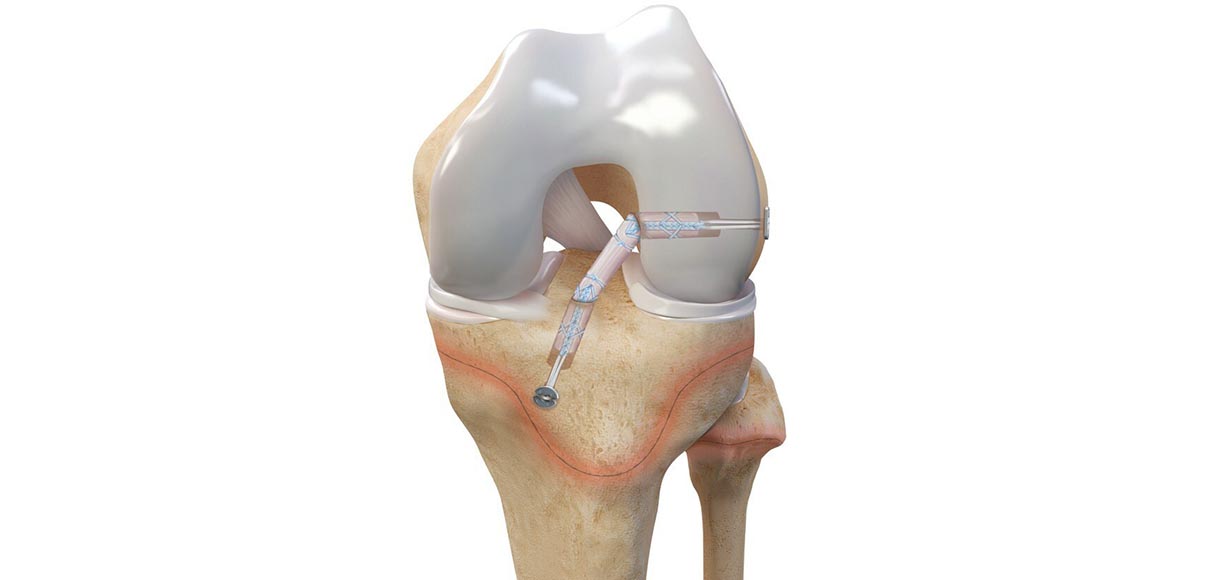
Arthrex’s ACL TightRope implant is a U.S. Food and Drug Administration (FDA)–cleared device for pediatric indications. It is used in the surgical treatment of orthopedic injuries. The company developed all-epiphyseal and transphyseal techniques and instrumentation for ACL surgery alongside top orthopedic surgeons Frank A. Cordasco, MD, MS, and Daniel W. Green, MD, MS, FAAP, FACS. The Arthrex all-epiphyseal technique was developed for skeletally immature patients and involves avoiding the pediatric growth plates to repair or reconstruct the ACL. With the pediatric-specific instrumentation guides, surgeons drill sockets for the new, reconstructed ACL by avoiding the growth plate to diminish the potential for growth disturbance. The Arthrex ACL TightRope portfolio of fixation devices includes the ACL TightRope II implant, the TightRope attachable button system (ABS) and implant, the FiberTag® TightRope implant, and the ACL Repair TightRope implant with FiberRing™ sutures.
Arthrex
800/933-7001