic agents such as antibiotics, metal nanoparticles, carbon materials, peptides, and natural extracts.42 Electrospun scaffolds are versatile – their innately small structure mimics the structure of the extracellular matrix and ability to be combined with bioactives augments their action at the wound bed.37,38,40 Electrospun nanofibers have select features which make them effective scaffolds for therapeutic agents including high surface area, high porosity, small pore size, morphological similarity to the extracellular matrix, and mechanical strength allowing for ease of handling.3,9,41,43
Electrospinning creates porous polymeric nanofibers which promote fibroblast adherence in wounds.3,11,39,40,43 One key advantage of electrospun dressings is the application process. Electrospun dressings are placed directly on the wound bed without having to touch the wound bed. Studies in porcine and rat models have found that, in combination with active nanomaterials, such as silver, copper and gold, electrospun dressings offer low cytotoxicity, antibacterial activity, and reduced inflammation, which together promote wound healing.11,14 Electrospun dressings may also be used as a network in which cellular markers are encapsulated to act at the wound bed. One such example is growth factors.44
Lai et al investigated a staged release of growth factors by an electrospun nanofiber dressing in the management of chronic wounds.44 A collagen-hyaluronic acid skin equivalent was designed to release various growth factors to promote epithelialization and re-vascularization of the wound bed. There was slow controlled release over one month which saw accelerated wound closure and more histologically normal collagen deposition. Leung et al confirmed that the ability to control the rate of drug release from a wound dressing by manipulating the release mechanisms allowed electrospun nanotechnology to play an active role in the wound healing process.9 Application of electrospun dressings in cases of chronic dermal ulcerations confirmed that nanofiber scaffold dressings can behave as both therapeutic and bioactive delivery vehicles.
As previously discussed, metal and metal oxide nanoparticles harbor the ability to disrupt bacterial cell membranes and ultimately cellular processes by generating reactive oxygen species. There is also some evidence to support loading these metal nanoparticles on to electrospun scaffolds as a means of delivery to the wound surface.41 There are biocompatible natural and synthetic polymers which are often combined to draw upon their individual benefits.3,9,38 With such actives in addition to the innate benefits of electrospun dressings, these combined dressings have antimicrobial effects, promote wound healing, induce angiogenesis, re-epithelialize, and promote granular tissue and collagen synthesis – all of which are key to the normal progression of wound healing and inhibiting wounds from being confined to the inflammatory stage of wound healing leading to chronicity.3,9,41,43,44
Natural polymers are derived from sources, such as silk fibroin, collagen, and polysaccharides found in the extracellular matrix of cells. Silk fibroin is a natural biopolymer fiber from silkworms and spiders with a repetitive amino acid sequence which allows it to form a biomaterial matrix.2,3,38 Wang et al investigated nerve regeneration using an electrospun silk scaffold. Their assessment of the scaffold’s ability to mimic a native ECM and provide support for regeneration of cells showed that silk fibers are biocompatible, biodegradable, and result in a low inflammatory response.2,45 The scaffold was also shown to have high levels of fibroblast cell attachment and to promote cell growth when functioning as a peripheral nerve graft.45 However, as a wound dressing, it is important to recognize that the solvent in which the silk is constituted during the electrospinning process is key to its success as a scaffold.38,45 For example, a previous study by the same authors found that in combination with a lactic acid backbone, silk was successfully electrospun into a matrix.45,46 When electrospun, nanofibers increase total surface area of an active molecule and allow for contact guidance for cell migration. In the setting of tissue regeneration, silk scaffolds have shown promising results in neural tissue, vascular tissue and epidermal tissue regeneration making it an ideal candidate in the dressing of chronic wounds.38 The unique properties of biocompatibility, biodegradability, flexibility, adherence, and absorption of exudates with minimal inflammatory reaction make silk a very promising material for wound dressings.2
Soy is a naturally derived plant-based biomaterial which has been successfully electrospun to form a functional scaffold. Soy has many reactive groups making it a strong candidate for chemical, physical, and enzymatic modification able to act at the wound bed.37 In a pig model, full thickness excisional wounds showed signs of re-epithelialization compared to the control group at two weeks after application of the electrospun soy scaffold dressing.47 Importantly, especially in the setting of chronic wounds, there was a decrease in the number of inflammatory cells noted. Dermal appendages, such as sweat glands and hair follicles, were present in the soy-treated wounds and absent in the control on histological exam, demonstrating that although electrospun soy scaffold did not accelerate wound healing, the quality of wound healing was augmented.47
In a comparison of polyethylene oxide (PEO) emulsions versus soy protein isolate emulsions, it was found that depending on the concentration of the mixture, the ability to be successfully electrospun changed.37 Soy + PEO scaffold showed significant upregulation in markers indicative of ECM deposition and remodeling.48 The scaffolding supported fibroblast adhesion, spread, growth, and proliferation in vitro.47,48 This study confirmed that trace amounts of PEO allowed for soy proteins to be successfully electrospun into a functional hydrolysis-resistant scaffold that supports wound bed healing and skin regeneration.37,48
Applications of Nanomaterials and Electrospinning
Nanoparticle-based treatments and therapies have been of recent interest largely due to their versatility. The ability of electrospun fiber dressings to be loaded with therapeutic nanoparticles allows them to function as a bioactive dressing tailored to address the deficiencies inherent in chronic wounds.9 There is a growing list of multi-drug resistant bacteria, and a high percentage of chronic wounds have a biofilm where resistance is transferred from bacteria to bacteria.52 The need for multifunctional dressings designed to target biofilms and reduce the formation of surface bioburden is great.
