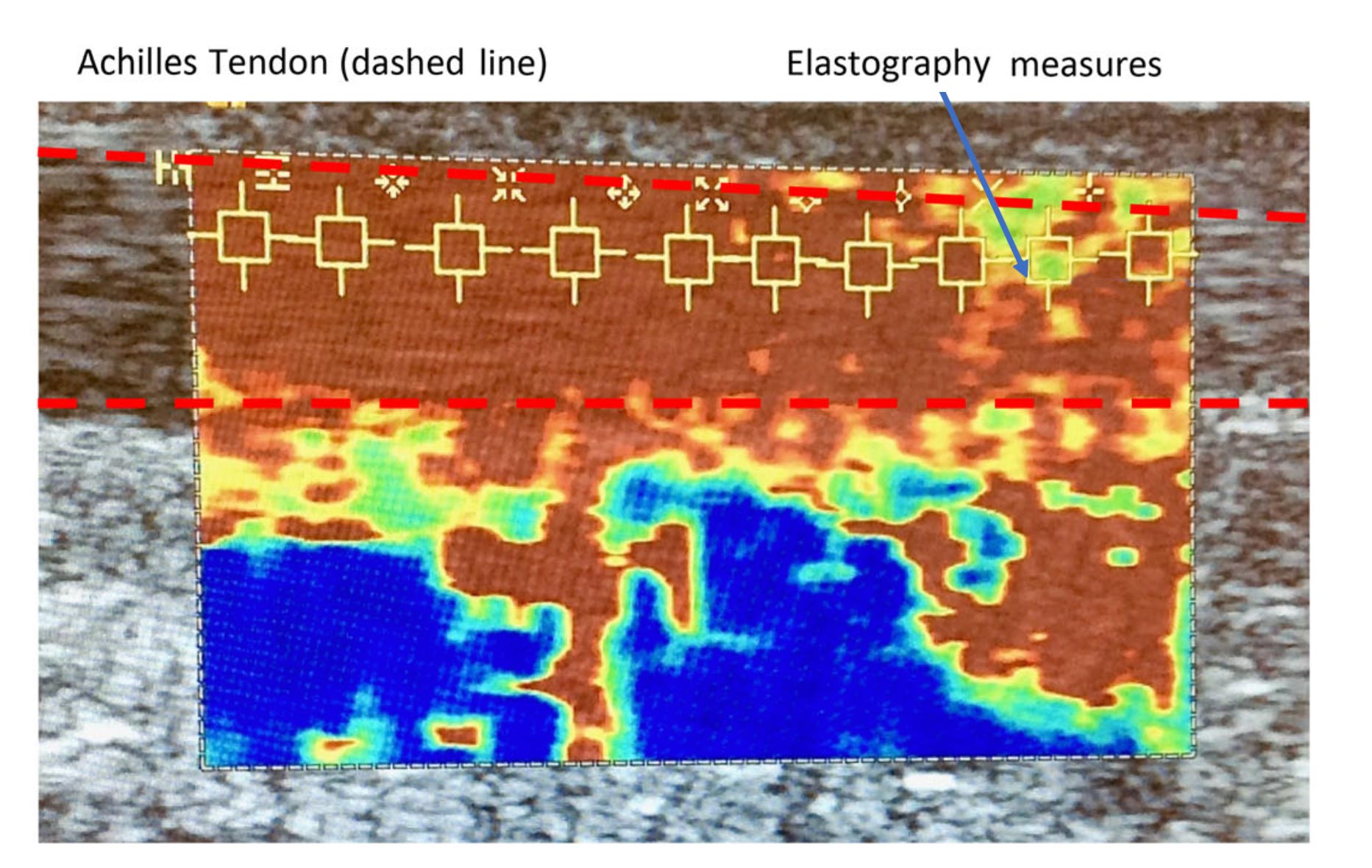
Inflammation associated with gout may change tissue elasticity. Ultrasound imaging using shear wave elastography (SWE) offers a non-invasive method of quantifying changes in tendon stiffness. These authors sought to determine differences in Achilles tendon stiffness in people with gout compared to controls (non-gout) using SWE.
By Simon Otter, Catherine Payne, Anna-Marie Jones, Nick Webborn, and Peter Watt
Gout has been associated with weaker foot/leg muscles and altered gait patterns. There is also evidence of on-going foot pain and an increased risk of tendinopathy, with the Achilles and patella tendons most frequently affected in gout. Additionally, the inflammation associated with gout may change tissue elasticity. We sought to determine differences in Achilles tendon stiffness in people with gout compared to controls (non-gout).
Methods
A cross sectional pilot study comparing 24 people with gout and 26 age/sex-matched controls. Clinical and demographic data were collated, and ultrasound (US) imaging used to determine tendon thickness, presence of gouty tophi and/or aggregates and levels of angiogenesis. Ten shear wave elastography (SWE) measures were taken along the center of a longitudinal section of the mid-portion of each Achilles tendon. [SWE is a non-invasive and dynamic method of quantifying changes in tendon elasticity, involving application of a focused ultrasound pulse to the tissue being imaged.] Data were summarized using descriptive statistics and a repeated measures ANCOVA was used to compare SWE measures between the two groups for the left and right foot separately after accounting for Body Mass Index.
