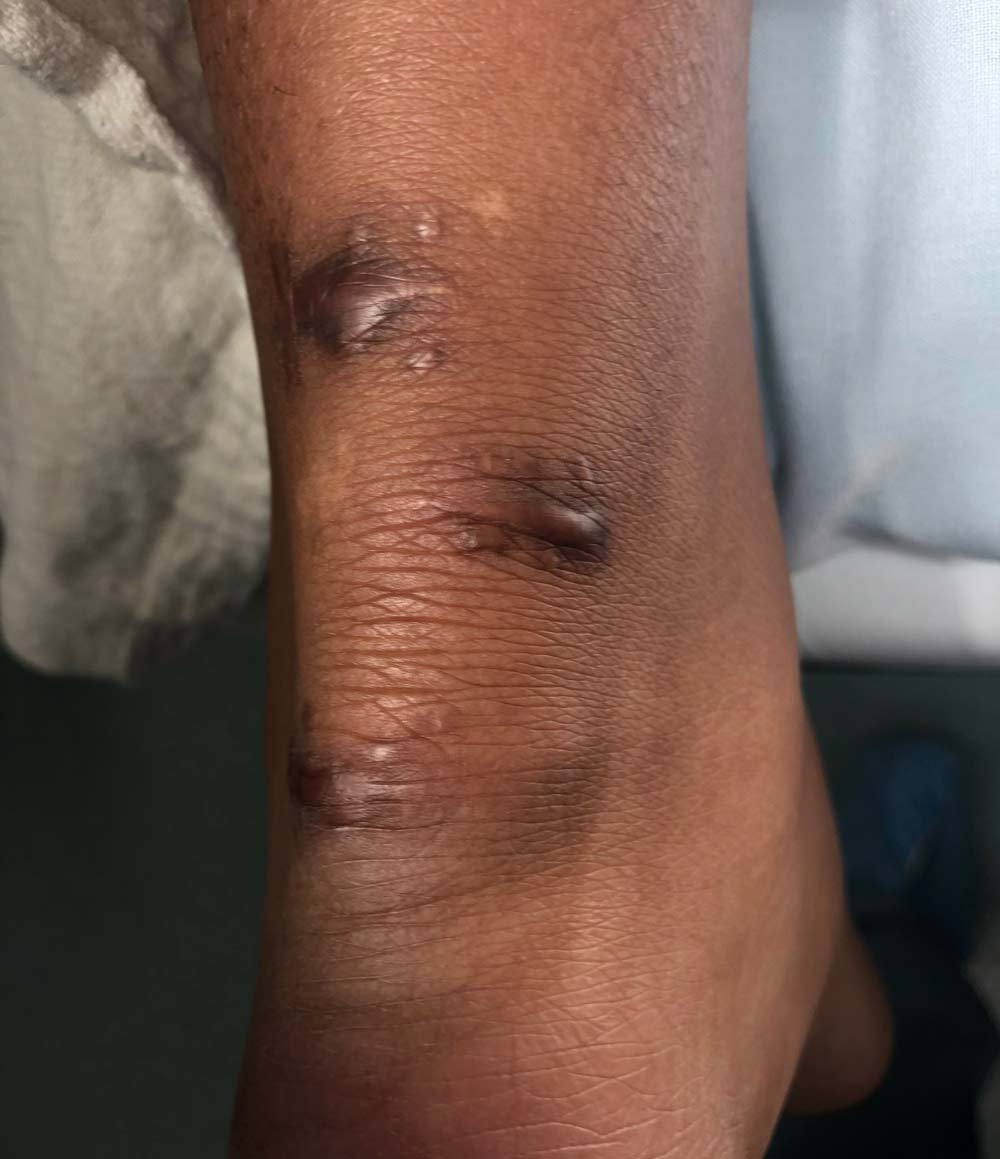
Managing patients with hypertrophic scars can be challenging, with a variety of conservative treatment options suggested in the medical literature. This review summarizes conservative treatment options for hypertrophic scars, focusing on the mechanisms of action, risks, complications, and efficacy of various options.
 By Laura-Ashley O’Connell, DPM; Adrienne Estes, DPM, MS, AACFAS, FAPWHc; and David Shofler, DPM, MSHS
By Laura-Ashley O’Connell, DPM; Adrienne Estes, DPM, MS, AACFAS, FAPWHc; and David Shofler, DPM, MSHS
Hypertrophic scarring is a cosmetically displeasing result that is symptomatic of wound healing disruption. Painful scarring is relatively common after surgical intervention, with 40% – 70% of patients experiencing some level of excess scar tissue formation.1 Hypertrophic scar formation is typically apparent by 4–8 weeks after skin injury. Scarring may occur on any body part after dermal trauma, but hypertrophic scarring most commonly affects the ankles, knees, shoulders and pre-sternal skin. Postsurgical scarring can be particularly painful on the legs and feet due to the rubbing caused by clothing, footwear, and ambulation.