During production of nanoparticle-loaded electrospun fiber scaffold dressings a key consideration is the mechanism by which the therapeutic agent is released. Modifications to the electrospinning process to slow or pulse the release of the bioactive agents were previously mentioned. For this reason, it is imperative that the type, properties, and composition of the polymer used for electrospinning be considered when determining the active agent with which to combine its action.50 It is also important to note the effect of the nanoparticle upon healthy cells. While it is advantageous that many metallic nanoparticles induce bacterial cell death by generating reactive oxygen species to interfere with normal cellular processes, the effect upon surrounding healthy cells cannot be ignored. Ultimately, the goal of active dressings – in this case, bioactive-loaded electrospun nanofiber dressings – is to promote healing of the wound, highlighting the importance of understanding the toxicology associated with these novel materials. Of importance when considering these for use are the nanoparticle’s size, surface chemistry, surface physiology, the dose which is present in or on the dressing, the degree to which it produces free radicals, and its chemical components.51
Summary
Chronic wounds impose a heavy burden on our healthcare system and in concert with the bioburden these wounds carry, treatment proves challenging and expensive.51 The goal in treatment with nanoparticles, nanocomposites, coatings, and scaffolds is to provide a viable matrix for tissue reconstruction, increase the selectivity and efficacy of antimicrobials utilized, and to decrease the amount of patient exposure to strong, sometimes toxic anti-microbials.11,40 The ability to deliver therapeutic agents directly to the wound surface, reduce the concern for systemic exposure and toxicity, and promote the rate at which wounds epithelialize are all enhanced with the use of nanomaterials whether in particle or fiber form. Novel treatment options using bioactive therapeutic nanomaterials and electrospun nanofiber scaffolds are increasing in use and present a necessary and beneficial solution to the problems of toxicity and overall effectiveness.
James McGuire, DPM, LPT, LPed, FAPWHc, is Director of the Leonard S. Abrams Center for Advanced Wound Healing and Clinical Professor at Temple University School of Podiatric Medicine.
Avnee J. Patel, DPM, is a first-year resident at the University Hospital Podiatric Surgical Residency Program in Newark, NJ.
- Kushwaha A, Goswami L, Kim BS. Nanomaterial-based therapy for wound healing. Nanomaterials. 2022;12(4):618.
- Boateng J, Catanzano O. Advanced therapeutic dressings for effective wound healing—a review. Journal of Pharmaceutical Sciences. 2015;104(11):3653-3680.
- Berthet M, Gauthier Y, Lacroix C, Verrier B, Monge C. Nanoparticle-Based Dressing: The Future of Wound Treatment? ]. Trends Biotechnol. 2017;35(8):770-784. Correction: Trends Biotechnol. 2018 Jan;36(1):119.
- Martin P, Nunan R. Cellular and molecular mechanisms of repair in acute and chronic wound healing. Br J Dermatol. 2015;173(2):370-378.
- Nethi SK, Das S, Patra CR, Mukherjee S. Recent advances in inorganic nanomaterials for wound-healing applications. Biomater Sci. 2019;7(7):2652-2674.
- Vijayakumar V, Samal SK, Mohanty S, Nayak SK. Recent advancements in biopolymer and metal nanoparticle-based materials in diabetic wound healing management. Int J Biol Macromol. 2019;122:137-148.
- Clinton A, Carter T. Chronic Wound Biofilms: Pathogenesis and Potential Therapies. Lab Med. 2015;46(4):277-284.
- Percival SL. Restoring balance: biofilms and wound dressings. J Wound Care. 2018;27(2):102-113.
- Leung V, Hartwell R, Yang H, Ghahary A, Ko F. (2011). Bioactive nanofibres for wound healing applications. J Fiber Bioengineer Inform. 2011;4(1):1-14.
- Haik J, Kornhaber R, Blal B, Harats M. The Feasibility of a Handheld Electrospinning Device for the Application of Nanofibrous Wound Dressings. Adv Wound Care (New Rochelle). 2017;6(5):166-174.
