 By Cary Groner
By Cary Groner
In recent years, studies have elucidated aspects of the biomechanical effects of posterior tibial tendon dysfunction that may ultimately lead to improvements in treatment. But it isn’t always clear which factors contribute to PTTD and which
develop in response.
Posterior tibial tendon dysfunction (PTTD), often known as adult acquired flatfoot deformity, remains a therapeutic challenge, particularly if patients and their clinicians wish to avoid surgical intervention.
The condition, which most often afflicts overweight women during or after menopause, occurs in about 3.3% of women older than 40 years, according to a UK study,1 but often goes undiagnosed. PTTD may also occur as an acute overuse syndrome in young athletes,2 however, and whether this predisposes these patients to the chronic condition later in life is unclear.
In recent years researchers have begun to unravel some of PTTD’s mysteries, including poorly understood aspects of its etiology, biomechanical complexities, and response to treatment. Much more remains to be discovered, but clinicians agree that any progress is welcome.
Definitions
PTTD is typically defined as a progressive, degenerative musculoskeletal process that ranges from a localized tenosynovitis to a rigid foot, ankle deformity, or both.3,4 The condition leads to the elongation of the posterior tibialis tendon, which is the primary dynamic stabilizer of the foot’s medial longitudinal arch (MLA).3 The posterior tibialis muscle, to which the tendon attaches, also serves as an ankle plantar flexor and invertor of the subtalar joint.5 Researchers are investigating the role of the muscle in PTTD, but it remains unclear.
Elongation of the tendon is associated with increased pronation and subsequent functional imbalances, including lowering or collapse of the MLA, valgus calcaneal alignment, forefoot abduction, and related problems such as Achilles tendon contracture.4,6 These imbalances are also associated with stress on important foot ligaments, including the plantar calcaneonavicular (spring), talocalcaneal interosseous, and deltoid ligaments, though it remains unclear whether ligament problems precede or follow muscle weaknesses.3 Regardless, add it all together and you have impaired foot biomechanics and pain.
PTTD is typically described as having four stages.4,7,8 In Stage I, patients have posterior tibial tenosynovitis or tendinosis without structural deformity. Stage II disease often involves pain medially along the tendon early on, with later subfibular impingement and sinus tarsi pain; there is also a flatfoot deformity that remains flexible, as well as attenuation or rupture of the tendon. By Stage III, patients usually have a fixed nonreducible deformity with marked calcaneal valgus and midfoot abduction. In Stage IV, there is a flatfoot deformity with associated ankle involvement, including a valgus talar tilt due to deltoid ligament failure.
Even staging is more complex than it sounds, however.
“We have this concept that PTTD progresses from stage one to stage four, but actually it seems that stage one conditions are almost unique to athletes and younger people, whereas stage two is almost exclusively experienced by older, sedentary people,” said Jeff Houck, PT, PhD, director of research in the Department of Physical Therapy at George Fox University in Newburg, OR. “A lot of these patients have flat feet as a risk factor, but the progression is not that well established.”
Research: Hit and miss
As noted, in recent years some research has elucidated aspects of the biomechanical effects of PTTD that may ultimately lead to improvements in treatment.
For example, Canadian investigators compared differences in arch height, ankle muscle strength, and biomechanical factors in 24 runners, 12 of whom were healthy and 12 of whom had Stage I PTTD.9 The authors reported that runners with PTTD had lower arch height as well as greater and more prolonged peak rearfoot eversion during gait than those who were healthy, suggesting that increased pronation may put more strain on the posterior tibial muscle. In another study, the same research group used an exercise fatigue protocol to reduce the force of the same muscle; the protocol disrupted typical shank and foot joint coupling patterns and increased coupling variability.10
But, as already noted, one problem with studying and managing PTTD is that it isn’t always clear what affects what.
“Some people argue that the ligament is damaged first, then the tendons,” Houck said. “But it’s not easy to tell. A student of mine did a study that looked at PTTD patients—who was strong, who was weak, what their foot motion was.11 They were markedly different, suggesting that either the strength protected their foot posture and ligaments, or that the ligaments helped them stay stronger. We don’t know which it was; it’s just as common for weakness to be a response to a problem as the cause of it.”