Hypertrophic scar tissue is visibly elevated with itching, pain, or discoloration and typically indicates the wound is stalled in the inflammatory phase of healing. Hypertrophic scars are characterized by their elevation of less than 4mm with red/pink coloration. They do not extend past their geographic margin and tend to regress spontaneously when the tissue enters the remodeling phase of healing.2 They mostly consist of Type 3 collagen that forms parallel to the epidermis.3 The undesirable appearance may lead to psychological effects on the patient including anxiety and depression.
Hypertrophic scars are most commonly seen in children and young adults, age 10–30 years. This group is more prone to trauma and has a greater quantity of elastic fibers in the skin, which increases tension and causes irritation with scar formation. A greater rate of collagen synthesis occurs in this population as well, enabling increased formation of scar tissue.3 Hypertrophic scarring is more common in patients with darker skin pigmentation, with a prevalence of 4.5% – 16% in the African American and Hispanic populations.4 Hypertrophic scars are common in burn patients and after surgery. Other risk factors include local inflammation and personal or family history of hypertrophic scarring.
Wound healing occurs in 4 phases: hemostasis, inflammation, proliferation, and remodeling. Initially, platelets control bleeding and a clot becomes a fibrin scaffold for cellular migration and ingrowth for tissue formation. Inflammatory cells secrete growth factors and cytokines, which initiate cellular proliferation. Devitalized tissue is removed with this process, as recruited monocytes differentiate into macrophages. The proliferation phase consists of angiogenesis and collagen synthesis, with keratinocytes migrating into the dermis to form granulation tissue. Myofibroblasts proliferate at wound margins with actin filaments to contract and bring the wound edges together. Keratinocytes promote growth of new tissue from the periphery, in toward the center of the wound. The remodeling phase converts healing tissue into mature tissue with degradation of excess components and extracellular matrix while type 3 collagen is converted to type 1.5,6
Disruptions of unknown origins that prolong the inflammatory phase cause increased fibroblast activity and production of excessive extracellular matrix by fibroblasts. Angiogenesis upregulation by epidermal cells via vascular endothelial growth factor (VEGF) causes excess capillary formation, excess type 1 collagen production, and increased scar volume. The remodeling phase never occurs, inflammation and proliferation continue, and excessive extracellular matrix is deposited.3 This results in an enlarged, fibrotic tissue mass at the site of the wound, which can become painful, discolored, and problematic.6
The most effective method for prevention of hypertrophic scars is to reduce inflammation and tension on the wound.4 All devitalized tissue should be debrided to prevent interference with healthy tissue ingrowth. Hemostatic dressing should be applied to prevent hematoma or seroma. Primary closure should be tension-free, with all incisions along resting skin tension lines when possible. Suture material should be of the appropriate strength with an acceptable number of sutures to reduce mechanical stress. Inert materials should be used in suturing and wound care. If epithelialization is delayed greater than 2 weeks, there is an increased risk of hypertrophic scar formation.5,6
Surgical treatment of scarring by debulking can be used to reduce scar mass, although it may also lead to recurrence or worsening of scar tissue due to increased inflammation and prolonged healing time. The patient must endure an additional procedure with risk of increased pain, as well as financial and emotional costs. Many nonsurgical treatments have been evaluated. While some have been proven effective, others have been shown to be detrimental to scar healing. The purpose of this review is to describe nonsurgical treatment options for symptomatic hypertrophic scar formation. The focus in this article will be to review the mechanism of action and outcomes of current and newly emerging treatment options.
