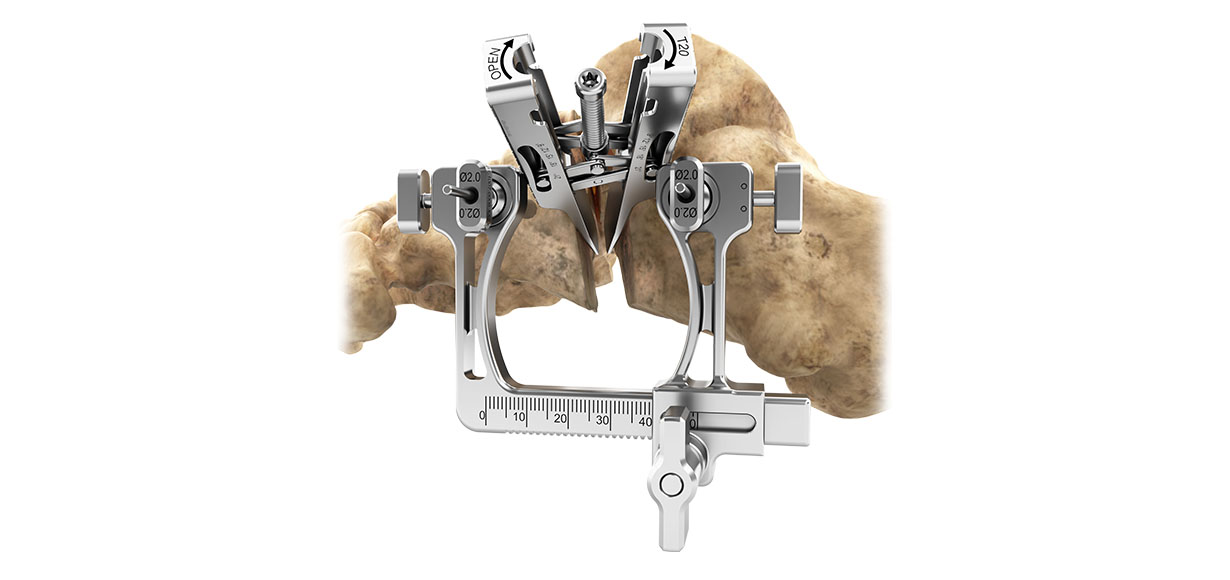
OrthoPediatrics announced the domestic launch of the PediFoot Deformity Correction System following 510(k) clearance by the US Food and Drug Administration. According to the company, this is the first pediatric-specific system to treat fractures and deformities of the foot and ankle. The PediFoot system is designed to focus on lateral column lengthening, calcaneal slide osteotomies, opening and closing wedge osteotomies, and arthrodesis procedures. It offers the smallest plates and screws within the company’s product portfolio, which enables greater effectiveness in addressing cavus foot, flatfoot, clubfoot, and hallux valgus. Patients will benefit from the innovative instrumentation, which flexibly follows the anatomic movement of the bones during the correction process. PediFoot is the first OrthoPediatrics product to feature StarLoc, a variable angle, locking-screw technology. The system also includes PediPedal, a combination of non-slip platforms and handle that assist the surgeon in taking repeatable, simulated weight bearing x-rays during surgery.
OrthoPediatrics Corp.
646/536-7035