PTTD’s prevalence in perimenopausal women has long provoked suspicion that the disease may be associated with metabolic or hormonal issues. But Penn State researchers in State College, PA, sampled tendon tissue from diseased tendons belonging to eight patients with PTTD requiring surgery (five women, mean age 52 years), then compared the tissue to samples from healthy tendons. There was no difference in estrogen receptor expression in the samples regardless of whether they were taken from normal or diseased tendons, or from male or female participants, undercutting (though probably not eliminating) speculation that hormonal changes in older women are a risk factor.12
Little if any research has been conducted on women’s shoes as a potential risk factor for the disease, even though male-female footwear differences—particularly high-heel wear—are increasingly suspect in other problems such as the gender disparity in rates of knee osteoarthritis.13 Of course, the biomechanics associated with high heels and OA are in some ways opposite to those found in PTTD; the former may be exacerbated by the shoes’ interference with pronation through the gait cycle, whereas the latter is worsened by excessive pronation. Researchers are skeptical, in any case, and a recent PubMed search found no published papers addressing the topic.
“The gender difference is puzzling, but it doesn’t seem to have to do with footwear,” Houck said. “The evidence for that is very weak.”
However, Michael Pinzur, MD, a professor of orthopedic surgery and rehabilitation at Loyola University Medical Center in Maywood, IL, noted that shoes may affect symptoms even if they don’t cause PTTD-related deformities.
“Generally, women’s shoes are not as supportive as men’s,” he said. “Just as many men have flat feet as women do, but they wear better shoes and tend to be less symptomatic.”
Various studies have looked at the relationship between PTTD and the kinetic chain. For example, researchers at the University of Southern California (USC) in Los Angeles reported that 17 middle-aged women with PTTD had decreased bilateral ankle and hip muscle performance—including being able to do fewer single-leg heel raises and leg lifts, and having lower endurance in the hip abductors and extensors—as well as worse performance and a significant increase in pain during a six-minute walk test, versus age-matched controls.14 The findings again bring up the chicken-and-egg conundrum that’s part of PTTD research, however: Did the women with PTTD walk slower because they were in more pain, and has slower walking contributed to their muscle weakness? Or did preexisting weakness slow down their walking?
“We found that patients were weaker on both sides even though [the PTTD] was a unilateral problem,” said Stephen Reischl, PT, PhD, one of that paper’s coauthors. Reischl, an adjunct associate professor of clinical physical therapy at USC, described the implications of this in terms of a possible biomechanical cascade.
“As the posterior tibialis fails, there may be more stress on the plantar fascia, which may fail,” he said. “Some of the deltoid spring ligament complex may fail; then the pull of the peroneus longus becomes a deformer of the foot rather than helping to stabilize the arch.”
Reischl noted that unilateral problems often have bilateral effects, as the USC team’s research showed.
“When a problem occurs on one side, something changes in the nervous system,” he said. “They walk slower, so the stimulus to the muscle decreases, and that happens in the entire lower extremity.”
One of Reischl’s colleagues, Eugene Chang, PT, PhD, who this summer completed his doctorate at USC, told LER that his dissertation examined just this issue—how local musculoskeletal disorders affect the control system, including the central nervous system (CNS).15
“It’s an adaptation on multiple levels to a localized disorder,” he said. “If the tendon is more compliant, the muscle has to work harder to compensate for the mechanical deficit. The CNS has to work harder to drive the muscle to compensate for the deficits.”
In unpublished research into patients with Achilles tendinosis, for example, Chang found that, even when the tendon problems were unilateral, patients tended toward biomechanical symmetry during bipedal activities.
“We had them run and looked at the kinematics—how they flexed their knees, ankles, and hips,” he said. “We saw a similar pattern on both sides; it looks like the uninvolved side tries to mimic the involved side. That has to be caused by the central nervous system, whether in Achilles tendinosis or PTTD.”
Neal Blitz, DPM, FACFAS, who is in private practice in New York City, also emphasized the importance of kinetic chain factors.
“If you internally rotate and have flat feet, that generally causes instability in the arch and unlocks the midfoot, and that causes the posterior tibial tendon to work harder to try to straighten up the arch,” Blitz said. “You get a tug-of-war between where the bones want to go and where the muscles are, and that’s where you get into trouble.”
Tibial internal rotation has been identified as a risk factor for PTTD, and in a 2011 article in this magazine, Christopher Neville PT, PhD, an assistant professor of physical therapy at SUNY Upstate Medical University in Syracuse, NY, said that such rotation was associated with several likely components of PTTD, including hindfoot eversion, lowering of the MLA, and forefoot abduction.16
Evaluation critical
Thorough patient evaluation is the first step in treatment, according to clinicians.
“To assess foot kinematics in the clinic, I tend to use the simplest tests—navicular drop, foot posture index, subtalar neutral,” said Houck, who added that he sees mostly older patients with Stage II PTTD. “I assess stability in the talonavicular and calcaneal-cuboid joints, and I think controlling the foot’s interaction with the tibia is important; it’s good to include functional retraining of the whole lower extremity when you’re working on the foot.”
