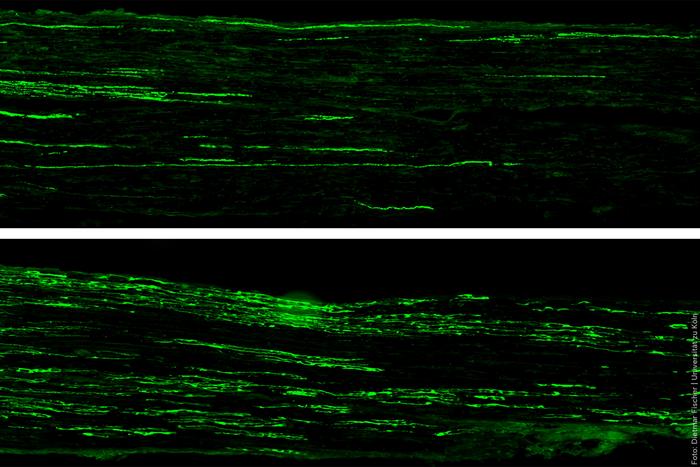
Longitudinal sections of 2 injured nerves with regenerating nerve fibers. Both specimens are from diabetic animals; in the lower image, the animal was treated with a peptide. Regeneration can be seen in the green-stained nerve fibers. Image courtesy of Dietmar Fischer.
A research team led by Professor Dr. Dietmar Fischer, professor of pharmacology at the University of Cologne’s Faculty of Medicine, and director of the Center for Pharmacology at University Hospital Cologne, both in Germany, has identified a central mechanism that explains limited regeneration in diabetes. Building on this, the researchers have developed a promising therapeutic approach that can be used to increase regeneration.
Using mouse models of type 1 and type 2 diabetes mellitus, the team demonstrated a high accumulation of the p35 protein in nerve cells. This protein activates an enzyme that triggers a signaling cascade, which in turn blocks the regrowth of nerve fibers. This considerably restricts the nerves’ natural regenerative capacity. Through targeted interventions in this signaling pathway–either using genetic methods or, pharmacologically, with newly developed small protein building blocks (peptides) that can be administered systemically–the scientists succeeded in removing the block. In the preclinical models, the nerve fibers then grew again at a similar rate to that observed in healthy animals. This was accompanied by significant motor and sensory improvements.
The study thus opens up new perspectives for the treatment and, potentially, the prevention of diabetic neuropathy.