Treatments
Pressure Therapy
The function of pressure therapy is to reduce tension on the wound surface and prevent motion between wound edges to allow healing. Pressure dressings are noninvasive, widely available, and inexpensive. This makes them an especially useful treatment option in impoverished areas. Patient compliance, however, is reported to be an issue with these dressings, as they can be irritating in warm climates and produce rashes. It is also important to obtain an appropriate level of pressure with compression to prevent wound formation or contracture deformity.2,7,8
It has been suggested that pressure applied to blood vessels limits flow and prevents an increase in inflammatory cytokine concentrations to the wound site.1 It has also been suggested that physical compression on an area promotes tissue remodeling and alters activity of matrixmetalloproteinase-28 (MMP-28). This facilitates scar maturation and reduces the thickness of scars. Studies demonstrate no significant difference between high and low pressure, although there is an optimal pressure range which should be targeted; pressures of 20 – 40mmHg are recommended.7 Pressures greater than 25mmHg are needed to overcome capillary pressure and reduce flow sufficiently. Pressures less than 40mmHg are recommended to prevent ischemia and possible paresthesia. Application is suggested for 23 hours per day for a period of at least 6 months.1 This requires significant commitment from the patient for proper application and regimen compliance. It is notable that pressure dressings may also be used prophylactically for prevention of hypertrophic scar tissue formation.8
Candy et al7 evaluated scar reduction with varying degrees of pressure application. The design was a double-blind, randomized controlled clinical trial with 53 hypertrophic scars in 17 patients. Scars were randomly assigned to one of two groups: 25 scars were assigned to the low-pressure (10 – 15mmHg) group; 28 scars were assigned to the high-pressure (20 – 25mmHg) group. Garments were worn 23 hours per day for 5 months with monthly assessments. Both groups demonstrated significant decreases in scar thickness over the 5-month period. Notably, the high-pressure group had a significantly greater improvement, with a 40% reduction in scar thickness. Both groups had a significant reduction in redness, while only the high-pressure group had significant decrease in pigmentation. Overall, a pressure of at least 20mmHg improved scar remodeling over the study period. It should be noted that garments in this study were noted to stretch with time and reduce with pressure applied, so they were replaced every 2 months.
Several additional studies have described the benefits of pressure garments.9,10 However, randomized trials on the topic are limited. Overall, pressure therapy may be considered a potentially effective, noninvasive, and inexpensive method for hypertrophic scar reduction with limited potential of complications.
Silicone Gel and Sheeting
 Silicone gel is transparent, easy to apply, readily available, and stores well for over-the-counter use. It is applied as a thin layer to the wound site twice daily. While the mechanism of action is poorly understood, it is believed that the pressure and increased temperature with application helps to promote collagen breakdown.2 The silicone also provides a hydrating layer to the wound surface, preventing water evaporation and maintenance of a moist wound environment.2,11 While the gel may be slow to dry, a hair dryer may help expedite the delay.12
Silicone gel is transparent, easy to apply, readily available, and stores well for over-the-counter use. It is applied as a thin layer to the wound site twice daily. While the mechanism of action is poorly understood, it is believed that the pressure and increased temperature with application helps to promote collagen breakdown.2 The silicone also provides a hydrating layer to the wound surface, preventing water evaporation and maintenance of a moist wound environment.2,11 While the gel may be slow to dry, a hair dryer may help expedite the delay.12
Signorini and Clementoni12 performed a prospective randomized controlled trial that included 160 surgical patients divided into two groups: in group 1, 72 were treated with topical silicone gel applications twice daily for 4 months, while group 2 served as the control. The silicone gel group reported no adverse reactions (rash, irritation, or infection). At the end of the trial, scars were graded 1 – 4: Grade 1, normal appearing skin; Grade 2, mildly hypertrophic; Grade 3, hypertrophic, and Grade 4, keloid. In the silicone gel group, 93% of patients had Grade 1 or 2 scar results. In the control group, 46% of patients had Grade 2 scar results and 26% had either Grade 3 or 4 scar results. The difference in scar quality between the 2 groups was statistically significant. This study concluded that the silicone gel was effective in reducing hypertrophy of new surgical scars.
Silicone sheets, which are thicker and a bit stiffer to shield the wound from the environment, are also available for scar treatment with the same proposed mechanism of action. The sheets are translucent and discreet, but may be cumbersome in more exposed body areas, and patients have reported noncompliance, especially in more exposed body areas.5
In a prospective, randomized controlled clinical trial by Gold et al,11 96 patients were divided into two groups: those at high risk for hypertrophic scar formation (defined as scarring in the past), and those at low risk for hypertrophic scar formation (no personal history of scarring). After surgery, patients were randomized to either routine postoperative care or silicone gel sheet application 48 hours post-operation. Patients were followed for 6 months. Thirty patients were lost to follow up, leaving 66 patients: 35 in the high-risk group and 31 in the low-risk group. At the final follow up, there were no significant differences in the low-risk group between those who received gel sheeting and those who received routine wound care. One patient receiving routine wound care developed a hypertrophic scar, while none of the gel sheeting patients developed a hypertrophic scar. Among high-risk patients, 39% of those who received silicone gel sheeting and 71% of the routine postoperative care group developed hypertrophic scars. Although the result was not statistically significant, it could be considered clinically notable.
Overall, silicone may be a useful option in preventing hypertrophic scar formation, especially among those with a history of uncontrolled scarring. Nonetheless, additional high-quality studies would help affirm the benefits of this option.