Houck said most of his patients with PTTD have bilateral pronation or flatfoot, but that the mechanical issues are worse on the side with the tendinopathy.
“I tend to think of things like muscle volume and muscle strength as associated with the cascading effect of the problem, not as much the cause of it,” he said.
Neal Blitz said that evaluation is crucial, and can be challenging for clinicians who are not familiar with PTTD.
“One reason it’s often missed by primary physicians is that it has a wide variety of presentations,” he said. “Younger, athletic patients could have some swelling in the tendon that is self-limiting and goes away. But that tenosynovitis can also become chronic; there’s an inflammatory process that, with continued stress, can lead to tendon rupture and attenuation.”
When the clinician suspects a rupture, an evaluation with magnetic resonance imaging (MRI) is indicated.
“A longitudinal tendon split is a game changer,” Blitz said. “Those patients have to have some form of immobilization to allow the tendon to heal.”
Treatment options
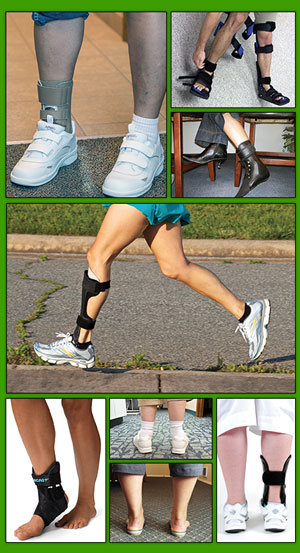
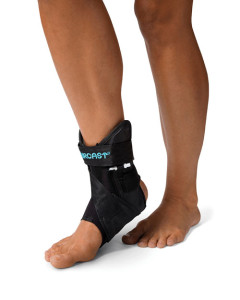
Although patients with advanced (Stage III-IV) PTTD often resort to surgery, several therapeutic approaches are available for those with less severe disease, or those who are poor candidates for surgical intervention. The standard strategy includes anti-inflammatory medication, in-shoe foot orthoses or ankle foot orthoses (AFOs), stretching and strengthening exercises, and physical therapy.4 The appropriateness of each modality is determined by clinician and patient preference, disease stage, the patient’s pathomechanics, and related issues.
“I tend to treat stage one patients with anti-inflammatories, rest, ice, and bracing, which can include simple ACE wraps or ankle straps,” Blitz said. “Sometimes, if it’s really tender, I’ll put them in a walking boot for anywhere from two to six weeks, assuming their muscle power is good.”
If patients have suspiciously high pain levels or weak muscles, Blitz may order an MRI to rule out tendon splits.
“There’s a bit of an art to PTTD treatment,” he continued. “You really have to be concerned about someone with a tendon split and flatfoot; their structure isn’t good and they’re putting a lot of stress on the tendon. An injured tendon heals as scar tissue, and if that happens repeatedly you’ve got problems. In those cases I may talk to them about surgery.”
Selene Parekh, MD, an associate professor of orthopedics at Duke University in Durham, NC, told LER he uses orthotic intervention and physical therapy in less severe cases.
“If patients have a weak posterior tibial tendon without a lot of malalignment issues, we can get them better by strengthening the tendon,” he said. “In those patients I’ll use a medial heel wedge orthotic, or sometimes a supramalleolar orthotic or an AFO. Most stage one patients will be successful with that, along with physical therapy and topical anti-inflammatories. I may treat stage two patients the same way, depending on their pain levels, or I may add an Arizona brace. If they fail therapy and bracing, they may end up in the operating room, as will those with significant malalignment, such as stage three and four patients.”
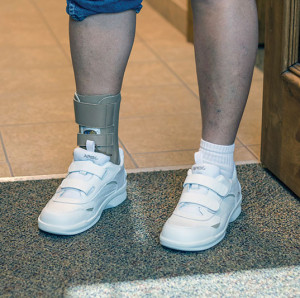
Strategies for orthotic intervention vary. For acute Stages I and II PTTD, the goal is to prevent the development of abnormal kinematics; patients may have the foot immobilized for three to six weeks in a cast or controlled ankle motion (CAM) boot, along with a foot orthosis inside the boot to control hindfoot eversion.8 For chronic PTTD Stages II to IV, possible approaches include UCBL (University of California Biomechanics Laboratory) and other foot orthoses, supramalleolar orthoses, and various types of AFOs.8
The literature generally supports the use of AFOs for treating chronic PTTD. In a 1996 study, investigators reported that 67% of patients with Stage I and II PTTD showed improvement in pain, function, and ambulation distance with a custom-molded AFO or medial-posted shoe insert.17 In 2008, researchers at Rush University Medical Center in Chicago reported results from a long-term study of 32 Stage II patients treated with a double-upright AFO. After an average follow-up of 8.6 years, 70% of patients were brace-free and had avoided surgery.18 In 2012, investigators from SUNY Upstate Medical University reported that custom articulated AFOs provided the greatest correction of flatfoot deformity while maintaining ankle motion and push-off function in 15 patients with Stage II PTTD.19
Jeff Houck agreed with those findings.