- Mihai MM, Dima MB, Dima B, Holban AM. Nanomaterials for Wound Healing and Infection Control. Materials (Basel). 2019;12(13):2176.
- Lau P, Bidin N, Islam S, et al. Influence of gold nanoparticles on wound healing treatment in rat model: Photobiomodulation therapy. Lasers Surg Med. 2017;49(4):380-386.
- Naraginti S, Kumari PL, Das RK, Sivakumar A, Patil SH, Andhalkar VV. Amelioration of excision wounds by topical application of green synthesized, formulated silver and gold nanoparticles in albino Wistar rats. Mater Sci Eng C Mater Biol Appl. 2016;62:293-300.
- Gu H, Ho PL, Tong E, Wang L, Xu B. Presenting Vancomycin on Nanoparticles to Enhance Antimicrobial Activities. Nano Letters. 2003;3(9):1261–1263.
- Balaure PC, Holban AM, Grumezescu AM, et al. In vitro and in vivo studies of novel fabricated bioactive dressings based on collagen and zinc oxide 3D scaffolds. Int J Pharm. 2019;557:199-207. doi:10.1016/j.ijpharm.2018.12.063
- Koga AY, Felix JC, Silvestre RGM, et al. Evaluation of wound healing effect of alginate film containing Aloe vera gel and cross-linked with zinc chloride. Acta Cir Bras. 2020;35(5):e202000507.
- Lu MM, Bai J, Shao D, et al. Antibacterial and biodegradable tissue nano-adhesives for rapid wound closure. Int J Nanomed. 2018;13:5849-5863.
- Fontana F, Mori M, Riva F, et al. Platelet Lysate-Modified Porous Silicon Microparticles for Enhanced Cell Proliferation in Wound Healing Applications. ACS Appl Mater Interfaces. 2016;8(1):988-996.
- Colilla M, Vallet-Regí M. Targeted Stimuli-Responsive Mesoporous Silica Nanoparticles for Bacterial Infection Treatment. Int J Mol Sci. 2020;21(22):8605.
- Mori M, Almeida PV, Cola M, et al. In vitro assessment of biopolymer-modified porous silicon microparticles for wound healing applications. Eur J Pharm Biopharm. 2014;88(3):635-642.
- Mebert AM, Baglole CJ, Desimone MF, Maysinger D. Nanoengineered silica: Properties, applications and toxicity. Food Chem Toxicol. 2017;109(Pt 1):753-770. doi:10.1016/j.fct.2017.05.054
- Rose S, Prevoteau A, Elzière P, Hourdet D, Marcellan A, Leibler L. Nanoparticle solutions as adhesives for gels and biological tissues. Nature. 2014;505(7483):382-385.
- Ahire JJ, Hattingh M, Neveling DP, Dicks LM. Copper-Containing Anti-Biofilm Nanofiber Scaffolds as a Wound Dressing Material. PLoS One. 2016;11(3):e0152755.
- Borkow G, Gabbay J, Dardik R, et al. Molecular mechanisms of enhanced wound healing by copper oxide-impregnated dressings. Wound Repair Regen. 2010;18(2):266-275.
- Melamed E, Rovitsky A, Roth T, Assa L, Borkow G. Stimulation of Healing of Non-Infected Stagnated Diabetic Wounds by Copper Oxide-Impregnated Wound Dressings. Medicina (Kaunas). 2021;57(10):1129.
- LewisOscar F, MubarakAli D, Nithya C, et al. One pot synthesis and anti-biofilm potential of copper nanoparticles (CuNPs) against clinical strains of Pseudomonas aeruginosa. Biofouling. 2015;31(4):379-391.
- Agarwala M, Choudhury B, Yadav RN. Comparative study of antibiofilm activity of copper oxide and iron oxide nanoparticles against multidrug resistant biofilm forming uropathogens. Indian J Microbiol. 2014;54(3):365-368.
- Xiao J, Zhu Y, Huddleston S, et al. Copper Metal-Organic Framework Nanoparticles Stabilized with Folic Acid Improve Wound Healing in Diabetes. ACS Nano. 2018;12(2):1023-1032.
- Guo C, Cheng F, Liang G, et al. Copper-based polymer-metal–organic framework embedded with ag nanoparticles: Long-acting and intelligent antibacterial activity and accelerated wound healing. Chem Engineer J. 2022;435:134915.
- Zhang P, Li Y, Tang Y, et al. Copper-Based Metal-Organic Framework as a Controllable Nitric Oxide-Releasing Vehicle for Enhanced Diabetic Wound Healing. ACS Appl Mater Interfaces. 2020;12(16):18319-18331.
- Yin IX, Zhang J, et al. The Antibacterial Mechanism of Silver Nanoparticles and Its Application in Dentistry. Int J Nanomedicine. 2020;15:2555-2562.