Corticosteroids
Corticosteroids have an anti-inflammatory effect that reduces collagen synthesis and degeneration, glycosaminoglycan production, and fibroblast proliferation. Corticosteroids also cause vasoconstriction via topical binding to glucocorticoid receptors to assist in reducing inflammation.13 Triamcinolone acetonide (typically Kenalog®, Bristol-Myers-Squibb) is a commonly used corticosteroid for the treatment of hypertrophic scars. An injection of 10 – 40mg/mL triamcinolone acetonide is performed every 2 – 4 weeks until the area has flattened sufficiently.13 This treatment can be combined with other therapies, most commonly with interferon, 5-Fluorouracil, verapamil, laser therapy, or cryotherapy. Potential adverse reactions of corticosteroid injection use include skin and fat atrophy, dyschromias, telangiectasias, and injection site pain. Variable resolution rates have been reported.13,14 For these and other reasons, such injections may be contraindicated children.
In a study by Chowdri et al,15 58 patients, 37 with keloids and 21 with hypertrophic scars, had their lesions excised. Patients received triamcinolone acetonide injections intraoperatively, 2 – 5 times weekly, then monthly for 4 – 6 months. Injection dosages ranged from 20 – 120mg per dose, which were calculated based on lesion size and patient age. Patients were followed for a minimum of 1 – 2 years postoperatively. Response to treatment was objective, based on recurrence. Fifty-four of the patients (93.1%) had no recurrence, while 3 had partial recurrence and 1 had full recurrence. None had recurrence worse than the initial preoperative state. The most common adverse reactions included pain, injection-site dermal atrophy, and hypopigmentation. It should be noted that dermal atrophy persisted after the 2-year follow-up visit, but hypopigmentation was noted to fade completely after 9 months.
An emerging method of intralesional steroid delivery includes laser assistance. Fractional ablative lasers can function at variable skin depths, allowing for assistance with wound healing, promoting collagen response, and delivering medications to deeper dermal layers. Triamcinolone acetonide is applied topically as a suspension and is delivered with a fractional ablative laser immediately postoperatively. In a study by Waibel et al,16 15 patients with hypertrophic scars from burns, surgery, or trauma were evaluated. Patients received 3 – 5 treatments every 2 – 3 months consisting of fractional ablative laser therapy, followed by topical application of 10 or 20mg/mL triamcinolone acetonide suspension. Patients were instructed to soak with acetic acid daily and use a moisturizer 3 times daily after treatment. Visual evaluation of the scars was performed at baseline and compared to results 6 months later. Average overall improvement was 2.73/3.0 on the modified Manchester quartile score. Texture improved most dramatically, with 12 out of 15 patients achieving a perfect score. All patients saw improvement in hypertrophy. Dyschromia improvement scores were more modest, although 7 patients received the highest possible score. Eleven out of 15 patients achieved a perfect score in overall improvement.
Corticosteroid therapy, whether via intralesional injection or with laser-assisted delivery, is supported as an effective treatment method for hypertrophic scarring with minimal potential adverse reactions.
Vitamin E oil
Vitamin E, also known as alpha-tocopherol, is a lipid-soluble antioxidant compound that protects cells from oxidative stress by decreasing peroxidation of lipids, stabilizing their membranes by reducing free radical production by neutrophils. Vitamin E is able to penetrate to the deep dermis and reticular tissue, supporting topical use. Systemically, vitamin E inhibits the inflammatory response, decreases collagen synthesis, and decreases the tensile strength of tissue.2 Vitamin E topical oil is inexpensive, readily available, and easy to apply. Disadvantages include reports of the potential of delaying healing. Adverse reactions include worsening of scar appearance, contact dermatitis, and eczema.
In vitro studies have demonstrated potential efficacy, but in vivo studies have not been as favorable.2 In a double-blinded study by Baumann et al,17 15 patients were evaluated post-Mohs surgery. The surgical site was divided into 2 sides: 1 received Aquaphor application twice daily for 4 weeks, and the other received supplemental vitamin E application with Aquaphor. Five out of the 15 patients experienced local erythema, irritation, rash or itching, and 4 discontinued the treatment as a result. There was no significant difference in appearance between the 2 sides, although authors noted clinically better appearance in the Aquaphor-only side. Appearance was not quantitative and was defined purely by expert opinion visually.
Vitamin E topical oil may flatten the scar and promote hydration, but it can also decrease scar strength and leave healed epidermis thin and vulnerable. There is little evidence to support that it may accelerate healing or improve scar appearance, and the adverse effects may outweigh the benefits.