“Those patients don’t need to have the ankle fixed,” he said. “They just need arch support and hindfoot control. Long term, a solid-ankle AFO leads to calf-muscle atrophy and dependence on the device.”
AFOs are helpful by themselves but they can’t do everything. Correcting mechanical defaults will ideally affect the tendons and ligaments that support the foot, but some foot postures—forefoot abduction, for example—are harder to influence than others.20 As a result, clinicians increasingly seek to combine stretching, strengthening, and physical therapy with bracing to achieve optimal outcomes.
In one study, researchers treated 47 patients with Stage I and II PTTD using a short, articulated AFO or a foot orthosis as well as a physical therapy regimen that included a variety of strengthening exercises and stretches. After an average of 10 sessions over four months, 83% of patients had successful subjective and functional outcomes.21
In a 2009 study, USC researchers reported that Stage I and II patients improved the most with a combination of foot orthoses, stretching, and an eccentric exercise regimen focused on the posterior tibial tendon.22
This June, in a literature review presented at the World Congress of the International Society for Prosthetics and Orthotics in Lyon, France, researchers analyzed 10 studies and concluded that the most effective treatment for treating Stage I and II PTTD was a custom foot orthosis in conjunction with an exercise program that included stretches and eccentric strengthening. The protocols prevented progression and decreased the need for surgery.23
In a just-published randomized controlled trial, Houck and colleagues compared the effects of augmenting orthosis treatment with either stretching (n = 17) or a combination of stretching and strengthening (n = 19) in Stage II PTTD patients. Although both groups showed significant improvements in pain and function over the 12-week trial, the added interventions were minimally effective in augmenting orthosis wear alone.24
“That approach didn’t show a strong benefit,” Houck said. “I think it’s more important, for PTTD, to have an aggressive global program that really targets the tendon.”
Others have arrived at similar conclusions. According to Patrick McKeon, PhD, ATC, it’s crucial to look at systems, not just isolated aspects of them.
“We have always looked at PTTD as a dysfunction within the tendon,” said McKeon, who is an assistant professor and clinical education coordinator for the athletic training program at Ithaca College in New York State. “But it’s really a dysfunction in the system, and the tendon is caught in the middle. If you use orthotics alone, how will you encourage the tendon to adapt positively? We need to think about how to use orthotics and rehabilitation exercises together so eventually we can wean people off orthotics.”
McKeon and several colleagues have developed a program that focuses on strengthening the intrinsic muscles of the foot to improve lower extremity injury prevention and rehabilitation for conditions including PTTD.25,26
“We often see that PTTD occurs when something else isn’t doing what it’s supposed to do,” he said. “We’ve started to look into the role of the intrinsic foot muscles in helping to stabilize the arch. If we can rely more on those muscles, it should decrease stress on the posterior tibial tendon and help ensure that tissue breakdown doesn’t exceed the ability of the body to remodel it. Bahram Jam [a physical therapist at York University in Toronto] has published a model regarding PTTD and the role of the plantar intrinsics,27 and we hope these approaches may end up creating a more robust system that can cope with change.”
Looking forward
This “Foot Core System,” as Jam’s model is called, has yet to be tested extensively, but it represents a growing acceptance of the need for PTTD management to go beyond the status quo, given that the current nonsurgical therapeutic arsenal mainly manages the disease rather than curing it. Ideally, as researchers and clinicians continue to clarify the causes of PTTD and develop strategies to combat them, patients may find increasing relief from this vexing and complex condition.
Cary Groner is a freelance writer in the San Francisco Bay Area.
- Kohls-Gatzoulis J, Woods B, Angel JC, Singh D. The prevalence of symptomatic posterior tibial tendon dysfunction in women over the age of 40 in England. Foot Ankle Surg 2009;15(2):75-81.
- Ross JA. Posterior tibial tendon dysfunction in the athlete. Clin Podiatr Med Surg 1997;14(3):479-488.
- Houck J, Neville C, Flemister A. Kinematics of PTTD dictate management. LER 2009;1(5):31-39.