- Radulescu M, Andronescu E, Dolete G, et al. Silver nanocoatings for reducing the exogenous microbial colonization of wound dressings. Materials (Basel). 2016;9(5):345.
- You C, Li Q, Wang X, et al. Silver nanoparticle loaded collagen/chitosan scaffolds promote wound healing via regulating fibroblast migration and macrophage activation. Sci Rep. 2017;7(1):10489.
- Guthrie KM, Agarwal A, Tackes DS, et al. Antibacterial efficacy of silver-impregnated polyelectrolyte multilayers immobilized on a biological dressing in a murine wound infection model. Ann Surg. 2012;256(2):371-377. doi:10.1097/SLA.0b013e318256ff99
- Pant B, Pant HR, Pandeya DR, et al. Characterization and antibacterial properties of AG NPS loaded nylon-6 nanocomposite prepared by one-step electrospinning process. Colloids and Surfaces A: Physicochemical and Engineering Aspects. 2012;395:94-99.
- Zamani M, Prabhakaran MP, Ramakrishna S. Advances in drug delivery via electrospun and electrosprayed nanomaterials. Int J Nanomedicine. 2013;8:2997-3017.
- Ramji K, Shah RN. Electrospun soy protein nanofiber scaffolds for tissue regeneration. J Biomater Appl. 2014;29(3):411-422.
- Bhardwaj N, Kundu SC. Electrospinning: a fascinating fiber fabrication technique. Biotechnol Adv. 2010;28(3):325-347.
- Zahedi P, Rezaeian I, Ranaei-Siadat SO, Jafari SH, Supaphol P. A review on wound dressings with an emphasis on electrospun nanofibrous polymeric bandages. Polymers Adv Tech. 2019;21(2):77-95.
- Patil JV, Mali SS, Kamble AS, Hong CK, Kim JH, Patil PS. Electrospinning: A versatile technique for making of 1d growth of nanostructured nanofibers and its applications: An experimental approach. Appl Surf Sci. 2017;423:641-674.
- Hamdan N, Yamin A, Hamid SA, Khodir WKWA, Guarino V. Functionalized Antimicrobial Nanofibers: Design Criteria and Recent Advances. J Funct Biomater. 2021;12(4):59.
- Alhusein N, Blagbrough IS, Beeton ML, Bolhuis A, De Bank PA. Electrospun Zein/PCL Fibrous Matrices Release Tetracycline in a Controlled Manner, Killing Staphylococcus aureus Both in Biofilms and Ex Vivo on Pig Skin, and are Compatible with Human Skin Cells. Pharm Res. 2016 Jan;33(1):237-46.
- Liu X, Lin T, Fang J, et al. In vivo wound healing and antibacterial performances of electrospun nanofibre membranes. J Biomed Mater Res A. 2010;94(2):499-508.
- Lai HJ, Kuan CH, Wu HC, et al. Tailored design of electrospun composite nanofibers with staged release of multiple angiogenic growth factors for chronic wound healing. Acta Biomater. 2014;10(10):4156-4166.
- Wang CY, Zhang KH, Fan CY, Mo XM, Ruan HJ, Li FF. Aligned natural-synthetic polyblend nanofibers for peripheral nerve regeneration. Acta Biomater. 2011;7(2):634-643.
- Zhang K, Wang H, Huang C, Su Y, Mo X, Ikada Y. Fabrication of silk fibroin blended P(LLA-CL) nanofibrous scaffolds for tissue engineering. J Biomed Mater Res A. 2010;93(3):984-93.
- Har-el Y, Gerstenhaber A, Brodsky R, Huneke RB, Lelkes PI. Electrospun Soy Protein Scaffolds as Wound Dressings: Enhanced Re-epithelialization in a Porcine Model of Wound Healing. Wound Med. 2014;5:9-15.
- Lin L, Perets A, Har-el YE, et al. Alimentary ‘green’ proteins as electrospun scaffolds for skin regenerative engineering. J Tissue Eng Regen Med. 2013;7(12):994-1008.
- Omar A, Wright JB, Schultz G, Burrell R, Nadworny P. Microbial biofilms and chronic wounds. Microorganisms. 2017;5(9).
- Gao Y, Bach Truong Y, Zhu Y, Louis Kyratzis I. Electrospun antibacterial nanofibers: Production, activity, and in vivo applications. J Appl Polymer Sci. 2014;131(18):40797
- Ai J, Biazar E, Jafarpour M, et al. Nanotoxicology and nanoparticle safety in biomedical designs. Int J Nanomedicine. 2011;6:1117-1127.
- Sen CK. Human Wounds and Its Burden: An Updated Compendium of Estimates. Adv Wound Care. 2019;8(2):39–48.