Bleomycin
 Bleomycin is a cytotoxic antineoplastic, antiviral, and antibacterial agent that inhibits DNA synthesis. It is a secondary metabolite of Streptomyces verticillus and is often used to treat warts. It functions by decreasing TGF-b1, which reduces collagen synthesis by fibroblasts. It also reduces levels of lysyl oxidase to influence collagen maturation and induces apoptosis to promote the remodeling phase of wound healing. As a treatment for hypertrophic scars and keloids, bleomycin is typically injected as 0.1mL, with quantities 1.5IU/mL monthly for 2 – 6 sessions total.5
Bleomycin is a cytotoxic antineoplastic, antiviral, and antibacterial agent that inhibits DNA synthesis. It is a secondary metabolite of Streptomyces verticillus and is often used to treat warts. It functions by decreasing TGF-b1, which reduces collagen synthesis by fibroblasts. It also reduces levels of lysyl oxidase to influence collagen maturation and induces apoptosis to promote the remodeling phase of wound healing. As a treatment for hypertrophic scars and keloids, bleomycin is typically injected as 0.1mL, with quantities 1.5IU/mL monthly for 2 – 6 sessions total.5
Bleomycin is easy to administer and is relatively inexpensive. However, reported adverse reactions include injection-site pain, ulceration, atrophy of surrounding tissue, and hyperpigmentation. Systemic symptoms are not typically seen with cutaneous injections, although toxicity is noted to be 200 – 300IU after total dosing (pulmonary fibrosis may occur at doses more than 400IU.)18 Systemic symptoms include Raynaud’s phenomenon, gangrene, neutrophilic eccrine hidradenitis, alopecia, hyperpigmentation, scleroderma or pulmonary fibrosis in exceedingly high levels of exposure.
Outcomes for use in hypertrophic scars and keloids have been demonstrated to be fairly good, with complete flattening in 54% – 73% of keloid patients19 in a study featuring 14 patients with 15 keloids or hypertrophic scars that were unresponsive to corticosteroid injections, pressure therapy, surgical excision, or cryotherapy. Authors used a needle-free injector to administer multiple bleomycin injections for a total of 0.4mL/cm2 every 4 weeks. Treatment continued until cosmetic appearance was acceptable (2 – 6 sessions over 16 – 24 months). Scars were evaluated based on scar thickness, scar pliability, erythema, and self-assessment. After treatment, 73.3% demonstrated complete flattening and 20% showed significant flattening, while 93% experienced complete softening. Eighty-seven percent of patients had no erythema. Temporary mild-to-moderate pain was noted after injections in half of the patients. Other side effects included hyperpigmentation, dermal atrophy, ulceration, and crusting.
Jet injections were also used to good benefit in a clinical trial with 50 hypertrophic scar and keloid patients. Injections were performed every 15 days for 3 rounds of injections, followed by a 2-month break, and then a fourth injection.20 Two months passed prior to final evaluation. A total of 44% of patients had complete flattening, 22% had significant flattening, 14% had adequate flattening and 20% had no improvement. Seven patients had recurrence of the lesion. The most commonly noted adverse reaction was itching.
Overall, bleomycin has the potential of yielding high regression, low recurrence, and few adverse reactions.20 Nonetheless, additional investigations would be helpful in confirming these findings.
Cryotherapy
Cryotherapy is performed with liquid nitrogen and delivered via intralesional cryoprobe needle, spray, or direct contact, thereby freezing the scar from the center outwards, producing an outcome similar to frostbite. This frostbite-type vascular damage (as opposed to burn injury) causes tissue necrosis, which promotes secretion of cytokines to alter the response of fibroblasts, reducing hypertrophic tissue formation. It is recommended to freeze the scar, and 5 – 10mm of uninvolved perilesional tissue. The literature reports variable success rates from 32% – 74% after multiple treatment sessions.5 It has been noted that hypertrophic scarring improved to a greater extent when cryotherapy was combined with corticosteroid injections.5
O’Boyle et al21 performed a review that evaluated 15 articles, which were all Level 4 (low evidence base) studies. Data regarding a total of 334 patients with 398 scars were reported. Overall, patients experienced 63% mean scar volume reduction and 58.5% mean surface area reduction. Recurrence rates were reported to be 0% – 24%. Mourad et al22 had demonstrated that intralesional delivery had statistically significantly better results than spray and direct contact administration Patients experienced decreased deformity, anxiety, pain, and itching. Some patients reported hypopigmentation, increased pain, prolonged healing, and recurrence. It was concluded that cryotherapy was a safe technique to produce effective scar reduction in few treatments.21
Botulinum Toxin A
Botulinum toxin A is a neurotoxin produced by Clostridium botulinum that blocks neuromuscular transmission. This blockade decreases muscle and skin tension via paralysis, stimulates collagen formation, and inhibits DNA synthesis during scar healing, causing the fibroblast cell cycle to pause in a non-proliferative stage. Lee and Jang reviewed several studies and found the standard protocol would have patients receive an injection of 70 – 140IU of the toxin per session every 1 – 3 months for a total of 3 – 9 months, making 3 sessions total.5,25 Treatment was usually performed as an adjunctive therapy to other conservative treatment options.
