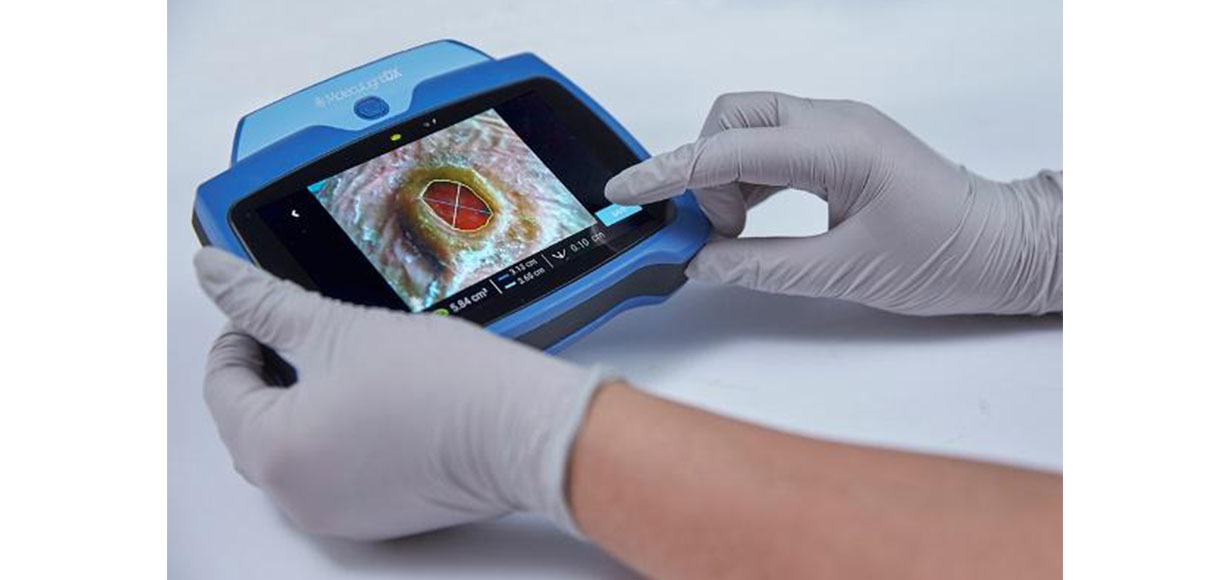
MolecuLightDX™ is a new point-of-care device that enables real-time detection of elevated bacterial burden in wounds. The DX is an expansion of the company’s product line and complements the MolecuLight i:X®. The MolecuLightDX has the following new features: comprehensive electronic medical record (EMR) integration options for multiple EMR environments; patient-centric user interface and workflow to allow for easy patient and wound tracking; an administrator workflow and system configuration capability; and a docking system for easier charging of the device. As with the i:X, the DX has the same accurate, rapid digital wound measurement for documentation of procedures and of wound progression. Newly available on the DX is a stickerless measurement capability, which automatically measures wound area without the need for wound stickers. The DX has received FDA clearance for sale in the U.S., and benefits from the reimbursement pathway available for the MolecuLight procedure.
MolecuLight
647/362-4684
moleculight.com