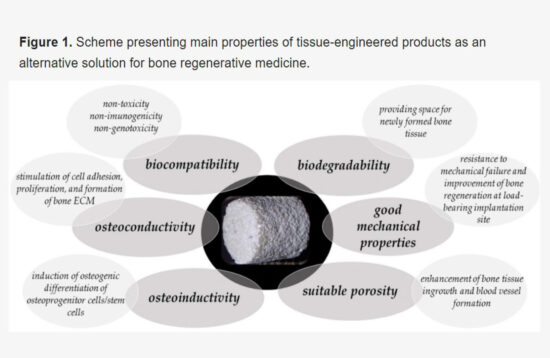
Reprinted from: Kazimierczak P, Przekora A. Osteoconductive and Osteoinductive Surface Modifications of Biomaterials for Bone Regeneration: A Concise Review. Coatings. 2020; 10(10):971. https://doi.org/10.3390/coatings10100971.
By Windy Cole, DPM, CWSP
Bone repair and regeneration (osteogenesis) relies on a series of intricate events supported by the processes of osteoinduction and osteoconduction. Osteoinduction is the mechanism by which osteogenesis is induced. At this stage, immature cells develop into preosteoblasts. Most bone healing is heavily reliant on osteoinduction, whereas osteoconduction is the step in which osteoprogenitor cells and neovascularization propagate into and adhere to a scaffold.
Keeping these processes in mind, orthobiologics that support bone repair, such as bone grafts and bone substitutes, have been developed. Although the use of these orthobiologics in foot and ankle surgery is widely regarded as standard-of-care, there remains a paucity of high-level evidence by way of randomized controlled trials and head-to-head studies. Most evidence exists around the use of allogenic bone grafts employed for use in primary arthrodesis and osteotomies. Outcomes reports are similar to those of autogenic bone grafts with fewer complications. While autogenic bone grafts may still be considered gold standard, complications such as donor site complications, infection, and pain do occur. Available studies on allogenic bone grafts have reported similar fusion rates and outcomes compared to autografts.
There are also a variety of commercially available bone substitutes that have shown a level of utility in foot and ankle surgery. Calcium phosphate, calcium sulphate, and hydroxyapatite can be used to fill bone voids, maintain reduction, and promote bone union. Additionally, demineralized bone matrix (DBM) is yet another type of orthobiologic on the market with utility in foot and ankle surgery. Let’s take a closer look at the uses, advantages, and disadvantages of these products.
Allografts
These are commercially available in cortical, cortical-cancellous, and cancellous forms. Cortical and cortical-cancellous grafts are best used if structural support is needed. Cancellous grafts have utility in filling defects and provide minimal structural support. These products are commercially available fresh, fresh-frozen, freeze dried, and demineralized. New advances in product processing have lessened the likelihood of disease transmission while also decreasing the chances of graft rejection. The primary use of allografts in foot and ankle surgery are in arthrodesis and osteotomies. Ease of use, wide availability, and high structural integrity are some of the advantages of allografts. While the main disadvantage is their relatively slow incorporation.
Bone Substitutes
Bone substitutes such as calcium phosphates and calcium sulphates are available in powders, pastes, granules, and blocks. These products have wide utility in foot and ankle surgery and can be used to fill bone voids, support fracture reductions, augment osteotomies, and promote unions in fusions.
Calcium Phosphate (CP) – resembles human cancellous bone having similar pore size to support osteoconduction. These products are rapidly resorbed (10-12 weeks). CP is biocompatible with the human body and comes sterile for use in the OR. There are a variety of brands on the market such as Skelite (EBI), Vitross (Orthovita), and Chron Os (DePuy).
Calcium Sulfate (CS) – is also osteoconductive, but has greater density than CP. Therefore, CS is better suited for use with fixation. Another advantage is that antibiotics can be added to CS allowing for use in the presence of osteomyelitis. Example product: Osteoset (Wright Medical).
Hydroxyapatite
These products are derived from natural coralline or synthetic calcium carbonate. They serve as an inorganic porous scaffold to support osteoconduction. The most common use is for back-filling bone voids. Advantages include the non-immunogenic nature of the product, but hydroxyapatites have minimum structural strength and are slow to degrade.
Demineralized Bone Matrix (DBM)
DBM is composed of processed bone allograft containing a collagen matrix. It is believed that DBM retains osteoconductive properties and has osteoinductive potential. There remain large variations in product processing based on the manufacturer. DBMs have received mixed reviews when used in foot and ankle surgery. There is a paucity of high-level evidence on DBMs and additional head-to-head trials are needed.
Conclusion
Allografts and bone substitutes are still the front runners when it comes to foot and ankle surgery. These products have a long track record of addressing bone voids and optimizing structural integrity of fracture fixation or fusions. There does not appear to be an orthobiologic product currently available that can simultaneously support ostoegenicity, osteo-inductivity, osteo-conductivity, and mechanical stability. Perhaps as advancements in tissue engineering arise, a new product that checks all of these boxes will emerge.
Windy Cole, DPM, CWSP, is an adjunct professor and Director of Wound Care Research at Kent State University College of Podiatric Medicine. Dr Cole also serves as the National Director of Professional Development and Clinical Education for Woundtech. She is board certified by the American Board of Foot and Ankle Surgery and the American Board of Wound Management. She is a wound care advocate on the forefront of wound research and was the 2020 World Union of Wound Healing Silver Medal Award recipient for her work in Technology-driven Research. And she is a member of the LER Editorial Advisory Board.
- Albrektsson T, Johansson C. Osteoinduction, osteoconduction and osseointegration. Eur Spine J. 2001;10 Suppl 2(Suppl 2):S96-101. doi: 10.1007/s005860100282.
- Wee J, Thevendran G. The role of orthobiologics in foot and ankle surgery: Allogenic bone grafts and bone graft substitutes. EFORT Open Reviews. 2017:2(6),272-280.