In a prospective clinical study that specifically looked at the use of botulinum toxin A in hypertrophic scars, Xiao et al23 randomly assigned 19 patients to receive 2.5U/cm3 botulinum toxin A injections monthly for 3 months, with a minimum follow up of 6 months. Patients and surgeons subjectively evaluated the result. Patients reported improved scores for redness, itching, and pliability as well as high rates of therapeutic satisfaction. Seven patients reported they had excellent outcomes, while the surgeons identified excellent outcomes in 4 patients. Twelve patients reported good outcomes while surgeons reported 15 patients with good outcomes. Overall, both surgeons and patients were satisfied with the result of botulinum toxin treatment alone.
Huang et al24 performed a split-face, double-blind, randomized controlled trial with 43 patients undergoing epicanthoplasty. Patients received either 5U botulinum toxin A or saline injection to the surgical site area of either eye at 6 – 7 days post-operatively at the time of suture removal. Patients were followed for a minimum of 6 months. A total of 30 patients completed the trial. The most notable improvements were seen at the 3-month follow up appointment, with results becoming more equivalent between eyes by the end of the 6-month follow-up period. Approximately 86% of patients perceived improved outcomes with botulinum treatment. No severe complications were reported. although one patient experienced temporary lid droop, which resolved after 6 weeks. Early application of botulinum toxin A can be used to prevent post-operative hypertrophy with good patient satisfaction and minimal complications. As the effects were noted to assimilate between sides by the 6-month follow up, it should be noted that the positive effects of botulinum toxin A may be temporary.
Kim et al25 performed a double-blind randomized controlled trial with 45 patients who had presented to the emergency room with forehead lacerations. Wounds were closed by 2 plastic surgeons and patients were sent home with instructions to report to a wound clinic every other day for a week. On postoperative day 5, sutures were removed, and the patients were randomized for the study: 24 patients received dermal injections of 5IU/cm botulinum toxin A at several points around the scar, while 21 received saline injection in similar locations. Follow up occurred at 1, 3, and 6 months postoperatively. On histological examination, there was a higher density of collagen fibers present in the control group, supporting the hypothesis that botulinum toxin A has favorable effects on scar formation. Elasticity, pigmentation, and erythema were more similar to normal skin than the control group. Improvements in scar width, height, pigment, pliability, and discomfort were statistically significant compared with the control group, but there was no significant difference in observer and patient scar assessments. There were no reported adverse reactions from the treatment.
Overall, botulinum toxin A injections may be an efficacious treatment for the prevention of hypertrophic scarring, with moderate benefit demonstrated and a low incidence of adverse events. It is questionable whether the results achieved by botulinum toxin A may diminish with time, thus a quality prospective study with long-term follow up is needed to determine significance.
5-Fluorouracil
5-Fluorouracil is a chemotherapeutic agent that acts to prevent angiogenesis and fibroblast proliferation by inhibiting DNA synthesis in tumor cells. It also prevents expression of collagen type 1 that is induced by TGF-beta.26 5-Fluorouracil is typically delivered as an intralesional injection of 50 – 150mg that is performed weekly for 12 weeks.27 It can be used alone or in combination with corticosteroid injections. Higher success rates have been reported when delivered with corticosteroid injection, due to significant pain when used alone.3
In a comprehensive review of 21 publications, Shah et al26 evaluated 5-fluorouracil in the treatment of hypertrophic scarring. Doses administered were 50 – 100mg per injection with 2 – 16 injections total at weekly or biweekly intervals. Follow up ranged from 6 – 18 months. Most patients were found to have had good-to-excellent flattening of lesions (typically 68% – 90%). Low recurrence rates were reported by all the authors included in the review, with a majority demonstrating zero patients with recurrence. Adverse reactions included pain in 95% – 100% of patients, along with ulceration, itching, superficial necrosis, tissue slough, hypopigmentation, and infection, though at much lower rates. Decreased incidence of adverse events overall was reported when 5-fluorouracil was combined with corticosteroid. Due to the high rate of side effects, it has been recommended that 5-fluorouracil be used with a corticosteroid, and only in patients who have failed other therapies.26
Cannabinoids
Cannabinoids can be divided into 3 groups based on their origins: phytocannabinoids from plants, endocannabinoids produced within the body, and laboratory-produced synthetic cannabinoids. Cannabidiol acts as an agonist at CB1 and CB2 receptors throughout the body. These receptors have differential binding to THC, cannabidiol, and endocannabinoids, based on their location. Studies have been performed recently to evaluate the effect of cannabidiol in neurologic and anti-inflammatory conditions, as CB1 and CB2 receptors are noted along sensory nerve fibers, inflammatory cells, and in adnexal structures. It is proposed that cannabinoids may have some utility as topical medications in wound healing.28
CB1 agonists have anti-inflammatory properties and may therefore reduce pain. In a study performed by Kim et al,29 wounded mice treated with topical CB1 agonists demonstrated greater recovery of epidermal barrier function, as well as significantly decreased skin fold thickness in chronic and acute inflammation. Topical CB1 agonists have been shown to decrease mast cell recruitment, blood histamine, inflammatory mediators, and myeloid immune cell infiltration.28 CB1 and CB2 agonists have an antinociceptive effect as well, which has been shown to be dose-dependent in mice models. It was demonstrated that topical CB1/CB2 agonists plus morphine reduced pain more effectively than topical morphine alone. 29 This finding supports that topical cannabinoids can be used to reduce pain.