Results
A small proportion of those with gout presented with intra-tendon aggregates and/or intra-tendon tophi in one or both tendons. There was no statistically significant difference in tendon thickness between groups. Neovascularity was present in a third of gout participants. SWE findings demonstrated significantly reduced tendon stiffness in those with gout compared to controls: right Achilles mdiff =1.04 m/s (95% CI (0.38 to 1.7) P = 0.003 and left Achilles mdiff = 0.7 m/s (95% CI 0.09 to 1.32) P = 0.025. No relationship between the presence of tophi and SWE values were detected.
Discussion
Using shear wave elastography, we have for the first time, demonstrated that people with gout exhibit significantly lower stiffness in the AT compared with age/sex matched non-gout participants. Previously, Aubrey and colleagues reported that those with mid-portion AT tendinopathy demonstrated significantly lower mean SWE velocities than did those with a non-pathological AT. The loss of Achilles tendon stiffness we identified in those with gout was often greater than that demonstrated using a similar protocol in otherwise healthy runners with known Achilles tendinopathy. The mean SWE values determined by Payne et al ranged from 7.91m/s – 9.56m/s compared with mean SWE values in our participants with gout of 4.75 m/s – 10 m/s. Recent work suggests Achilles tendinopathy in healthy subjects involves both thickening of the tendon and softening (as reflected in lower SWE values), however our participants with gout did not report symptoms of AT.
Tendon pathology in the lower limbs has been shown to be more frequent in those with gout than in those with either osteoarthritis or healthy controls. A systematic review and meta-analysis revealed that abnormal US findings (i.e. tendon thickening, increased vascularity, hypoechogenicity) in the AT were predictive of tendinopathy. However in our findings from those with gout, tendon thickening did not appear to be present, nor were considerably higher intra-tendon power Doppler readings noted, possibly suggesting a different pathological model from standard tendinopathy. A previous US study demonstrated a much higher prevalence of intra-tendinous tophi (73%) than in non-gout participants. Monosodium urate (MSU) deposition has been reported in 15% – 48% of the AT using dual energy CT scanning techniques. Our conventional US findings were toward the upper end of these results, but not as high as Carroll et al, which may reflect different population sampling strategies.
Previous studies have reported weaker foot/leg muscles in those with gout than in non-gout participants. This muscle weakness was also associated with greater levels of foot pain. Those with gout also exhibited slower walking speeds than control participants. Current international recommendations for gout management highlight the importance of diet together with lifestyle changes such as increased levels of physical activity. In 2008, the UK National Institute for Health and Care Excellence (NICE) produced management recommendations for osteoarthritis that focused on the lower limb and emphasized that muscle strengthening should be undertaken before increasing aerobic activity.
A key strength of our work is that our participants were broadly representative of patients with gout treated in primary care. None were experiencing symptoms of an acute gout flare, nor Achilles tendinosis, yet significant loss of tendon stiffness was demonstrated.
Conclusions
Subjects with gout demonstrated significantly reduced Achilles tendon stiffness compared to non-gout controls. From a clinical standpoint, our SWE findings for the AT in those with gout were similar to SWE measurements in subjects with Achilles tendinopathy who did not have gout. This finding suggests the AT in people with gout may be less effective at transmitting muscle force and potentially more susceptible to further pathology and injury.
This excerpt is from Otter S, Payne C, Jones AM, et al. Differences in Achilles tendon stiffness in people with gout: a pilot study. BMC Musculoskelet Disord. 2020;21:658. Editing has occurred; references were deleted due to space limitations. Use is per the Creative Commons license 4.0. Readers are encouraged to read the full article online at https://doi.org/10.1186/s12891-020-03598-3.
Clinical Trials in Gout
There are currently 30 clinical trials registered with ClinicalTrials.gov recruiting patients with gout. Of the 30, 12 are in recruiting in the United States. Trials are examining an array of potential therapies including SEL-212, adrenocorticotropic hormone (ACTH), pegloticase, pegloticase with methotrexate, tart cherry extract, and SAP-001. Other trials are looking at the pharmacokinetics and pharmacodynamics of drugs in gout, gut biota and probiotics, the value of DECT scans, the role of regulatory B cells in inflammatory rheumatisms, colchicine, and the evolution of tophus.
For more information, visit clinicaltrials.gov and search Gout. You can add various filters to narrow your search.
ClinicalTrials.gov is a Web-based resource that provides patients, their family members, health care professionals, researchers, and the public with easy access to information on publicly and privately supported clinical studies on a wide range of diseases and conditions. The Web site is maintained by the “https://www.nlm.nih.gov/” National Library of Medicine (NLM) at the “https://www.nih.gov/” National Institutes of Health (NIH). Information on ClinicalTrials.gov is provided and updated by the sponsor or principal investigator of the clinical study. Studies are generally submitted to the Web site (that is, registered) when they begin, and the information on the site is updated throughout the study.