- Mendicino R, Catanzariti A, Peterson K. Adult acquired flatfoot: nonoperative options. LER 2012;4(1):45-51.
- Kitaoka HB, Ahn TK, Luo ZP, An KN. Stability of the arch of the foot. Foot Ankle Int 1997;18(10):644-648.
- Olszewski H. Evidence-based orthotic management of PTTD. LER 2011;3(11):25-31.
- Johnson KA, Strom DE. Tibial posterior tendon dysfunction. Clin Orthop 1989;239:196-206.
- Myerson MS. Instructional course lectures, the American Academy of Orthopedic Surgeons — Adult acquired flatfoot deformity. Treatment of dysfunction of the posterior tibial tendon. J Bone Joint Surg Am 1996;78(5):780-792.
- Rabbito M, Pohl MB, Humble N, Ferber R. biomechanical and clinical factors related to stage one posterior tibial tendon dysfunction. J Orthop Sports Phys Ther 2011;41(10):776-784.
- Ferber R, Pohl MB. Changes in joint coupling and variability during walking following tibial posterior muscle fatigue. J Foot Ankle Res 2011;4:6.
- Neville C, Flemister AS, Houck JR. Deep posterior compartment strength and foot kinematics in subjects with stage II posterior tibial tendon dysfunction. Foot Ankle Int 2010;31(4):320-328.
- Bridgeman JT, Zhang Y, Donahue H, et al. Estrogen receptor expression in posterior tibial tendon dysfunction: a pilot study. Foot Ankle Int 2010;31(12):1081-1084.
- Groner C. Footwear and risk of knee OA: The search for meaning in moments. LER 2015;7(7):18-27.
- Kulig K, Popovich JM Jr, Noceti-Dewit LM, et al. Women with posterior tibial tendon dysfunction have diminished ankle and hip muscle performance. J Orthop Sports Phys Ther 2011;41(9):687-694.
- Chang YJ, Kulig K. The neuromechanical adaptations to Achilles tendinosis. J Physiol June 5. [Epub ahead of print]
- Groner C. Rotational mechanics: bracing’s next frontier. LER 2011;3(10):29-36.
- Chao W, Wapner KL, Lee TH, et al. Nonoperative management of posterior tibial tendon dysfunction. Foot Ankle Int 1996;17(12):736-741.
- Lin JL, Balbas J, Richardson EG. Results of a non-surgical treatment of stage II posterior tibial tendon dysfunction: a 7 to 10 year follow-up. Foot Ankle Int 2008;29(8):781-786.
- Neville CG, Lemley FR. Effect of ankle-foot orthotic devices on foot kinematics in stage II posterior tibial tendon dysfunction. Foot Ankle Int 2012;33(5):406-414.
- Groner C. AFO choices for PTTD grow clearer. LER 2010;2(12):76.
- Alvarez RG, Marini A, Schmitt C, Saltzman CL. Stage I and II posterior tibial tendon dysfunction treated by a structured nonoperative management protocol: an orthosis and exercise program. Foot Ankle Int 2006;27(1):2-8.
- Kulig K, Lederhaus ES, Reischl S, et al. Effect of eccentric exercise program for early tibial posterior tendinopathy. Foot Ankle Int 2009;30(9):877-885.
- Phimister H, Figgins E. Conservative management of stage I and II posterior tibial tendon dysfunction. Presented at the 15th World Congress of the International Society for Prosthetics and Orthotics, Lyon, France, June 2015.
- Houck J, Neville C, Tome J, Flemister A. Randomized controlled trial comparing orthosis augmented by either stretching or stretching and strengthening for stage II tibialis posterior tendon dysfunction. Foot Ankle Int 2015 April 9. [Epub ahead of print]
- McKeon P, Hertel J, Bramble D, Davis I. The Foot Core System: a new paradigm for understanding intrinsic foot muscle function. Br J Sports Med 2015;49(5):290.
- McKeon P, Fourchet F. Freeing the foot: integrating the Foot Core System into rehabilitation for lower extremity injuries. Clin Sports Med 2015;34(2):347-361.
- Jam B. Evaluation and retraining of the intrinsic foot muscles for pain syndromes related to abnormal control of pronation. Advanced Physical Therapy Education Institute website. http://www.aptei.com/articles/pdf/IntrinsicMuscles.pdf. Accessed July 30, 2015.












I have a patented novel treatment for Grade I-II PTTD that requires research for its efficacy, and am looking for a principle investigator from an institution to undertake this research. My article is found in the Dec. 2017 issue of the Journal of the America Podiatric Medial Association. My website Stickmanhq.com describes the treatment and the theory behind it.