The CB2 receptor is expressed by effector cells in the skin during wound healing. This, combined with its known involvement in inflammation, fibrogenesis, and cellular proliferation, led some to believe it could have a role in the regulation of skin wound healing. To test that hypothesis, Wang et al30 treated mice excisional wounds with a topical CB2 agonist and analyzed presence of inflammatory infiltrates, expression of cytokines, fibrogenesis, and wound re-epithelialization. It was found that CB2 receptor activation decreased neutrophil and macrophage infiltration, enhanced keratinocyte proliferation and migration, accelerated wound re-epithelialization, decreased accumulation of fibroblasts, and decreased collagen production. This study concluded that CB2 agonists may aid in wound healing by decreasing inflammation, accelerating re-epithelialization, and decreasing scar formation.
Due to the ubiquitous availability of over-the-counter cannabidiol products, clinicians would be wise to ask all patients about their use, as drug-drug interactions are not well-studied at this time.
Conclusion
There are many emerging treatments for hypertrophic scarring. Consideration of clinical evidence is helpful in guiding the use of currently available options, with deference to patient desires. Future investigations may help bolster clinical evidence, as randomized trials are generally lacking on this topic. Currently, there is a need to develop a consistently successful regimen to be regarded as a standard by which to treat. The most significant limiting factor to determining a standardized treatment protocol appears to be lack of prospective randomized controlled trials with large sample sizes or studies with unbiased results. It is recommended that additional research be performed to determine the best method or gold standard for nonsurgical scar treatment.
Laura-Ashley O’Connell, DPM, is a resident physician at the Chino Valley Medical Center in Chino, California.
Adrienne Estes, DPM, MS, AACFAS, FAPWHc, is Assistant Professor, Department of Podiatric Medicine and Surgery at Western University of Health Sciences in Pomona, California.
David Shofler, DPM, MSHS, is Assistant Professor, Department of Podiatric Medicine and Surgery at Western University of Health Sciences in Pomona, California.
LER is proud to partner with the American College of Podiatric Medicine to present clinically relevant peer-reviewed content, curated by Jarrod Shapiro, DPM, FACFAS, FACPM, FFPM RCPS (Glasg).
- Gauglitz G, Korting H, Pavicic, Ruzicka T, Jeschke M. Hypertrophic scarring and keloids: pathomechanisms and current and emerging treatment strategies. Mol Med. 2011;17(1-2):113-125.
- Atiyeh B. Nonsurgical management of hypertrophic scars: evidence-based therapies, standard practices, and emerging methods. Aesth Plast Surg. 2007;31(5):468-492.
- Rabello F, Souza C, Farina J. Update on hypertrophic scar treatment. Clinics. 2014;69(8):565-573.
- Davidson S, Aziz N, Rashid R, Khachemoune A. A primary care perspective on keloids. Medscape J Med. 2009;11(1):18.
- Lee H, Jung Y. Recent understandings of biology, prophylaxis and treatment strategies for hypertrophic scars andeloids. Int J Molec Sci. 2018; 19(3):711-730.
- Armour A, Scott P, Tredget E. Cellular and molecular pathology of HTS: basis for treatment. Wound Repair Regen. 2007;15(Suppl 1):S6-S17.
- Candy L, Cecilia L, Ping Z. Effect of different pressure magnitudes on hypertrophic scar in a Chinese population. Burns. 2010;36(8):1234-1241.
- MacIntyre L, Baird M. Pressure garments for use in the treatment of hypertrophic scars—a review of the problems associated with their use. Burns. 2006;32(1):10-15.
- Powell B, Haylock C, Clarke J. A semi-rigid transparent face mask in the treatment of post-burn hypertrophic scars. Br J Plast Surg. 1985;38(4):561-566.
- Engrav L, Heimbach D, Rivara F.. 12-year within-wound study of the effectiveness of custom pressure garment therapy. Burns. 2010;36(7):975-983.
- Gold M, Foster T, Adair M, Burlison K, Lewis T. Prevention of hypertrophic scars and keloids by the prophylactic use of topical silicone gel sheets following a surgical procedure in an office setting. Dermatol Surg. 2001;27(7):641-644.
- Signorini M, Clementoni MT. Clinical evaluation of a new self-drying silicone gel in the treatment of scars: a preliminary report. Aesthetic Plastic Surgery. 2007;31(2):183-187.
- Juckett G, Hartman-Adams H. Management of keloids and hypertrophic scars. Am Fam Physician. 2009;80(3):253-260.
- Manuskiatti W, Fitzpatrick R. Treatment Response of Keloidal and Hypertrophic Sternotomy Scars: comparison among intralesional corticosteroid, 5-fluorouracil, and 585-nm flashlamp-pumped pulsed-dye laser treatments. Arch Dermatol. 2002;138(9):1149-1155.
- Chowdri N, Matoo M, Darzi M. Keloids and hypertrophic scars: results with intraoperative and serial postoperative corticosteroid injection therapy. Aust N Z J Surg. 1999;69(9):655-659.
- Waibel J, Wulkan A, Schumaker P. Treatment of hypertrophic scars using laser and laser assisted corticosteroid delivery. Lasers Surg Med. 2013;45(3):135-140.
- Baumann L, Spencer J. The effects of topical vitamin E on the cosmetic appearance of scars. Dermatol Surg. 1999;25(4):311-315.
- Kabel AM, Moharm FM. Insights into dermatological applications and cutaneous toxicities of bleomycin. M J Dermatol. 2017;2(1):010 .
- Saray Y, Gulec A. Treatment of keloids and hypertrophic scars with dermojet injections of bleomycin: a preliminary study. Int J Dermatol. 2005;44(9):777-784.
- Aggarwal H, Saxena A, Lubana P, Mathur R, Jain D. Treatment of keloids and hypertrophic scars using bleom. J Cosmet Dermatol. 2008;7(1):43-49.
- O’Boyle C, Shayan-Arani H, Hamada M. Intralesional cryotherapy for hypertrophic scars and keloids: a review. Scars Burn Heal. 2017;3:2059513117702162.
- Mourad B, Elfar N, Elsheikh S. Spray versus intralesional cryotherapy for keloids. J Dermatolog Treat. 2015;27(3):264-269.
- Xiao Z, Zhang F, Cui Z. Treatment of hypertrophic scars with intralesional botulinum toxin type A injections: a preliminary report. Aesthestic Plast Surg. 2009;33(3):409-412.
- Huang R, Ho C, Tremp M, Xie Y, Li Q, Zan T. Early postoperative application of botulinum toxin type A prevents hypertrophic scarring after epicanthoplasty: a split-face, double-blind randomized trial. Plast Reconstr Surg. 2019;144(4):835-844.
- Kim S, Lee S, Lee J, Jeong H, Suh I. Clinical trial to evaluate the efficacy of botulinum toxin type A injection for reducing scars in patients with forehead laceration. Medicine (Baltimore). 2019;98(34):e16952.
- Shah V, Aldahan A, Mlacker S, Alsaidan M. 5-Fluorouracil in the Treatment of Keloids and Hypertrophic Scars: A Comprehensive Review of the Literature. Dermatol Ther (Heidelb). 2016;6(2):169-183.
- Gupta S, Kalra A. Efficacy and safety of intralesional 5-fluorouracil in the treatment of keloids. Dermatology. 2002;204(2):130-132.
- Hashim P, Cohen J, Pompei D, Goldenberg G. Topical cannabinoids in dermatology. Cutis 2017;100(1):50-52.
- Kim HJ, Kim B, Park BM, et al. Topical cannabinoid receptor 1 agonist attenuates the cutaneous inflammatory responses in oxazolone-induced atopic dermatitis model. Int J Dermatol. 2015;54:E401-E408 .
- Wang L, Zhao R, Li J. Pharmacological activation of cannabinoid 2 receptor attenuates inflammation, fibrogenesis and promotes re-epithelialization during skin wound healing. Eur J Pharmacol. 2016;786:128-136.